

氧化偶联反应的最新研究进展
收稿日期: 2014-11-14
修回日期: 2015-01-24
网络出版日期: 2015-01-28
基金资助
科技部973计划(Nos. 2012CB725302, 2011CB808600)、国家自然科学基金(Nos. 21390400, 21272180, 21302148)、高等学校博士学科点专项科研基金资助(No. 20120141130002)、教育部长江学者和创新团队发展计划(No. IRT1030)、国家科学技术部基金(No. 2012YQ120060)、高等学校学科创新引智计划(111项目)资助项目.
Recent Advances in Oxidative Coupling Reactions
Received date: 2014-11-14
Revised date: 2015-01-24
Online published: 2015-01-28
Supported by
Project supported by the 973 Program (Nos. 2012CB725302, 2011CB808600), the National Natural Science Foundation of China (Nos. 21390400, 21272180, and 21302148), and the Research Fund for the Doctoral Program of Higher Education of China (No. 20120141130002), the Program for Changjiang Scholars and Innovative Research Team in University (No. IRT1030), the Ministry of Science and Technology of China (No. 2012YQ120060) and the Program of Introducing Talents of Discipline to Universities of China (111 Program).
张剑 , 陆庆全 , 刘超 , 雷爱文 . 氧化偶联反应的最新研究进展[J]. 有机化学, 2015 , 35(4) : 743 -759 . DOI: 10.6023/cjoc201411028
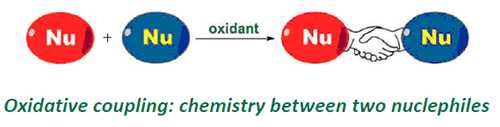
Recently, transition-metal-catalyzed oxidative coupling reactions have emerged as the most important and attractive methods to construct carbon-carbon bond and carbon-heteroatom bonds in organic synthetic chemistry. This review mainly summarizes our research progress in the area of oxidative coupling and their versatility, application in organic synthesis, especially for the third generation of oxidative coupling.
[1] Negishi, E.-I.; de Meijere, A. Handbook of Organopalladium Chemistry for Organic Synthesis, Wiley-Interscience, New York, 2002.
[2] (a) Heck, R. F.; Nolley, J. P. J. Org. Chem. 1972, 37, 2320.
(b) Miyaura, N.; Suzuki, A. J. Chem. Soc., Chem. Commun. 1979, 866.
(c) Milstein, D.; Stille, J. K. J. Am. Chem. Soc. 1979, 101, 4992.
(d) Baba, S.; Negishi, E. J. Am. Chem. Soc. 1976, 98, 6729.
(e) Hatanaka, Y.; Hiyama, T. J. Org. Chem. 1988, 53, 918.
(f) Guram, A. S.; Rennels, R. A.; Buchwald, S. L. Angew. Chem., Int. Ed. 1995, 34, 1348.
[3] (a) Liu, C.; Zhang, H.; Shi, W.; Lei, A. Chem. Rev. 2011, 111, 1780.
(b) Liu, Q.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 10788.
(c) Shi, W.; Liu, C.; Lei, A. Chem. Soc. Rev. 2011, 40, 2761.
(d) Liu, C.; Jin, L. Synlett 2010, 2527.
[4] Lipshutz, B. H.; Siegmann, K.; Garcia, E.; Kayser, F. J. Am. Chem. Soc. 1993, 115, 9276
[5] Zhao, Y.; Wang, H.; Hou, X.; Hu, Y.; Lei, A.; Zhang, H.; Zhu, L. J. Am. Chem. Soc. 2006, 128, 15048.
[6] Jin, L.; Zhao, Y.; Zhu, L.; Zhang, H.; Lei, A. Adv. Synth. Catal. 2009, 351, 630.
[7] Oi, S.; Fukita, S.; Inoue, Y. Chem. Commun. 1998, 2439
[8] Zhao, Y.; Jin, L.; Li, P.; Lei, A. J. Am. Chem. Soc. 2008, 130, 9429.
[9] (a) Luh, T.; Leung, M.; Wong, K. Chem. Rev. 2000, 100, 3187.
(b) Woell, J. B.; Fergusson, S. B.; Alper, H. J. Org. Chem. 1985, 50, 2134.
