

铼催化的醇脱羟基反应研究进展
收稿日期: 2014-09-11
修回日期: 2014-11-11
网络出版日期: 2014-11-20
基金资助
黑龙江省教育厅海外学人科研(No. 1154H14)、黑龙江省教育厅(No. 1154G53)、国家自然科学基金联合基金(No. U1362110)、国家自然科学基金(Nos. 21322203, 21272238)资助项目.
Recent Progress in Re-Catalyzed Dehydroxylation Reactions
Received date: 2014-09-11
Revised date: 2014-11-11
Online published: 2014-11-20
Supported by
Project supported by the Scientific Research Foundation for Overseas Chinese Scholars of Heilongjiang Province (No. 1154H14), the Department of Education of Heilongjiang Province (No. 1154G53), the Joint Funds of National Natural Science Foundation of China (No. U1362110), and the National Natural Science Foundation of China (Nos. 21322203, 21272238).
毛国梁 , 贾冰 , 王从洋 . 铼催化的醇脱羟基反应研究进展[J]. 有机化学, 2015 , 35(2) : 284 -293 . DOI: 10.6023/cjoc201409027
The development of novel effective processes for the conversion of abundant renewable resources to fuels and value-added chemicals has spurred new interest in the discovery of selective chemical transformations of polyols and carbohydrates. These compounds have high oxygen content, which is mostly present in the form of hydroxyl groups. Therefore, partial or complete removal of the hydroxyl groups is of great importance. Herein a brief overview on the development of rhenium-catalyzed dehydroxylation reactions in recent years is given, which are roughly classified into two categories: (1) dehydroxylation reactions of monools for new chemical bond formation and (2) dehydroxylation reactions of diols and polyols for olefin syntheses.
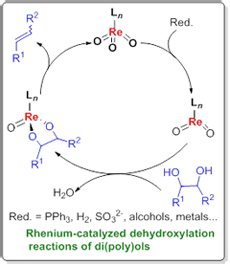
Key words: rhenium catalysis; alcohols; dehydroxylation; reductants; olefins
[1] (a) Cao, X. H. Chem. Ind. Eng. Prog. 2007, 26, 905 (in Chinese). (曹湘洪, 化工进展, 2007, 26, 905.)
(b) Min, E. CIESC J. 2006, 57, 1739 (in Chinese). (闵恩泽, 化工学报, 2006, 57, 1739.)
(c) Vennestrom, P. N. R.; Osmundsen, C. M.; Christensen, C. H.; Taarning, E. Angew. Chem., Int. Ed. 2011, 50, 10502.
[2] Ragauskas, A. J.; Williams, C. K.; Davison, B. H.; Britovsek, G.; Cairney, J.; Eckert, C. A.; Frederik, W. J. Jr.; Hallett, J. P.; Leak, D. J.; Liotta, C. L.; Mielenz, J. R.; Murphy, R.; Templer, R.; Tschaplinski, T. Science 2006, 311, 484.
[3] Werpy, T.; Peterson, G. Top Value Added Chemicals from Biomass: Vol. 1, U. S. Department of Energy, Washington D. C., 2004.
[4] Corma, A.; Iborra, S.; Velty, A. Chem. Rev. 2007, 107, 2411.
[5] For our work, see: (a) Xia, D.; Wang, Y.; Du, Z.; Zheng, Q.-Y.; Wang, C. Org. Lett. 2012, 14, 588.
(b) Wang, Y.; Zhang, L.; Yang, Y.; Zhang, P.; Du, Z.; Wang, C. J. Am. Chem. Soc. 2013, 135, 18048.
(c) Tang, Q.; Xia, D.; Jin, X.; Zhang, Q.; Sun, X.-Q.; Wang, C. J. Am. Chem. Soc. 2013, 135, 4628.
(d) Tang, H.; Zhou, B.; Huang, X.-R.; Wang, C.; Yao, J.; Chen, H. ACS Catal. 2014, 4, 649. For selected work from other groups, see:
(e) Kuninobu, Y.; Takai, K. Chem. Rev. 2011, 111, 1938.
(f) Jin, H.; Xie, J.; Pan, C.; Zhu, Z.; Cheng, Y.; Zhu, C. ACS Catal. 2013, 3, 2195.
(g) Fukumoto, Y.; Daijo, M.; Chatani, N. J. Am. Chem. Soc. 2012, 134, 8762.
(h) Hua, R.; Tian, X. J. Org. Chem. 2004, 69, 5782.
(i) Liu, Q.; Li, Y.-N.; Zhang, H.-H.; Chen, B.; Tung, C.-H.; Wu, L.-Z. J. Org. Chem. 2011, 76, 1444.
