

葡萄糖酸溶液中三组分一锅法合成新型β-吲哚衍生物
收稿日期: 2014-06-18
修回日期: 2014-07-09
网络出版日期: 2014-08-29
基金资助
国家科技支撑计划(No.2009BAI75B02)、江西省研究生创新基金(No.YC2014)、江西师范大学优秀博士论文培育计划资助项目.
One-Pot Three-Component Synthesis of New β-Indole Derivatives in Gluconic Acid Aqueous Solution
Received date: 2014-06-18
Revised date: 2014-07-09
Online published: 2014-08-29
Supported by
Project supported by the National Science and Technology Support Program of China (No. 2009BAI75B02), the Innovation Fund Designated for Graduate Students of Jiangxi Province (No. YC2014), and the Outstanding Doctoral Dissertation Cultivation Program of Jiangxi Normal University.
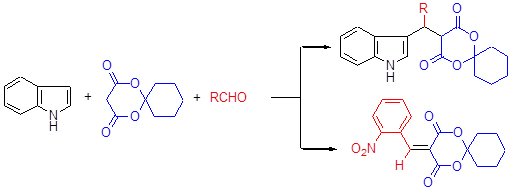
在生物质葡萄糖酸水溶液中, 醛、吲哚和2,2-亚戊基-1,3-二噁烷-4,6-二酮通过"一锅煮"三组分反应合成了13种新型β-吲哚衍生物, 产率为66.4%~98.5%. 该方法具有反应条件温和、底物普适性好、收率高及对环境友好等优点, 为β-取代吲哚类化合物的合成提供了一种有效的新方法.
关键词: 2,2-亚戊基-1,3-二噁烷-4,6-二酮; 葡萄糖酸; 三组分反应; β-吲哚衍生物
严楠 , 夏剑辉 , 熊云奎 , 熊斌 , 林春花 , 廖维林 . 葡萄糖酸溶液中三组分一锅法合成新型β-吲哚衍生物[J]. 有机化学, 2014 , 34(12) : 2487 -2492 . DOI: 10.6023/cjoc201406010
One-pot three-component reactions of aldehydes with indole and 2,2-pentamethylene-1,3-dioxane-4,6-dione afforded thirteen kinds of 5-[(indol-3-yl)-methyl]-2,2-pentamethylene-1,3-dioxane-4,6-dione derivatives (β-indole derivatives) in the presence of gluconic acid aqueous solution in 66.4%~98.5% yields. The advantages of this protocol were mild reaction conditions, wide substrate scope, high yields and benign to environment, which afforded an effective method to synthesize β-indole derivatives.

[1] Yang, D.-L.; Li, J.-R.; Sun, K.-N.; Lu, H.-Y.; Liu, M.-X.; Shi, D.-X. Chin. J. Org. Chem. 2013, 33, 2341 (in Chinese). (杨德利, 李加荣, 孙克宁, 路红燕, 刘明星, 史大昕, 有机化学, 2013, 33, 2341.)
[2] Tong, G.-J.; Fan, W.; Jiang, B. Chin. J. Org Chem. 2013, 33, 2578 (in Chinese). (佟光进, 范威, 姜波, 有机化学, 2013, 33, 2578.)
[3] Dömling, A. Chem. Rev. 2006, 106, 17.
[4] Dömling, A.; Wang, W.; Wang, K. Chem. Rev. 2012, 112, 3083.
[5] Francesco, E.; Salvatore, G.; Ornelio, R.; Silvia, T.; Caroline, P.; Massimo, C. Tetrahedron Lett. 2011, 52, 568.
[6] Csaba, N.; Laurent, J.; Janos, S.-T. Tetrahedron Lett. 2000, 56, 5479.
[7] Stéphane, G.; Bérangère, L.; Antonella, F.; Paolode, M.; Janos, S. Tetrahedron 2010, 66, 3065.
[8] Armstrong, E.-L.; Grover, H.-K.; Kerr, M.-A. J. Org. Chem. 2013, 78, 10534.
[9] Oikawa, Y., Hirasawa, H., Yonemitsu, O. Tetrahedron Lett. 1978, 19(20), 1759.
[10] Wang, H.-Y.; Li, L.-L.; Li, W.; Huang, Z.-B.; Shi, D.-Q. Chin. J. Org. Chem. 2013, 33, 1616 (in Chinese). (汪辉员, 李丽丽, 林伟, 黄志斌, 史达清, 有机化学, 2013, 33, 1616.)
[11] Renzetti, A.; Dardennes, E.; Fontana, A.; Maria, P.-D.; Sapi, J.; Gerard, S. J. Org. Chem. 2008, 73, 6824.
[12] Chandrasekhar, S.; Patro, V.; Reddy, G.-P.; Grée, R. Tetrahedron Lett. 2012, 53, 6223.
[13] Qu, Y.-Y.; Ke, F.; Zhou, L.; Li, Z.-K.; Xiang, H.-F.; Wu, D.; Zhou, X.-G. Chem. Commun. 2011, 47, 3912.
[14] Liu, B.-M.; Zhang, D.-L.; Guo, X.-M.; Li, H.-Z. Chin. J. Synth. Chem. 2004, 12, 505 (in Chinese). (刘卉闵, 张冬暖, 果秀敏, 李惠章, 合成化学, 2004, 12, 505.)
[15] Wang, C.; Zhang, Y.-Q.; Li, G.-S.; Li, J.-C.; Li, X.-L. Chin. J. Org. Chem. 2003, 23, 1416 (in Chinese). (王春, 张英群, 李贵深, 李敬慈, 李晓陆, 有机化学, 2003, 23, 1416.)
[16] Sheng, W.-L.; Du, Y.-Y.; Tian, F.-L.; Han, L.-M.; Zhu, N. Chemistry 2012, 75, 1026 (in Chinese). (盛万里, 杜玉英, 田福利, 韩利民, 竺宁, 化学通报, 2012, 75, 1026.)
[17] Zhou, B.-H.; Yang, J.; Li, M.-H.; Gu, Y.-L. Green Chem. 2011, 13, 2204.
[18] Guo, R.-Y.; Wang, P.; Wang, G.-D.; Mo, L.-P.; Zhang, Z.-H. Tetrahedron 2013, 69, 2056.
[19] Yan, N.; Xiong, B.; Liao, W.-L.; Xu, Z.-H. Chin. J. Org. Chem. 2010, 30, 1391 (in Chinese). (严楠, 熊斌, 廖维林, 许招会, 有机化学, 2010, 30, 1391.)
/
| 〈 |
|
〉 |