[10] Frisch, A. C.; Beller, M. Angew. Chem., Int. Ed. 2005, 44, 674.
[11] Liu, Q.; Li, G.; He, J.; Liu, J.; Li, P.; Lei, A. Angew. Chem., Int. Ed. 2010, 49, 3371.
[12] Yoshida, H.; Yamaryo, Y.; Ohshita, J.; Kunai, A. Tetrahedron Lett. 2003, 44, 1541.
[13] (a) Inoue, S.; Shiota, H.; Fukumoto, Y.; Chatani, N. J. Am. Chem. Soc. 2009, 131, 6898.
(b) Guan, Z.; Ren, Z.; Spinella, S. M.; Yu, S.; Liang, Y.; Zhang, X. J. Am. Chem. Soc. 2008, 131, 729.
(c) Giri, R.; Yu, J. J. Am. Chem. Soc. 2008, 130, 14082.
[14] (a) Inoue, S.; Shiota, H.; Fukumoto, Y.; Chatani, N. J. Am. Chem. Soc. 2009, 131, 6898.
(b) Guan, Z. H.; Ren, Z. H.; Spinella, S. M.; Yu, S. C.; Liang, Y. M.; Zhang, X. M. J. Am. Chem. Soc. 2009, 131, 729.
(c) Giri, R.; Yu, J. Q. J. Am. Chem. Soc. 2008, 130, 14082.
(d) Orito, K.; Horibata, A.; Nakamura, T.; Ushito, H.; Nagasaki, H.; Yuguchi, M.; Yamashita, S.; Tokuda, M. J. Am. Chem. Soc. 2004, 126, 14342.
(e) Zhao, Y.; Jin, L.; Li, P.; Lei, A. J. Am. Chem. Soc. 2008, 130, 9429.
[15] (a) Eckhardt, M.; Fu, G. C. J. Am. Chem. Soc. 2003, 125, 13642.
(b) Altenhoff, G.; Wuertz, S.; Glorius, F. Tetrahedron Lett. 2006, 47, 2925.
(c) Cahiez, G.; Gager, O.; Buendia, J. Angew. Chem., Int. Ed. 2010, 49, 1278.
[16] Chen, M.; Zheng, X.; Li, W.; He, J.; Lei, A. J. Am. Chem. Soc. 2010, 132, 4101.
[17] Izawa, Y.; Shimizu, I.; Yamamoto, A. Bull. Chem. Soc. Jpn. 2004, 77, 2033.
[18] Murata, S.; Teramoto, K.; Miura, M.; Nomura, M. J. Chem. Res., Synop. 1993, 434.
[19] Fuchita, Y.; Utsunomiya, Y.; Yasutake, M. J. Chem. Soc., Dalton Trans. 2001, 2330.
[20] Dick, A. R.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 2300
[21] (a) Kamata, K.; Yamaguchi, S.; Kotani, M.; Yamaguchi, K.; Mizuno, N. Angew. Chem., Int. Ed. 2008, 47, 2407.
(b) Eglinton, G.; Galbraith, A. R. Chem. Ind. 1956, 737.
(c) Hay, A. S. J. Org. Chem. 1960, 25, 1275.
(d) Hay, A. S. J. Org. Chem. 1962, 27, 3320.
(e) Ranu, B. C.; Banerjee, S. Lett. Org. Chem. 2006, 3, 607.
(f) Jiang, H.-F.; Tang, J.-Y.; Wang, A. Z.; Deng, G.-H.; Yang, S.-R. Synthesis 2006, 1155.
(g) Yadav, J. S.; Reddy, B. V. S.; Reddy, K. B.; Gayathri, K. U.; Prasad, A. R. Tetrahedron Lett. 2003, 44, 6493.
(h) Nishihara, Y.; Ikegashira, K.; Hirabayashi, K.; Ando, J.-i.; Mori, A.; Hiyama, T. J. Org. Chem. 2000, 65, 1780.
(i) Siemsen, P.; Livingston, R. C.; Diederich, F. Angew. Chem., Int. Ed. 2000, 39, 2632.
[22] (a) Glaser, C. Ber. Dtsch. Chem. Ges. 1869, 2, 422.
(b) Glaser, C. Ann. Chem. Pharm. 1870, 154, 137.
