

3-甲氧基-6-取代-5,6-二氢吡咯[3,4-b]吡啶-1-酮的合成与生物活性
收稿日期: 2013-09-24
修回日期: 2013-10-14
网络出版日期: 2013-11-20
基金资助
国家科技重大新药创制专项(No. 2010ZX09401-404)资助项目.
Synthesis and Biological Activities of 3-Methoxy-6-substituted-5,6-dihydropyrrolo[3,4-b]pyridin-7-ones
Received date: 2013-09-24
Revised date: 2013-10-14
Online published: 2013-11-20
Supported by
Project supported by the National Significant and Special Project of New Created Drugs (No. 2010ZX09401-404).
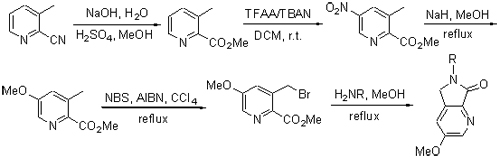
以3-甲基-2-氰基吡啶为原料,经水解、酯化、硝化、甲氧基取代、溴代反应制得中间体3-溴甲基-5-甲氧基吡啶-2-甲酸甲酯(7),然后中间体与不同的有机胺经环合反应得到一系列新的沙利度胺衍生物3-甲氧基-6-取代-5,6-二氢吡咯[3,4-b]吡啶-7-酮1a~1l. 其结构经1H NMR,13C NMR及HRMS确证. 采用MTT(噻唑蓝)法测试了目标化合物抑制HCT-116,MG-63,MCF-7,HUVEC及HMVEC细胞的活性,结果表明,几乎所有化合物对人体正常细胞无明显抑制作用,化合物1h~1l只对MG-63细胞株有明显的抑制活性,化合物1c~1g对这三种肿瘤细胞都有较强的抑制活性,其中化合物1d和1e活性最强.
关键词: 沙利度胺衍生物; 3-甲基-2-氰基吡啶; 合成; 生物活性
孙广龙 , 张蔚蔚 , 詹晓平 , 刘增路 , 毛振民 . 3-甲氧基-6-取代-5,6-二氢吡咯[3,4-b]吡啶-1-酮的合成与生物活性[J]. 有机化学, 2014 , 34(3) : 546 -551 . DOI: 10.6023/cjoc201309033
A series of thalidomide derivatives named 3-methoxy-6-substituted 5,6-dihydropyrrolo[3,4-b]pyridin-7-ones 1a~1l, which have never been reported in literature, were synthesized from 3-methyl pyridine-2-carboxylic acid methyl ester (7) and different amines by cyclization reaction. The intermediate 7 was produced via hydrolysis, esterification, nitration reaction, methoxy substitution, bromination reaction using 3-methyl-pyridine-2-carbonitrile as the starting material. The structures of all compounds have been confirmed by 1H NMR, 13C NMR and HRMS techniques. The target compounds were evaluated for their inhibitory activity against HCT-116, MG-63, MCF-7, HUVEC and HMVEC cells by MTT (thiazolyl blue tetrazolium bromide) method, and the results indicated that almost all of them had no obvious inhibitory effect on normal human cells, compounds 1h~1l only displayed obvious inhibitory effect on MG-63 cells while compounds 1c~1g presented excellent inhibitory activities against all the three kinds of tumor cells, among which compounds 1d and 1e exhibited most potent activities.

[1] D'Amato, R. J.; Loughnan, M. S.; Flynn, E.; Folkman, J. Proc. Natl. Acad. Sci. U. S. A. 1994, 91, 4082.
[2] Dunzendorfer, S.; Schratzberger, P.; Reinisch, N.; Kahler, C. M.; Wiedermann, C. J. Naunyn. Schmiedeberg's Arch. Pharmacol. 1997, 356, 529.
[3] Richardson, P.; Hideshima, T.; Anderson, K. Annu. Rev. Med. 2002, 53, 629.
[4] Chen, L. H.; Li, D. D. Chin. J. New Drugs 2006, 15, 1141 (in Chinese).
(陈立慧, 李电东, 中国新药杂志, 2006, 15, 1141.)
[5] Greig, N. H.; Holloway, H.; Brossi, A.; Zhu, X. X.; Giordano, T.; Yu, Q. S.; Figg, W. D. WO 2005028436, 2005 [Chem. Abstr. 2005, 142, 355170].
[6] Miyachi, H.; Azuma, A.; Ogasawara, A.; Uchimura, E.; Watanabe, N.; Kobayashi, Y.; Kato, F.; Kato, M.; Hashimoto, Y. J. Med. Chem. 1997, 40, 2858.
