

磷钨酸在吲哚与磺酰亚胺Friedel-Crafts反应中的应用
收稿日期: 2013-11-11
修回日期: 2013-12-17
网络出版日期: 2014-02-12
基金资助
新疆自治区青年科学基金(No. 2011211B05)资助项目.
Friedel-Crafts Reaction of Indoles with N-Sulfonyl Imines Catalyzed by H3PW12O40
Received date: 2013-11-11
Revised date: 2013-12-17
Online published: 2014-02-12
Supported by
Project supported by the Xinjiang Autonomous Region Youth Science Foundation (No. 2011211B05).
以磷钨酸为催化剂,成功催化了吲哚与磺酰亚胺的Friedel-Crafts反应,并合成了一系列双吲哚甲烷衍生物. 研究结果显示,10 mol%磷钨酸能够很好地催化此反应,收率达到97%. 所合成的双吲哚甲烷衍生物的结构经核磁共振谱和X射线单晶衍射确定.
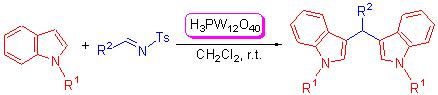
关键词: 吲哚; 亚胺; 磷钨酸; Friedel-Crafts反应
惠永海 , 王长春 , 石伟 , 孟华 , 解正峰 . 磷钨酸在吲哚与磺酰亚胺Friedel-Crafts反应中的应用[J]. 有机化学, 2014 , 34(5) : 898 -902 . DOI: 10.6023/cjoc201311014
H3PW12O40 was found to be effective catalysts for the reactions of indoles with various N-sulfonyl imines to afford the corresponding bis(indolyl)methanes. The results indicated that the good yield (up to 97%) was obtained by using 10 mol% H3PW12O40. The structures of these products were characterized by NMR and X-ray crystallography.
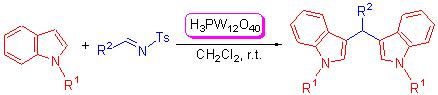
Key words: indoles; imines; H3PW12O40; Friedel-Crafts reaction
[1] Olah, G. A.; Khrisnamurti, R.; Prakash, G. K. S. In Comprehensive Organic Synthesis, Vol. 3, Pergamon, New York, 1991, p. 293.
[2] Peddibhotla, S. Curr. Bioact. Compd. 2009, 5, 20.
[3] Marugan, J. J.; Manthey, C.; Anaclerio, B.; Latrance, L.; Lu, T.; Markotan, T.; Leonard, K. A.; Crysler, C.; Eisennagel, S.; Dasgupta, M.; Tomczuk, B. J. Med. Chem. 2005, 48, 926.
[4] Zhang, H. C.; Ye, H.; Moretto, A. F.; Brumfield, K. K.; Maryanoff, B. E. Org. Lett. 2000, 2, 89.
[5] Poulsen, T. B.; Jørgensen, K. A. Chem. Rev. 2008, 108, 2903.
[6] Morteza, S. Chem. Rev. 2012, 112, 3508.
[7] Chen, Y. C.; Xie, Z. F. Chin. J. Org. Chem. 2012, 32, 452 (in Chinese).
(陈永诚, 解正峰, 有机化学, 2012, 32, 452.)
[8] Shirini, F.; Khaligh, N. G.; Jolodar, O. G. Dyes Pigm. 2013, 98, 290.
[9] Lin, H.; Zang, Y.; Sun, X. W.; Lin, G. Q. Chin. J. Chem. 2012, 30, 2309.
[10] Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F.; Amani, K. Green Chem. 2001, 3, 131.
[11] Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F.; Amani, K. Chem. Commun. 2003, 764.
[12] Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F.; Amani, K. Synthesis 2005, 299.
[13] Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F.; Amani, K. J. Chem. Soc., Perkin Trans. 1 2002, 2601.
[14] Purnima, K. V.; Sreenu, D.; Bhasker, N.; Nagaiah, K.; Lingaiah, N.; Subba Reddy, B. V.; Yadav, J. S. Chin. J. Chem. 2013, 31, 534.
[15] Chen, J. L.; Xie, Z. F.; Xing, Y. Chin. J. Org. Chem. 2011, 31, 1714 (in Chinese).
(陈君丽, 解正峰, 邢烨, 有机化学, 2011, 31, 1714.)
[16] Zhou, G. P.; Yu, L.; Hui, Y. H.; Xie, Z. F. Acta Chim. Sinica 2012, 11, 1289.
[17] Ren, S. Y.; Xie, Z. F.; Cao, L. Q.; Xie, X. P.; Qin, G. F.; Wang, J. D. Catal. Commun. 2009, 10, 464.
[18] Xie, Z. F.; Li, G. L.; Zhao, G.; Wang, J.-D. Chin. J. Chem. 2009, 27, 925.
[19] Osawa, T.; Namiki, M. Tetrahedron Lett. 1983, 24, 4719.
[20] Bandgar, B. P.; Shaikh, K. A. Tetrahedron Lett. 2003, 44, 1959
[21] Li, J. T.; Dai, H. G.; Xu, W. Z.; Li, T. S. Ultras. Sonochem. 2006, 13, 27.
[22] Sadaphal, S. A.; Kategaonkar, A. H.; Labade, V. B.; Shingare, M. S. Chin. Chem. Lett. 2010, 21, 39.
[23] Azizian, J.; Teimouri, F.; Mohammadizadeh, M. R. Catal. Commun. 2007, 8, 1117.
[24] Alonso, I.; Esquivias, J.; Gómez-Arrayás, R.; Carretero, J. C. J. Org. Chem. 2008, 73, 6401.
[25] Hui, Y. H.; Zhang, Q.; Jiang, J.; Lin, L. L.; Liu, X. H.; Feng, X. M. J. Org. Chem. 2009, 74, 6878.
[26] Trost, B. M.; Müller, C. J. Am. Chem. Soc. 2008, 130, 2438. Zwierzak. A.; Napieraj, A. Tetrahedron 1996, 52, 8789.
/
| 〈 |
|
〉 |