(c) Yang, F.; Cui, X.; Li, Y.-N.; Zhang, J.; Ren, G.-R.; Wu, Y. Tetrahedron 2007, 63, 1963.
(d) Shi, M.; Qian, H.-X. Appl. Organomet. Chem. 2006, 20, 771.
(e) Rossi, R.; Carpita, A.; Bigelli, C. Tetrahedron Lett. 1985, 26, 523.
(f) Qian, M.; Negishi, E. Org. Process Res. Dev. 2003, 7, 412.
(g) Lei, A.; Srivastava, M.; Zhang, X. J. Org. Chem. 2002, 67, 1969.
(h) Chalifoux, W. A.; Ferguson, M. J.; Tykwinski, R. R. Eur. J. Org. Chem. 2007, 1001.
(i) Yan, J.; Lin, F.; Yang, Z. Synthesis 2007, 1301.
[23] (a) Shun, A. L. K. S.; Tykwinski, R. R. Angew. Chem., Int. Ed. 2006, 45, 1034.
(b) Haley, M. M.; Tykwinski, R. R. Carbon-Rich Compounds: From Molecules to Materials, Wiley-VCH, Weinheim, 2006.
(c) Diederich, F.; Stang, P. J.; Tykwinski, R. R. Acetylene Chemistry: Chemistry, Biology and Material Science, Wiley-VCH, Weinheim, 2005.
[24] (a) Kim, S.; Kim, S.; Lee, T.; Ko, H.; Kim, D. Org. Lett. 2004, 6, 3601.
(b) Nishihara, Y.; Ikegashira, K.; Mori, A.; Hiyama, T. Tetrahedron Lett. 1998, 39, 4075.
(c) Montierth, J. M.; DeMario, D. R.; Kurth, M. J.; Schore, N. E. Tetrahedron 1998, 54, 11741.
(d) Alami, M.; Ferri, F. Tetrahedron Lett. 1996, 37, 2763.
[25] Yin, W.; He, C.; Chen, M.; Zhang, H.; Lei, A. Org. Lett. 2008, 11, 709.
[26] (a) Matsumoto, F.; Chujo, Y. Pure Appl. Chem. 2006, 78, 1407.
(b) Butler, P.; Gallagher, J. F.; Manning, A. R.; Mueller-Bunz, H.; McAdam, C. J.; Simpson, J.; Robinson, B. H. J. Organomet. Chem. 2005, 690, 4545.
(c) McAdam, C. J.; Manning, A. R.; Robinson, B. H.; Simpson, J. Inorg. Chim. Acta 2005, 358, 1673.
(d) Gallagher, J. F.; Butler, P.; Hudson, R. D. A.; Manning, A. R. J. Chem. Soc., Dalton Trans. 2002, 75.
(e) Butler, P.; Gallagher, J. F.; Manning, A. R. Inorg. Chem. Commun. 1998, 1, 343.
(f) Onitsuka, K.; Tao, X. Q.; Sonogashira, K. Bull. Chem. Soc. Jpn. 1994, 67, 2611.
(g) Matsumoto, M.; Kuroda, K. Tetrahedron Lett. 1980, 21, 4021.
(h) Miyano, S.; Izumi, Y.; Hashimoto, H. J. Chem. Soc., Chem. Commun. 1978, 446.
[27] (a) Lanci, M. P.; Brinkley, D. W.; Stone, K. L.; Smirnov, V. V.; Roth, J. P. Angew. Chem., Int. Ed. 2005, 44, 7273.
(b) Kieber-Emmons, M. T.; Riordan, C. G. Acc. Chem. Res. 2007, 40, 618.
[28] (a) Smith, E. H.; Whittall, J. Organometallics 1994, 13, 5169.
(b) Klein, H. F.; Beck-Hemetsberger, H.; Reitzel, L.; Rodenhaeuser, B.; Cordier, G. Chem. Ber. 1989, 122, 43.
[29] (a) Kamata, K.; Yamaguchi, S.; Kotani, M.; Yamaguchi, K.; Mizuno, N. Angew. Chem., Int. Ed. 2008, 47, 2407.
(b) Siemsen, P.; Livingston, R. C.; Diederich, F. Angew. Chem., Int. Ed. 2000, 39, 2632.
