

铁催化的N-芳基四氢异喹啉α-sp3-C—H键在空气中的氧化磷酸化
收稿日期: 2014-08-21
修回日期: 2014-09-28
网络出版日期: 2014-10-09
基金资助
国家自然科学基金(No.21302067)和江苏省自然科学基金(No.BK20130120)资助项目.
Iron-Catalyzed Oxidative Phosphonation of α-sp3-C—H Bonds of N-Aryl Tetrahydroisoquinolines with Air as Oxidant
Received date: 2014-08-21
Revised date: 2014-09-28
Online published: 2014-10-09
Supported by
Project supported by the National Natural Science Foundation of China (No.21302067) and the National Natural Science Foundation of Jiangsu Province (No.BK20130120).
张艳 , 罗莎 , 冯柏年 . 铁催化的N-芳基四氢异喹啉α-sp3-C—H键在空气中的氧化磷酸化[J]. 有机化学, 2014 , 34(11) : 2249 -2254 . DOI: 10.6023/cjoc201408025
An efficient cross-dehydrogenative-coupling (CDC) between sp3-C—H bond adjacent to a nitrogen atom of tertiary amine and H—P bonds of dialkyl phosphites and diaryl phosphate was developed using FeCl3 as catalyst and air as oxidant under mild reaction conditions. The safe, convenient, environmental and efficiently benign to synthesize a series of biologically important α-aminophosphonates.
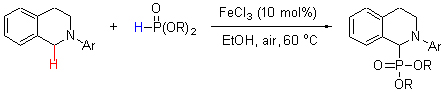
[1] (a) Li, Z.; Li, C. J. J. Am. Chem. Soc. 2005, 127, 3672.(b) Basle, C.; Li, C. J. Green Chem. 2007, 9, 1047. (c) Boess, E.; Sureshkumar, D.; Sud, A.; Wirtz, C.; Fares, C.; Klussmann, M. J. Am. Chem. Soc. 2011, 133, 8106.(d) Xiao, T. B.; Li, L. Y.; Lin, G. L.; Mao, Z. W.; Zhou, L. Org. Chem. 2014, 16, 4232.(e) Zheng, Z. S.; Dian, L. Y.; Yuan, Y. C.; Zhang, N. D.; Du, Y. F.; Zhao, K. J. Org. Chem. 2014, 79, 7451.
[2] (a) Han, W.; Ofial, A. R. Chem. Commun. 2009, 5024.(b) Liu, P.; Liu, Y.; Wong, E. L. M.; Xiang, S.; Che, C. M. Chem. Sci. 2011, 2, 2187.(c) Yu, L.; Wang, M.; Wang, L. Tetrahedron 2014, 70, 5391.
[3] (a) Yoo, W. J.; Li, C. J. Top. Curr. Chem. 2009, 292, 281. (b) Rout, S. K.; Guin, S.; Ali, W.; Gogoi, A.; Patel, B. K. Org. Lett. 2014, 16, 3086. (c) Yoo, W. J.; Kobayashi, S. Green Chem. 2014, 16, 2438.(d) Yan, G. B.; Yu, J.; Zhang, L. Chin. J. Org. Chem. 2012, 32, 294 (in Chinese).(严国兵, 于健, 张玲, 有机化学, 2012, 32, 294.)(e) Liu, W.; Bi, Y. L. Chin. J. Org. Chem. 2012, 32, 1041 (in Chinese). (刘伟, 毕艳兰, 有机化学, 2012, 32, 1041.)(f) Pan, F.; Shi, Z. J. Acta Chim. Sinica 2012, 71, 1679 (in Chinese).(潘菲, 施章杰, 化学学报, 2012, 71, 1679.) (g) Xu, Q.; Zhao, C. Q.; Zhou, Y. B.; Yi, S. F.; Han, L. B. Chin. J. Org. Chem. 2012, 32, 1761 (in Chinese).(徐清, 赵长秋, 周永波, 尹双凤, 韩立彪, 有机化学, 2012, 32, 1761.)
[4] (a) Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215.(b) Sun, C. L.; Li, B, J,; Shi, Z. J. Chem. Rev. 2011, 111, 1293.(c) Zhang, Y.; Luo, S.; Zhu, C. J. Chin. J. Org. Chem. 2012, 32, 2073 (in Chinese).(张艳, 罗莎, 朱成建, 有机化学, 2012, 32, 2073.)(d) Zhang, Y.; Zhu, C. J. Chin. J. Org. Chem. 2012, 32, 2090 (in Chinese).(张艳, 朱成建, 有机化学, 2012, 32, 2090.)
[5] (a) Liu, C.; Zhang, H.; Shi, W.; Li, A. Chem. Rev. 2011, 111, 1780. (b) Li, B. J.; Shi, Z. J. Chem. Soc. Rev. 2012, 41, 5588. (c) Zhang, B.; Guan, H. X.; Liu, B.; Shi, B. F. Chin. J. Org. Chem. 2014, 34, 1487 (in Chinese). (张博, 管晗曦, 刘斌, 史炳锋, 有机化学, 2014, 34, 1487.) (d) Li, H.; Ding, C. H.; Xu, B.; Hou, X. L. Acta Chim. Sinica 2014, 72, 765 (in Chinese).(李浩, 丁昌华, 许斌, 侯雪龙, 化学学报, 2014, 72, 765.)
