

2,3-二取代喹唑啉-4(3H)-酮化合物的合成及生物活性研究
收稿日期: 2013-10-16
修回日期: 2013-11-13
网络出版日期: 2013-11-21
基金资助
国家自然科学基金(Nos. 20972186,21172256);国家重点基础研究发展计划(No. 2010CB126104)和绿色生态农药的研发及产业化(No. 2011BAE06B03)资助项目.
Synthesis and Bioactivity Studies of 2,3-Disubstituted Quinazolin-4(3H)-one
Received date: 2013-10-16
Revised date: 2013-11-13
Online published: 2013-11-21
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 20972186, 21172256), the National Basic Research Program of China (No. 2010CB126104) and the Project of Green Pesticide Research, Development and Industrialization (No. 2011BAE06B03).
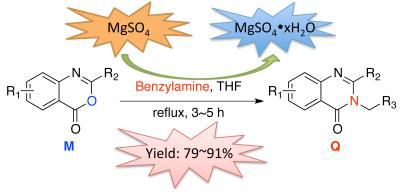
设计合成了一系列2,3-二取代喹唑啉-4(3H)-酮化合物,并研究了常用无机干燥剂对目标化合物合成的影响,发现无水硫酸镁能够促进2,3-二取代喹唑啉-4(3H)-酮化合物的形成. 所有化合物通过核磁共振氢谱、核磁共振碳谱、高分辨质谱等方法进行结构表征,并首次报道了6-氯-3-苄氨基-2-新戊基-8-甲基喹唑啉-4(3H)-酮的单晶结构. 通过生物活性测试发现部分化合物在200 mg/L浓度下对小麦白粉病(Blumeria graminis)具有较好的活体防效.
关键词: 2,3-二取代喹唑啉-4(3H)-酮; 合成; 生物活性; 单晶结构
欧俊军 , 刘克昌 , 王毅 , 张浩 , 刘瑞全 , 李奇博 , 汪清民 , 李永强 , 芮昌辉 , 刘尚钟 . 2,3-二取代喹唑啉-4(3H)-酮化合物的合成及生物活性研究[J]. 有机化学, 2014 , 34(3) : 526 -536 . DOI: 10.6023/cjoc201310022
In this research, a series of new 2,3-disubstituted quinazolin-4(3H)-ones were designed and synthesized. The influence of common drying agents on the formation of 2,3-disubstituted quinazolin-4(3H)-ones was investigated and firstly reported. It is found that the anhydrous magnesium sulfate can increase the yield of 2,3-disubstituted quinazolin-4(3H)-ones significantly. The structures of new compounds were all confirmed via 1H NMR, 13C NMR and HRMS. At the same time, the crystal structure of 3-(benzylamino)-6-chloro-8-methyl-2-neopentyl-quinazolin-4(3H)-one was firstly reported. The results of bioactivity assay showed that some of these new compounds have some control effects on wheat powdery mildew (Blumeria graminis) in vivo at a concentration of 200 mg/L.

[1] Chen, H.-J.; Jiang, Y.-L.; Lin, C.-M. Int. J. Oncol. 2013, 43, 141.
[2] Patel, M. B.; Kumar, S. P.; Valand, N. N. J. Mol. Model. 2013, 19, 3201.
[3] Chao, Q.; Deng, L.; Shih, H. J. Med. Chem. 1999, 42, 3860.
[4] Beaulieu, P. L.; Coulombe, R.; Duan, J. Bioorg. Med. Chem. Lett. 2013, 23, 4132.
[5] Sahoo, B. M.; Dinda, S. C.; Kumar, B. V. V. R. Int. J. Pharm. Sci. Nanotechnol. 2013, 6, 2046.
[6] Abou-Seri, S. M.; Abouzid, K.; Abou El Ella, D. A. Eur. J. Med. Chem. 2011, 46, 647.
[7] Antonelli, A.; Fallahi, P.; Ferrari, S. M. Curr. Genomics 2011, 12, 626.
[8] Morgensztern, D.; Govindan, R. Chemother. Source Book, 4th ed., 2008, p. 191.
[9] (a) Zhou, J.; Fang, J. J. Org. Chem. 2011, 76, 7730.
(b) Sim, Y.-L.; Omer, N.; Khan, M. N. Tetrahedron 2013, 69, 2524.
(c) Chen, Y.; Shan, W.; Lei, M. Tetrahedron Lett. 2012, 53, 5923.
(d) Badri, R.; Alizadeh-Haddad, A.; Adlu, M. Bull. Chem. Soc. Ethiop. 2013, 27, 131.
[10] (a) Gao, X.; Cai, X.; Yan, K. Chin. J. Org. Chem. 2008, 28, 1785 (in Chinese).
(高兴文, 蔡学建, 严凯, 有机化学, 2008, 28, 1785.;
b) Gao, X.; Cai, X.; Yan, K. Molecules 2007, 12, 2621.
[11] Xie, M.; Chen, G.; Zou, L.Chem. Res. Appl. 2010, 864 (in Chinese).
(谢敏, 陈广民, 邹丽娟, 化学研究与应用, 2010, 864.)
[12] (a) Wang, X.; Li, P.; Li, Z. J. Agric. Food Chem. 2013, 61, 9575.
(b) Nanda, A. K.; Ganguli, S.; Chakraborty, R. Molecules 2007, 12, 2413.
(c) Ouyang, G.; Zhang, P.; Xu, G. Molecules 2006, 11, 383.
[13] (a) Liu, J.-F. Curr. Org. Synth. 2007, 4, 223.
(b) Besson, T.; Chosson, E. Comb. Chem. High Throughput Screening 2007, 10, 903.
(c) El-Mekabaty, A. Int. J. Mod. Org. Chem. 2013, 2, 81.
[14] Patil, D. A.; Patil, P. O.; Patil, G. B. Mini-Rev. Med. Chem. 2011, 11, 633.
[15] (a) Salehi, P.; Dabiri, M.; Zolfigol, M. A. Tetrahedron Lett. 2005, 41, 7051.
(b) Baghbanzadeh, M.; Dabiri, M.; Salehi, P. Heterocycles 2008, 11, 2809.
(c) Chen, J.; Wu, D.; He, F. Tetrahedron Lett. 2008, 23, 3814.
(d) Ishikawa, K.; Hosoe, T.; Itabashi, T. Sci. Pharm. 2011, 79, 937.
[16] O'Neil, M. J. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th ed., Merck & Co., Inc., New Jersey, 2006, Monograph Number: 1658, 2653, 5691 and 8680.
[17] (a) Sati, N.; Kumar, S.; Rawat, M. S. M. Indian J. Pharm. Sci. 2009, 71, 572.
(b) Tseng, M.-C.; Chu, Y.-H. Tetrahedron 2008, 64, 9515.
[18] (a) Nerkar, A. G.; Kudale, S. A.; Joshi, P. P. Int. J. Pharm. Pharm. Sci. 2012, 4, 449.
(b) Chikhale, H.; Lade, K.; Joshi, P. Int. J. Pharm. Pharm. Sci. 2012, 4, 466.
[19] Armarego, W. L. F.; Chai, C. L. L. Purification of Laboratory Chemicals, 5th ed., Butterworth Heinemann Press, London, 2003, p. 361, p. 378.
[20] Muller, P.; Herbst, I. R.; Spek, A. L.; Schneider, T. R.; Sawaya, M. R. Crystal Structure Refinement—A Crystallographer's Guide to SHELXL, Oxford University Press, New York, 2006.
/
| 〈 |
|
〉 |