[30] Liu, Q.; Li, G.; He, J.; Liu, J.; Li, P.; Lei, A. Angew. Chem., Int. Ed. 2010, 49, 3371.
[31] (a) Belmont, P. In Silver in Organic Chemistry, Ed.: Harmata, M., John Wiley & Sons Inc., Hoboken, NJ, 2010, Chapter 5, pp. 143~165.
(b) Alvarez-Corral, M.; Munoz-Dorado, M.; Rodriguez-Garcia, I. Chem. Rev. 2008, 108, 3174.
(c) Weibel, J. M.; Blanc, A.; Pale, P. Chem. Rev. 2008, 108, 3149.
[32] (a) Hou, X. L.; Cheung, H. Y.; Hon, T. Y.; Kwan, P. L.; Lo, T. H.; Tong, S. Y.; Wong, H. N. C. Tetrahedron 1998, 54, 1955.
(b) Kirsch, S. F. Org. Biomol. Chem. 2006, 4, 2076.
(c) Lipshutz, B. H. Chem. Rev. 1986, 86, 795.
(d) Keay, B. A. Chem. Soc. Rev. 1999, 28, 209.
(e) Brown, R. C. D. Angew. Chem., Int. Ed. 2005, 44, 850.
(f) Balme, G.; Bouyssi, D.; Monteiro, N. Heterocycles 2007, 73, 87.
[33] Tang, P.; Ritter, T. Tetrahedron 2011, 67, 4449.
[34] Viterisi, A.; Orsini, A.; Weibel, J.-M.; Pale, P. Tetrahedron Lett. 2006, 47, 2779.
[35] (a) Iqbal, J.; Bhatia, B.; Nayyar, N. K. Chem. Rev. 1994, 94, 519.
(b) Snider, B. B. Chem. Rev. 1996, 96, 339.
(c) Alagoz, O.; Yilmaz, M.; Tarik Pekel, A. Synth. Commun. 2006, 36, 1005.
(d) Lee, Y. R.; Byun, M. W.; Kim, B. S. Bull. Korean Chem. Soc. 1998, 19, 1080.
(e) Liu, W.; Jiang, H.; Zhang, M.; Qi, C. J. Org. Chem. 2010, 75, 966.
(f) Yan, R.; Huang, J.; Luo, J.; Wen, P.; Huang, G.; Liang, Y. Synlett 2010, 1071.
(g) Sivan, A.; Deepthi, A.; Nandialath, V. Synthesis 2011, 2466.
[36] Katritzky, A. R. Comprehensive Heterocyclic Chemistry III, Elsevier, Amsterdam, New York, 2008.
[37] (a) Katritzky, A. R.; Xu, Y.-J.; Tu, H. J. Org. Chem. 2003, 68, 4935.
(b) Langer, S. Z.; Arbilla, S.; Benavides, J.; Scatton, B. Adv. Biochem. Psychopharmacol. 1990, 46, 61.
(c) Carminati, G. M. Farmaco, Ed. Prat. 1978, 33, 68.
(d) Allen, J.; Parent, G.; Tizot, A. J. Labelled Compd. Radiopharm. 1986, 23, 807.
[38] (a) Groebke, K.; Weber, L.; Mehlin, F. Synlett 1998, 661.
(b) Yadav, J. S.; Reddy, B. V. S. Y.; Rao, G.; Srinivas, M.; Narsaiah, A. V. Tetrahedron Lett. 2007, 48, 7717.
(c) Bakherad, M.; Nasr-Isfahani, H.; Keivanloo, A.; Doostmohammadi, N. Tetrahedron Lett. 2008, 49, 3819.
(d) Adib, M.; Mohamadi, A.; Sheikhi, E.; Ansari, S.; Bijanzadeh, H. R. Synlett. 2010, 1606.
[39] He, C.; Hao, J.; Xu, H.; Mo, Y.; Liu, H.; Han, J.; Lei, A. Chem. Commun. 2012, 48, 11073.
[40] He, C.; Guo, S.; Ke, J.; Hao, J.; Xu, H.; Chen, H.; Lei, A. J. Am. Chem. Soc. 2012, 134, 5766.
[41] Ueda, S.; Nagasawa, H. J. Am. Chem. Soc. 2009, 131, 15080.
