

海洋抗肿瘤环酯肽Apratoxin A研究进展
收稿日期: 2013-10-21
修回日期: 2013-12-04
网络出版日期: 2013-12-06
基金资助
国家自然科学基金(No. 81001392,81172975);复旦大学青年教师科研能力提升项目和国家高技术研究发展计划(863计划,No. 2013AA092903)资助项目.
Research Progress of Apratoxin A:A Marine Cyclicdepsipeptide with Significant Anti-cancer Activity
Received date: 2013-10-21
Revised date: 2013-12-04
Online published: 2013-12-06
Supported by
Project supported by the National Natural Science Foundation of China (No. 81001392, 81172975), the Project to Enhance the Research Ability of Young Teachers (Fudan University), and the National High Technology Research and Development Program of China (863 Program, No. 2013AA092903).
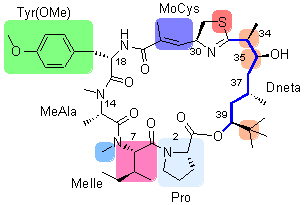
apratoxin A具有新颖复杂的化学结构,是近年来发现的高效广谱抗肿瘤海洋天然环酯肽. 从化学合成、构效关系、作用机制及生物合成角度,对其研究进展作了小结.
关键词: apratoxin A; 海洋活性肽; 抗肿瘤
张伟 , 刘国运 , 尹溽纹 , 李英霞 . 海洋抗肿瘤环酯肽Apratoxin A研究进展[J]. 有机化学, 2014 , 34(3) : 475 -484 . DOI: 10.6023/cjoc201310033
Apratoxin A, a marine natural cyclicdepsipeptide bearing novel and complex structure, showed significant antiproliferative activity against several cancer cell lines. A brief introduction on the research progress of this natural product including total synthesis, structure-activity relationship, pharmacological studies, and biosynthetic pathway was summarized.

Key words: apratoxin A; marine bioactive peptide; anti-cancer
[1] Newman, D. J.; Cragg, G. M.; Snader, K. M. J. Nat. Prod. 2003, 66, 1022.
[2] Wang, C.-Y.; Shao, C.-L. Marine Drugs, Science Press, Beijing, 2011, pp. 1~10 (in Chinese).
(王长云, 邵长伦, 海洋药物学, 科学出版社, 北京, 2011, pp. 1~10.)
[3] Luesch, H.; Yoshida, W. Y.; Moore, R. E.; Paul, V. J.; Corbett, T. H. J. Am. Chem. Soc. 2001, 123, 5418.
[4] Chen, J.; Forsyth, C. J. J. Am. Chem. Soc. 2003, 125, 8734.
[5] Chen, J.; Forsyth, C. J. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 12067.
[6] Doi, T.; Numajiri, Y.; Munakata, A.; Takahashi, T. Org. Lett. 2006, 8, 531.
[7] Ma, D.; Zou, B.; Cai, G.; Hu, X.; Liu, J. O. Chem. Eur. J. 2006, 12, 7615.
[8] Numajiri, Y.; Takahashi, T.; Doi, T. Chem. Asian J. 2009, 4, 111.
[9] Doi, T.; Numajiri, Y.; Takahashi, T.; Takagi, M.; Shin-ya, K. Chem. Asian J. 2011, 6, 180.
[10] Paterson, I.; Wallace, D. J.; Cowden, C. J. Synthesis 1998, 639.
[11] Chen, J.; Forsyth, C. J. Org. Lett. 2003, 5, 1281.
[12] Ma, D. 8th Chinese International Peptide Symposium, Kunming, 2004 (in Chinese).
(马大为, 第八届中国国际多肽学术会议, 昆明, 2004.)
[13] List, B.; Pojarliev, P.; Castello, C. Org. Lett. 2001, 3, 573.
[14] Raman, P.; Razavi, H.; Kelly, J. W. Org. Lett. 2000, 2, 3289.
[15] You, S.; Razavi, H.; Kelly, J. W. Angew. Chem., Int. Ed. 2003, 42, 83.
[16] Xu, Z.; Chen, Z.; Ye, T. Tetrahedron: Asymmetry 2004, 15, 355.
[17] Gilles, A.; Martinez, J.; Cavelier, F. C. R. Chim. 2011, 14, 437.
[18] Tidgewell, K.; Engene, N.; Byrum, T.; Media, J.; Doi, T.; Valeriote, F. A.; Gerwick, W. H. ChemBioChem 2010, 11, 1458.
[19] Thornburg, C. C.; Cowley, E. S.; Silorska, J.; Shaala, L. A.; Ishmael, J. E. Youssef, D. T. A.; McPhail, K. L. J. Nat. Prod. 2013, 76, 1781.
[20] Matthew, S.; Schupp, P. J.; Luesch, H. J. Nat. Prod. 2008, 71, 1113.
[21] Chen, Q.-Y.; Liu, Y.; Luesch, H. ACS Med. Chem. Lett. 2011, 2, 861.
[22] Luesch, H.; Yoshida, W. Y.; Moore, R. E.; Paul, V. J. Bioorg. Med. Chem. 2002, 10, 1973.
[23] Gutiérrez, M.; Suyama, T. L.; Engene, N.; Wignerd, J. S.; Matainaho, T.; Gerwick, W. H. J. Nat. Prod. 2008, 71, 1099.
[24] Luesch, H.; Chanda, S. K.; Raya, R. M.; Dejesus, P. D.; Orth, A. P.; Walker, J. R.; Belmonte, J. C. I.; Schultz, P. G. Nat. Chem. Biol. 2006, 2, 158.
[25] Shen, S.; Zhang, P.; Lovchik, M. A.; Li, Y.; Tang, L.; Chem, Z.; Zeng, R.; Ma, D.; Yuan, J.; Yu, Q. J. Cell Biol. 2009, 185, 629.
[26] Liu, Y.; Law, B. K.; Luesch, H. Mol. Pharm. 2009, 76, 91.
[27] Grindberg, R. V.; Ishoey, T.; Brinza, D.; Esquenazi, E.; Coates, R. C.; Liu, W.; Gerwick, L.; Dorrestein, P. C.; Pevzner, P.; Lasken, R.; Gerwick, W. H. PLoS One 2011, 6, e18565.
/
| 〈 |
|
〉 |