[6] Zhu, Z.; Espenson, J. H. J. Org. Chem. 1996, 61, 324.
[7] Sherry, B. D.; Radosevich, A. T.; Toste, F. D. J. Am. Chem. Soc. 2003, 125, 6076.
[8] Liu, Y.; Hua, R. M.; Sun, H. B.; Qiu, X. Q. Organometallics 2005, 24, 2819.
[9] Ohri, R. V.; Radosevich, A. T.; Hrovat, K. J.; Musich, C.; Huang, D.; Holman, T. R.; Toste, F. D. Org. Lett. 2005, 7, 2501.
[10] Abdukader, A.; Jin, H.; Cheng, Y.; Zhu, C. Tetrahedron Lett. 2014, 55, 4172.
[11] Xu, Q.; Li, Q. Chin. J. Org. Chem. 2013, 33, 18 (in Chinese). (徐清, 李强, 有机化学, 2013, 33, 18.)
[12] Luzung, M. R.; Toste, F. D. J. Am. Chem. Soc. 2003, 125, 15760.
[13] Kennedy-Smith, J. J.; Young, L. A.; Toste, F. D. Org. Lett. 2004, 6, 1325.
[14] Kuninobu, Y.; Ishii, E.; Takai, K. Angew. Chem., Int. Ed. 2007, 46, 3296.
[15] Nielsen, M. B.; Diederich, F. Synlett 2002, 544.
[16] Kuninobu, Y.; Ueda, H.; Takai, K. Chem. Lett. 2008, 8, 878.
[17] Korstanje, T. J.; Jastrzebski, J. T. B. H.; Klein Gebbink, R. J. M. ChemSusChem 2010, 3, 695.
[18] Korstanje, T. J.; Waard, E. F.; Jastrzeki, J. T. B. H.; Klein Gebbink, R. J. M. ACS Catal. 2012, 2, 2173.
[19] Cook, G. K.; Andrews, M. A. J. Am. Chem. Soc. 1996, 118, 9448.
[20] Raju, S.; Jastrzebski, J. T. B. H.; Robertus, M. L.; Klein Gebbink, R. J. M. ChemSusChem 2013, 6, 1673.
[21] Ziegler, J. E.; Zdilla, M. J.; Evans, A. J.; Abu-Omar, M. M. Inorg. Chem. 2009, 48, 9998.
[22] Denning, A. L.; Dang, H.; Liu, Z. M.; Nicholas, K. M.; Jentoft, F. C. ChemCatChem 2013, 12, 3567.
[23] (a) Holm, R. H.; Donahue, J. P. Polyhedron 1993, 12, 571.
(b) Lee, S. C.; Holm, R. H. Inorg. Chim. Acta 2008, 361, 1166.
[24] Kaksonen, A. H.; Puhakka, J. A. Eng. Life Sci. 2007, 7, 541.
[25] Vkuturi, S.; Chapman, G.; Ahmad, I.; Nicholas, K. M. Inorg. Chem. 2010, 49, 4744.
[26] Ahmad, I.; Chapman, G.; Nicholas, K. M. Organometallics 2011, 30, 2810.
[27] Arceo, E.; Ellman, J. A.; Bergman, R. G. J. Am. Chem. Soc. 2010, 132, 11408.
[28] Yi, J.; Liu, S.; Abu-Omar, M. M. ChemSusChem 2012, 5, 1401.
[29] Shiramizu, M.; Toste, F. D. Angew. Chem., Int. Ed. 2012, 51, 8082.
[30] Shiramizu, M; Toste, F. D. Angew. Chem., Int. Ed. 2013, 52, 12905.
[31] For selected transformations, see: (a) Bellemin-Laponnaz, S. ChemCatChem 2009, 1, 357.
(b) Morrill, C.; Grubbs, R. H. J. Am. Chem. Soc. 2005, 127, 2842.
(c) Morrill, C.; Beutner, G. L.; Grubbs, R. H. J. Org. Chem. 2006, 71, 7813.
(d) Hansen, E. C.; Lee, D. J. Am. Chem. Soc. 2006, 128, 8142.
(e) Herrmann, A. T.; Saito, T.; Stivala, C. E.; Tom, J.; Zakarian, A. J. Am. Chem. Soc. 2010, 132, 5962.
[32] Jacobs, C. B.; Nicholas, K. M. ChemSusChem 2013, 6, 597.
[33] McClain, J. M.; Nicholas, K. M. ACS Catal. 2014, 4, 2109.
/
| 〈 |
|
〉 |