[42] (a) Schoenberg, A.; Bartoletti, I.; Heck, R. F. J. Org. Chem. 1974, 39, 3318.
(b) Schoenberg, A.; Heck, R. F. J. Org. Chem. 1974, 39, 3327.
[43] (a) Zhao, Y.; Jin, L.; Li, P.; Lei, A. J. Am. Chem. Soc. 2008, 130, 9429.
(b) Ritleng, V.; Sirlin, C.; Pfeffer, M. Chem. Rev. 2002, 102, 1731.
(c) Chen, X.; Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Angew. Chem., Int. Ed. 2009, 48, 5094.
(d) Lu, W.; Yamaoka, Y.; Taniguchi, Y.; Kitamura, T.; Takaki, K.; Fujiwara, Y. J. Organomet. Chem. 1999, 580, 290.
(e) Dupont, J.; Consorti, C. S.; Spencer, J. Chem. Rev. 2005, 105, 2527.
(f) Giri, R.; Yu, J.-Q. J. Am. Chem. Soc. 2008, 130, 14082.
(g) Guan, Z.-H.; Ren, Z.-H.; Spinella, S. M.; Yu, S.; Liang, Y.-M.; Zhang, X. J. Am. Chem. Soc. 2009, 131, 729.
(h) Inoue, S.; Shiota, H.; Fukumoto, Y.; Chatani, N. J. Am. Chem. Soc. 2009, 131, 6898.
(i) Yu. L. M.; Xu, Q. H.; Cai, M. Z. Chin. J. Org. Chem. 2008, 28, 256 (in Chinese).
(余腊妹, 许秋华, 蔡明中, 有机化学, 2008, 28, 256.)
[44] (a) Orito, K.; Horibata, A.; Nakamura, T.; Ushito, H.; Nagasaki, H.; Yuguchi, M.; Yamashita, S.; Tokuda, M. J. Am. Chem. Soc. 2004, 126, 14342.
(b) Ohashi, S.; Sakaguchi, S.; Ishii, Y. Chem. Commun. 2005, 486.
(c) Haffemayer, B.; Gulias, M.; Gaunt, M. J. Chem. Sci. 2011, 2, 312.
[45] (a) Bew, S. P. Compr. Org. Funct. Group Transform. II 2005, 5, 19.
(b) Goossen, L. J.; Rodriguez, N.; Goossen, K. Angew. Chem. 2008, 120, 3144; Angew. Chem., Int. Ed. 2008, 47, 3100.
(c) Itahara, T. Chem. Lett. 1982, 1151.
(d) Itahara, T. Chem. Lett. 1983, 127.
[46] (a) Okamoto, N.; Miwa, Y.; Minami, H.; Takeda, K.; Yanada, R. Angew. Chem. 2009, 121, 9873; Angew. Chem. Int. Ed. 2009, 48, 9693.
(b) Cacchi, S.; Fabrizi, G. Chem. Rev. 2005, 105, 2873.
(c) Wolfe, J. P.; Thomas, J. S. Curr. Org. Chem. 2005, 9, 625.
(d) Joucla, L.; Djakovitch, L. Adv. Synth. Catal. 2009, 351, 673.
[47] Zhang, H.; Liu, D.; Chen, C.; Liu, C.; Lei, A. Chem. Eur. J. 2011, 17, 9581.
[48] (a) Bew, S. P. Compr. Org. Funct. Group Transform. II 2005, 5, 19.
(b) Goossen, L. J.; Rodriguez, N.; Goossen, K. Angew. Chem., Int. Ed. 2008, 47, 3100.
[49] (a) Grimster, N. P.; Gauntlett, C.; Godfrey, C. R. A.; Gaunt, M. J. Angew. Chem., Int. Ed. 2005, 44, 3125.
(b) Cacchi, S.; Fabrizi, G. Chem. Rev. 2005, 105, 2873.
(c) Wolfe, J. P.; Thomas, J. S. Curr. Org. Chem. 2005, 9, 625.
(d) Stuart, D. R.; Fagnou, K. Science 2007, 316, 1172.
(e) Seregin, I. V.; Gevorgyan, V. Chem. Soc. Rev. 2007, 36, 1173.
(f) Joucla, L.; Djakovitch, L. Adv. Synth. Catal. 2009, 351, 673.
