

有机介质中脂肪酶催化N-丙基曲克芦丁酰胺类似物的合成研究
收稿日期: 2013-07-15
修回日期: 2013-09-25
网络出版日期: 2013-10-25
基金资助
国家自然科学基金(No.21172055)资助项目.
Synthesis of Novel Fatty Amide Analogues of Troxerutin Catalyzed by Lipase in Organic Medium
Received date: 2013-07-15
Revised date: 2013-09-25
Online published: 2013-10-25
Supported by
Project supported by the National Natural Science Foundation of China (No. 21172055).
王宏雁 , 杨亮茹 , 肖咏梅 , 毛璞 , 朱晓亮 , 袁金伟 , 屈凌波 . 有机介质中脂肪酶催化N-丙基曲克芦丁酰胺类似物的合成研究[J]. 有机化学, 2014 , 34(2) : 355 -361 . DOI: 10.6023/cjoc201307022
In nonaqueous medium, a series of troxerutin amidation analogues were synthesized by the reaction of vinyl-troxerutin esters with fatty amines. The influence parameters including enzyme source, the amount of the enzyme, reactive medium, water content of solvents and reaction time were investigated. The results showed that the amidation occurred regioselectively at vinyl-ester bond. Using pyridine as solvent (water content <1%), the amidation reaction catalyzed by lipase LS-10 (20 mg/mL) reached plateau after 21 h at 50 ℃.
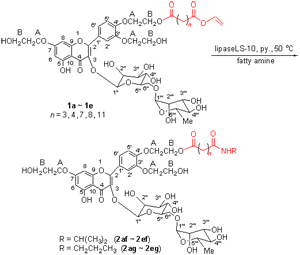
Key words: amidation; troxerutin; enzymatic synthesis; fatty amine; regioselectivity
[1] Chen, A. Y.; Chen, Y. C. Food Chem. 2013, 138, 2099.
[2] Zeng, S.; Liu, W.; Nie, F. F.; Zhao, Q.; Rong, J. J.; Wang, J.; Tao, L.; Qi, Q.; Lu, N.; Li, Z. Y.; Guo, Q. L. Biochem. Biophys. Res. Commun. 2009, 385, 551.
[3] Suresh Babu, K.; Hari Babu, T.; Srinivas, P. V.; Hara Kishore, K.; Murthy, U. S. N.; Madhusudana Rao, J. Bioorg. Med. Chem. Lett. 2006, 16, 221.
[4] Li, H. Q.; Shi, L.; Li, Q. S.; Liu, P. G.; Luo, Y.; Zhao, J.; Zhu, H. L. Bioorg. Med. Chem. 2009, 17, 6264.
[5] Shin, J. S.; Kim, K. S.; Kim, M. B.; Jeong, J. H.; Kim, B. K. Bioorg. Med. Chem. Lett. 1999, 9, 869.
[6] Biasutto, L.; Marotta, E.; De Marchi, U.; Zoratti, M.; Paradisi, C. J. Med. Chem. 2007, 50, 241.
[7] Sun, X.; Hu, C. Q.; Huang, X. D.; Dong, J. C. Chin. J. Org. Chem. 2003, 23, 81 (in Chinese).
(孙逊, 胡昌奇, 黄晓东, 董纪昌, 有机化学, 2003, 23, 81.)
[8] Babu, T. H.; Rao, V. R. S.; Tiwari, A. K.; Babu, K. S.; Srinivas, P. V.; Ali, A. Z.; Rao, J. M. Bioorg. Med. Chem. Lett. 2008, 18, 1659.
[9] Liu, T.; Hu, Y. Z. Chin. J. Org. Chem. 2006, 26, 983 (in Chinese).
(刘滔, 胡永洲, 有机化学, 2006, 26, 983.)
[10] Zhang, S. X.; Ma, J. G.; Bao, Y. M.; Yang, P. W.; Zou, L.; Li, K. J.; Sun, X. D. Bioorg. Med. Chem. 2008, 16, 7127.
