

基于有机小分子的焦磷酸根荧光探针研究进展
收稿日期: 2013-11-26
修回日期: 2013-12-12
网络出版日期: 2013-12-23
基金资助
国家自然科学基金(No. 21272027)、北京市自然科学基金(No. 2122031)和北京市教育委员会共建项目专项资助项目.
Recent Progress in Fluorescent Probes for Pyrophosphate Based on Small Organic Molecules
Received date: 2013-11-26
Revised date: 2013-12-12
Online published: 2013-12-23
Supported by
Project supported by the National Natural Science Foundation of China (No. 21272027), the Beijing Natural Science Foundation (No. 2122031), and the Beijing Municipal Commission of Education.
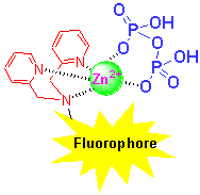
焦磷酸根(PPi)作为一种重要的生物功能阴离子在生命科学、环境科学、药物领域和化学过程等方面起着非常重要的作用. 鉴于荧光分析具有操作简便、灵敏度高等突出优点,设计合成高效的PPi荧光探针成为近年来超分子化学研究的热点之一. 综述了近年来PPi荧光识别与传感的多种设计策略与原理,主要包括基于荧光增强或淬灭型识别,激基缔合物识别,荧光指示剂置换,静电或氢键作用识别等. DPA-金属离子络合物,尤其是DPA-Zn2+络合物,作为识别基团对PPi有着显著的亲和性和选择性识别能力. DPA-Zn2+络合物与多种荧光团或者荧光指示剂组合而形成的化学传感体系已经被广泛应用于PPi荧光识别与传感.
徐勤超 , 金灿 , 朱雪慧 , 邢国文 . 基于有机小分子的焦磷酸根荧光探针研究进展[J]. 有机化学, 2014 , 34(4) : 647 -661 . DOI: 10.6023/cjoc201311043
Pyrophosphate (PPi), a kind of important biological functional anion, plays crucial roles in life science, environmental science, medicine and chemical process. It has attracted extensive interests in supramolecular chemistry to design efficient fluorescent PPi sensors due to the simplicity and high sensitivity of fluorometric analysis. In this review, several kinds of strategies and theories of PPi fluorescent recognition and sensing are summarized, including off-on/on-off fluorescent response, excimer formation, hydrogen-bonding or electrostatic interactions and fluorescent indicator displacement assay. DPA-metal ion complexes, especially DPA-Zn2+ complex, have remarkable binding affinity and high selectivity for PPi. The chemosensing ensembles, consisting of DPA-Zn2+ complex and various fluorophores or fluorescent indicators, have been widely applied to the PPi fluorescent recognition and sensing.

[1] Heinonen, J. K. Biological Role of Inorganic Pyrophosphate, Kluwer Academic Publishers, Norwell, 2001.
[2] Ronaghi, M.; Karamohamed, S.; Pettersson, B.; Uhlén, M.; Nyrén, P. Anal. Biochem. 1996, 242, 84.
[3] Xu, S.; He, M.; Yu, H.; Cai, X.; Tan, X.; Lu, B.; Shu, B. Anal. Biochem. 2001, 299, 188.
[4] (a) Kim, S. K.; Lee, D. H.; Hong, J.-I.; Yoon, J. Acc. Chem. Res. 2009, 42, 23.(b) Hargrove, A. E.; Nieto, S.; Zhang, T.; Sessler, J. L.; Anslyn, E. V. Chem. Rev., 2011, 111, 6603. (c) Ngo, H. T.; Liu, X.; Jolliffe, K. A. Chem. Soc. Rev. 2012, 41, 4928.
[5] Vance, D. H.; Czarnik, A. W. J. Am. Chem. Soc. 1994, 116, 9397.
[6] Lee, D. H.; Im, J. H.; Son, S. U.; Chung, Y. K.; Hong, J.-I. J. Am. Chem. Soc. 2003, 125, 7752.
[7] Jang, Y, J.; Jun, E. J.; Lee, Y. J.; Kim, Y. S.; Kim, J. S.; Yoon, J. J. Org. Chem. 2005, 70, 9603.
[8] Lee, H. N.; Swamy, K. M. K.; Kim, S. K.; Kwon, J.-Y.; Kim, Y.; Kim, S.-J.; Yoon, Y. J.; Yoon, J. Org. Lett. 2007, 9, 243.
[9] Chen, W.-H.; Xing, Y.; Pang, Y. Org. Lett. 2011, 13, 1362.
[10] Kim, M. J.; Swamy, M. K. K.; Lee, K. M.; Jagdale, A. R.; Kim, Y.; Kim, S.-J.; Yoo, K. H.; Yoon, J. Chem. Commun. 2009, 7215.
[11] Das, P.; Bhattacharya, S.; Mishra, S.; Das, A. Chem. Commun. 2011, 47, 8118.
[12] Kim, H. J.; Lee, J. H.; Hong, J.-I. Tetrahedron Lett. 2011, 52, 4944.
[13] Zhang, J. F.; Kim, S.; Han, J. H.; Lee, S.-J.; Pradhan, T.; Cao, Q. Y.; Lee, S. J.; Kang, C.; Kim, J. S. Org. Lett. 2011, 13, 5294.
[14] Jia, J.; Tang, X.; He, Y. F.; Zhang, M. Y.; Xing, G. W. Chin. J. Org. Chem. 2012, 32, 1803 (in Chinese). (郏佳, 唐茜, 何颖芳, 张梦雨, 邢国文, 有机化学, 2012, 32, 1803.)
[15] Su, G.; Liu, Z.; Xie, Z.; Qian, F.; He, W.; Guo, Z. Dalton Trans. 2009, 7888.