[6] (a) Mihovilovic, M. D.; Schnuerch, M. ChemCatChem 2014, 6, 2194. (b) Labre, F.; Gimbert, Y.; Bannwarth, P.; Olivero, S.; Dunach, E.; Chavant, P. Y. Org. Lett. 2014, 16, 2366.(c) Xu, T.; Cheung, C. W.; Hu, X. Angew. Chem., Int. Ed. 2014, 53, 4910. (d) Li, Z. P.; Cao, L.; Li. C. J. Angew. Chem., Int. Ed. 2007, 46, 6505. (e) He, R. Y.; Huang, Z. T.; Zheng, Q. Y.; Wang, C. Y. Angew. Chem., Int. Ed. 2014, 53, 4950.
[7] (a) Jia, F.; Li, Z. P. Org. Chem. Front. 2014, 1, 194. (b) Asako, S.; Ilies, L.; Nakamura, E. J. Am. Chem. Soc. 2013, 135, 17755. (c) Chiranjeevi, B.; Koyyada, G.; Prabusreenivasan, S.; Kumar, V.; Sujitha, P.; Kumar, C. G.; Sridhar, B.; Shaik, S.; Chandrasekharam, M. RSC Adv. 2013, 3, 16475. (d) Guo, X. W.; Yu, R.; Li, H. J.; Li, Z. P. J. Am. Chem. Soc. 2009, 131, 17387. (e) Zhou, B. W.; Chen, H.; Wang, C. Y. J. Am. Chem. Soc. 2013, 135, 1264.
[8] (a) Malhotra, S.; Seng, P. S.; Koenig, S. G.; Deese, A. J.; Ford, K. A. Org. Lett. 2013, 15, 3698. (b) Lu, L. Q.; Li, Y. H.; Junge, K.; Beller, M. Angew. Chem., Int. Ed. 2013, 52, 8382. (c) Li, Y. M.; Ma, L. N.; Jia, F.; Li, Z. P. J. Org. Chem. 2013, 78, 5638. (d) Wang, C. Y. Synlett 2013, 24, 1606.
[9] (a) Manabe, K.; Kobayashi, S. Chem. Commun. 2000, 669.(b) Yadav, J. S.; Reddy, B. V. S.; Raj, K. S.; Reddy, K. B.; Prasad, A. R. Synthesis 2001, 2277.
[10] (a) Saidi, M. R.; Azizi, N. Synlett 2002, 1347. (b) Akiyama, T.; Sanada, M.; Fuchibe, K. Synlett 2003, 1463.
[11] (a) Han, W.; Ofial, R. Chem. Commun. 2009, 6023. (b) Hari, D. P.; Konig, B. Org. Lett. 2011, 13, 3852.
[12] (a) Xue, Q. C.; Xie, J.; Jin, H. M.; Cheng, Y. X.; Zhu, C. J. Org. Biomol. Chem. 2013, 11, 1606. (b) Rueping, M.; Zhu, S. Q.; Koenigs, R. M. Chem. Commun. 2011, 47, 8679. (c) Dhineshkumar, J.; Lamani, M.; Alagiri, K.; Prabhu, K. R. Org. Lett. 2013, 15, 1092.
[13] (a) Xie, J.; Li, H. M.; Xue, Q. C.; Cheng, Y. X.; Zhu, C. J. Adv. Synth. Catal. 2012, 354, 1646. (b) Alagiri, K.; Devadig, P.; Prabhu, K. R. Chem. Eur. J. 2012, 18, 5160. (c) Alagiri, K.; Devadig, P.; Prabhu, K. R. Tetrahedron Lett. 2012, 53, 1456. (d) Wang, H. L.; Li, X. C.; Wu, F.; Wan, B. S. Tetrahedron Lett. 2012, 53, 681.
[14] (a) Zhang, Y.; Peng, H.; Zhang, M.; Cheng, Y. X.; Zhu, C. J. Chem. Commun. 2011, 47, 2354. (b) Zhang, Y.; Zhu, C. J. Catal. Commun. 2012, 28, 134.(c) Zhang, Y.; Feng, B. N.; Zhu, C. J. Org. Biomol. Chem. 2012, 10, 9137.
[15] (a) Huo, C. D.; Wang, C.; Wu, M. X.; Jia, X. D.; Yuan, Y.; Xie, H. S. Org. Biomol. Chem. 2014, 12, 3123.(b) Basle O.; Li, C. J. Chem. Commun. 2009, 4124.
[16] Li, Z. P.; Li, C. J. J. Am. Chem. Soc. 2005, 127, 3672.
/
| 〈 |
|
〉 |