[7] Man, H. W.; Corral, L. G.; Stirling, D. I.; Muller, G. W. Bioorg. Med. Chem. Lett. 2003, 13, 3415.
[8] Kotla, V.; Goel, S.; Nischal, S.; Heuck, C.; Vivek, K.; Das, B.; Verma, A. J. Hematol. Oncol. 2009, 2, 36.
[9] Kaplan, G.; Sampaio, E. P. US 5385901, 1992 [Chem. Abstr. 1992, 117, 226313].
[10] Kennenth, S. B.; Shannon, C. D.; William, D. F. Biochem. Pharmacol. 1998, 55, 1827.
[11] Fan, B.-L. Ph.D. Dissertation, Shanghai Jiaotong University, Shanghai, 2008 (in Chinese).
(范柏林, 博士论文, 上海交通大学, 上海, 2008.)
[12] Shi, Q.-F. M.S. Thesis, Shanghai Jiaotong University, Shanghai, 2012 (in Chinese).
(史群峰, 硕士论文, 上海交通大学, 上海, 2012.)
[13] Kale, R.; Tayade, P.; Saraf, M.; Juvekar, A. Drug Dev. Ind. Pharm. 2008, 34, 149.
[14] Shibata, Y.; Sasaki, K.; Hashimoto, Y.; Iwasaki, S. Chem. Pharm. Bull. 1996, 44, 156.
[15] Capitosti, S. M.; Hansen, T. P.; Brown, M. L. Bioorg. Med. Chem. 2004, 12, 327.
[16] Zhou, H. Ph.D. Dissertation, Shanghai Jiaotong University, Shanghai, 2013 (in Chinese).
(周恒, 博士论文, 上海交通大学, 上海, 2013.)
[17] Xie, Y.; Wu, G. Q.; Zheng, S. C.; Chen, F. E. Chin. J. Med. Chem. 2006, 16, 49 (in Chinese).
(谢艳, 吾国强, 郑士才, 陈芬儿, 中国药物化学杂志, 2006, 16, 49.)
[18] Zhang, W.-J.; Luo, Y.; Zhang, Y. M. Chin. J. Pharm. 2006, 37, 434 (in Chinese).
(张万金, 罗艳, 张燕梅, 中国医药工业杂志, 2006, 37, 434.)
[19] Njoroge, F. G.; Vibulbhan, B.; Pinto, P.; Chan, T. M.; Osterman, R.; Remiszewski, S.; Rosario, J. D.; Doll, R.; Girijavallabhan, V.; Ganguly, A. K. J. Org. Chem. 1998, 63, 445.
[20] Wang, Y.-H.; Song, Y. J.; Hu, Z.; Jing, J. H.; Meng, X. L.; Huang, Y. D. Chin. J. Org. Chem. 2009, 29, 780 (in Chinese).
(王艳红, 宋元军, 胡 桢, 景介辉, 孟祥丽, 黄玉东, 有机化学, 2009, 29, 780.)
[21] Xie, C.; Runnegar, M. T. C.; Snider, B. B. J. Am. Chem. Soc. 2000, 122, 5017.
[22] Clarke, K.; Goulding, J.; Scrowston, R. M. J. Chem. Soc., Perkin Trans. 1 1984, 1501.
[23] Zhang, E.; Li, C.; Zhang, B. Y.; Lü, A. Q.; Fang, Y.; Feng, S. Q.; Liu, H. M. Chin. J. Org. Chem. 2013, 33, 1100 (in Chinese).
(张恩, 李聪, 张保寅, 吕爱桥, 方园, 冯思琦, 刘宏民, 有机化学, 2013, 33, 1100.)
[24] Mamiko, S.; Yohei, M.; Hiroki, K.; Hiroyuki, K.; Aya, T.; Kazuo, N.; Yuichi, H. Chem. Pharm. Bull. 2003, 51, 1098.
[25] Caroline, S.; Jean-michel, R. J. Enzyme Inhib. Med. Chem. 2008, 23, 659.
[26] Yang, M.; He, J. B.; Cheng, Y. X.; Jiang, S. Chin. J. Org. Chem. 2013, 33, 1319 (in Chinese).
(杨梅, 何江波, 程永现, 蒋晟, 有机化学, 2013, 33, 1319.)
[27] Du, C.; Ren, Y. J.; Wang, Q. W.; Jin, L. Chin. J. Org. Chem. 2013, 33, 1279 (in Chinese).
(杜成, 任玉杰, 王庆伟, 金鹭, 有机化学, 2013, 33, 1279.)
/
| 〈 |
|
〉 |