[16] Yang, S.; Feng, G.; Williams, N. H. Org. Biomol. Chem. 2012, 10, 5606.
[17] Gu, Z. Y, Xu, Q. C, Xing, G. W. Prog. Chem. 2013, 25, 1302 (in Chinese). (古振远, 徐勤超, 邢国文, 化学进展, 2013, 25, 1302.)
[18] Lee, D. H.; Kim, S. Y.; Hong, J.-I. Angew. Chem., Int. Ed. 2004, 43, 4777.
[19] Roy, B.; Rao, A. S.; Ahn, K. H. Org. Biomol. Chem. 2011, 9, 7774.
[20] Pathberiya, L. G.; Barlow, N.; Nguyen, T.; Graham, B.; Tuck, K. L. Tetrahedron 2012, 68, 9435.
[21] Lee, D.-N.; Jo, A.; Park, S. B.; Hong, J.-I. Tetrahedron Lett. 2012, 53, 5528.
[22] Huang, X.; Guo, Z.; Zhu, W.; Xie, Y.; Tian, H. Chem. Commun. 2008, 5143.
[23] Shao, N.; Wang, H.; Gao, X.; Yang, R.; Chan, W. Anal. Chem. 2010, 82, 4628.
[24] Xu, Q. C; Wang, X. F.; Xing, G. W.; Zhang, Y. RSC Adv. 2013, 3, 15834.
[25] Park, C.; Hong, J.-I. Tetrahedron Lett. 2010, 51, 1960.
[26] Ravikumar, I.; Ghosh, P. Inorg. Chem. 2011, 50, 4229.
[27] Cheng, T.; Wang, T.; Zhu, W.; Chen, X.; Yang, Y.; Xu, Y.; Qian, X. Org. Lett. 2011, 13, 3656.
[28] Cho, H. K.; Lee, D. H.; Hong, J.-I. Chem. Commun. 2005, 1690.
[29] Kim, S. Y.; Hong, J.-I. Bull. Korean Chem. Soc. 2010, 31, 716.
[30] Lee, H. N.; Xu, Z.; Kim, S. K.; Swamy, K. M. K.; Kim, Y.; Kim, S.-J.; Yoon, J. J. Am. Chem. Soc. 2007, 129, 3828.
[31] Wen, J.; Geng, Z.; Yin, Y.; Zhang, Z.; Wang, Z. Dalton Trans. 2011, 40, 1984.
[32] Han, M. S.; Kim, D. H. Angew. Chem., Int. Ed. 2002, 41, 3809.
[33] Han, M. S.; Kim, D. H. Bioorg. Med. Chem. Lett. 2003, 13, 1079.
[34] Hanshaw, R. G.; Hilkert, S. M.; Jiang, H.; Smith, B. D. Tetrahedron Lett. 2004, 45, 8721.
[35] Morgan, B. P.; He, S.; Smith, R. C. Inorg. Chem. 2007, 46, 9262.
[36] Lee, J. H.; Park, J.; Lah, M. S.; Chin, J.; Hong, J. Org. Lett. 2007, 9, 3729.
[37] Huang, F.; Cheng, C.; Feng, G. J. Org. Chem. 2012, 77, 11405.
[38] Surman, A. J.; Bonnet, C. S.; Lowe, M. P.; Kenny, G. D.; Bell, J. D.; Toth, E.; Vilar, R. Chem. Eur. J. 2011, 17, 223.
[39] Nonaka, A.; Horie, S.; Jamesb, T. D.; Kubo, Y. Org. Biomol. Chem. 2008, 6, 3621.
[40] McDonough, M. J.; Reynolds, A. J.; Lee, W. Y. G.; Jolliffe, K. A. Chem. Commun. 2006, 2971.
[41] Butler, S. J.; Jolliffe, K. A. Org. Biomol. Chem. 2011, 9, 3471.
[42] Carolan, J. V.; Butler, S. J.; Jolliffe, K. A. J. Org. Chem. 2009, 74, 2992.
[43] Lee, D. H.; Kim, S. Y.; Hong, J.-I. Tetrahedron Lett. 2007, 48 4477.
[44] Chen, Z.; Lu, Y.; He, Y.; Huang, X. Sens. Actuators, B: Chem. 2010, 149, 407.
[45] Sokkalingam, P.; Kim, D. S.; Hwang, H.; Sessler, J. L.; Lee, C.-H. Chem. Sci. 2012, 3, 1819.
[46] Kim, S. Y.; Hong, J.-I. Tetrahedron Lett. 2009, 50, 1951.
[47] Nishizawa, S.; Kato, Y.; Teramae, N. J. Am. Chem. Soc. 1999, 121, 9463.
[48] Kim, S. K.; Singh, N. J.; Kwon, J.; Hwang, I.-C.; Park, S. J.; Kim, K. S.; Yoon, J. Tetrahedron 2006, 62, 6065.
[49] Anzenbacher, P. Jr; Jursíková, K.; Sessler, J. L. J. Am. Chem. Soc. 2000, 122, 9350.
[50] Lee, J. H.; Jeong, A. R.; Jung, J.-H.; Park, C.-M.; Hong, J.-I. J. Org. Chem. 2011, 76, 417.
[51] (a) Zhu, C.; Liu, L.; Yang, Q.; Lv, F.; Wang, S. Chem. Rev. 2012, 112 4687.(b) Wang, Y.-J.; Gu, Z.-Y., Xing, G. W. Chem. Asian J. 2012, 7, 489.(c) Wang, Y.-J.; Xin, B.-J.; Duan, X.-R.; Xing, G.-W.; Wang, S. Macromol. Rapid Commun. 2010, 31, 1473. Zhao, X.; Schanze, K. S. Chem. Commun. 2010, 46, 6075.
/
| 〈 |
|
〉 |