[11] Xanthakis, E.; Theodosiou, E.; Magkouta, S.; Stamatis, H.; Loutrari, H.; Roussos, C.; Kolisis, F. Pure Appl. Chem. 2010, 82, 1.
[12] Ma, X.; Yan, R.; Yu, S. Q.; Lu, Y. Y.; Li, Z.; Lu, H. H. J. Agric. Food Chem. 2012, 60, 10844.
[13] Al-Maharik, N. I.; Kaltia, S. A.; Mutikainen, I.; Wähälä, K. J. Org. Chem. 2000, 65, 2305.
[14] Zheng, X.; Meng, W. D.; Qing, F. L. Tetrahedron Lett. 2004, 45, 8083.
[15] Lambusta, D.; Nicolosi, G.; Patti, A.; Sanfilippo, C. J. Mol. Catal. B: Enzym. 2003, 22, 271.
[16] Roh, C.; Choi, K. Y.; Pandey, B. P.; Kim, B. G. J. Mol. Catal. B: Enzym. 2009, 59, 248.
[17] Roh, C.; Seo, S. H.; Choi, K. Y.; Cha, M.; Pandey, B. P.; Kim, J. H.; Park, J. S.; Kim, D. H.; Chang, I. S.; Kim, B. G. J. Biosci. Bioeng. 2009, 108, 41.
[18] Mellou, F.; Loutrari, H.; Stamatis, H.; Roussos, C.; Kolisis, F. N. Process Biochem. 2006, 41, 2029.
[19] Salem, J. H.; Humeau, C.; Chevalot, I.; Harscoat-Schiavo, C.; Vanderesse, R.; Blanchard, F.; Fick, M. Process Biochem. 2010, 45, 382.
[20] Nakajima, N.; Ishihara, K.; Itoh, T.; Furuya, T.; Hamada, H. J. Biosci. Bioeng. 1999, 87, 105.
[21] Ziaullah; Bhullar, K. S.; Warnakulasuriya, S. N.; Rupasinghe, H. P. V. Bioorg. Med. Chem. 2013, 21, 684.
[22] Xiao, Y. M., Li, J. T.; Mao, P.; Zhao, Z.; Lin, X. F. Chin. J. Org. Chem. 2010, 30, 551 (in Chinese).
(肖咏梅, 栗俊田, 毛璞, 赵贞, 林贤福, 有机化学, 2010, 30, 551.)
[23] Hernández-Rodríguez, B.; Córdova, J.; Bárzana, E. J. Mol. Catal. B: Enzym. 2009, 61, 136.
[24] Chebil, L.; Anthoni. J.; Humeau, C.; Gerardin, C.; Engasser, J. M.; Ghoul, M. J. Agric. Food Chem. 2007, 55, 9496.
[25] Tongboriboon, K.; Cheirsilp, B.; H-Kittikun, A. J. Mol. Catal. B: Enzym. 2010, 67, 52.
[26] He, W.; Jia, C. S.; Ma, Y. A.; Yang, Y. B.; Zhang, X. M.; Feng, B. A.; Yue, L. J. Mol. Catal. B: Enzym. 2010, 67, 60.
[27] Horchani, H.; Gargouri, A.; Sayari, A. J. Mol. Catal. B: Enzym. 2012, 75, 35.
[28] Gayot, S.; Santarelli, X.; Coulon, D. J. Biotechnol. 2003, 101, 29.
[29] Ardhaoui, M.; Falcimaigne, A.; Ognier, S.; Engasser, J. M.; Moussou, P.; Pauly, G.; Ghoul, M. J. Biotechnol. 2004, 110, 265.
[30] Kontogianni, A.; Skouridou, V.; Sereti, V.; Stamatis, H.; Kolisis, F. N. J. Mol. Catal. B: Enzym. 2003, 21, 59.
[31] Mellou, F.; Lazari, D.; Skaltsa, H.; Tselepis, A. D.; Kolisis, F. N.; Stamatis, H. J. Biotechnol. 2005, 116, 295.
/
| 〈 |
|
〉 |