[50] (a) Stuart, D. R.; Fagnou, K. Science 2007, 316, 1172.
(b) Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215.
(c) Liu, C.; Zhang, H.; Shi, W.; Lei, A. Chem. Rev. 2011, 111, 1780.
(d) Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068.
[51] Liu, Q.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 10788.
[52] (a) Peres, V.; Nagem, T. J.; de Oliveira, F. F. Phytochemistry 2000, 55, 683.
(b) Pinto, M. M. M.; Sousa, M. E.; Nascimento, M. S. J. Curr. Med. Chem. 2005, 12, 2517.
(c) Vieira, L. M. M.; Kijjoa, A. Curr. Med. Chem. 2005, 12, 2413.
(d) Riscoe, M.; Kelly, J. X.; Winter, R. Curr. Med. Chem. 2005, 12, 2539.
(e) El-Seedi, H. R.; El-Ghorab, D. M. H.; El-Barbary, M. A.; Zayed, M. F.; Goeransson, U.; Larsson, S.; Verpoorte, R. Curr. Med. Chem. 2009, 16, 2581.
(f) Saleem, M.; Nazir, M.; Ali, M. S.; Hussain, H.; Lee, Y. S.; Riaz, N.; Jabbar, A. Nat. Prod. Rep. 2010, 27, 238.
[53] Zhao, J.; Larock, R. C. Org. Lett. 2005, 7, 4273.
[54] Zhang, H.; Shi, R.; Gan, P.; Liu, C.; Ding, A.; Wang, Q.; Lei, A. Angew. Chem., Int. Ed. 2012, 51, 5204.
[55] Labadie, J. W.; Stille, J. K. J. Am. Chem. Soc. 1983, 105, 6129.
[56] (a) Izawa, Y.; Shimizu, I.; Yamamoto, A. Bull. Chem. Soc. Jpn. 2004, 77, 2033.
(b) Hu, Y.; Liu, J.; Lu, Z.; Luo, X.; Zhang, H.; Lan, Y.; Lei, A. J. Am. Chem. Soc. 2010, 132, 3153.
[57] Otera, J. Esterification: Methods, Reactions, and Applications, Wiley-VCH, Weinheim, 2003.
[58] (a) Schoenberg, A.; Heck, R. F. J. Org. Chem. 1974, 39, 3327.
(b) Brennfuhrer, A.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2009, 48, 4114.
[59] (a) Larock, R. C. Comprehensive Organic Transformations: A Guide to Functional Group Preparations, 2nd ed., Wiley-VCH, New York, 1999.
(b) Li, Q. L.; Gu, C. Z.; Yin, H.; Zhang, Y. Chin. J. Org. Chem. 2005, 25, 1416 (in Chinese).
(李前荣, 顾承志, 尹浩, 张毅, 有机化学, 2005, 25, 1416.)
[60] (a) Ekoue-Kovi, K.; Wolf, C. Chem. Eur. J. 2008, 14, 6302.
(b) Yoo, W.-J.; Li, C.-J. Tetrahedron Lett. 2007, 48, 1033.
(c) Yoo, W.-J.; Li, C.-J. J. Org. Chem. 2006, 71, 6266.
(d) Xu, B.; Liu, X.; Haubrich, J.; Friend, C. M. Nat. Chem. 2010, 2, 61.
(e) Wu, X.-F.; Darcel, C. Eur. J. Org. Chem. 2009, 1144.
[61] (a) Xu, B.; Liu, X.; Haubrich, J.; Madix, R. J.; Friend, C. M. Angew. Chem., Int. Ed. 2009, 48, 4206.
(b) Oliveira, R. L.; Kiyohara, P. K.; Rossi, L. M. Green Chem. 2009, 11, 1366.
(c) Miyamura, H.; Yasukawa, T.; Kobayashi, S. Green Chem. 2010, 12, 776.
(d) Owston, N. A.; Parker, A. J.; Williams, J. M. J. Chem. Commun. 2008, 624.
(e) Zeng, T.; Song, G.; Li, C.-J. Chem. Commun. 2009, 6249.
(f) Zhang, J.; Gandelman, M.; Shimon, L. J. W.; Milstein, D. Dalton Trans. 2007, 107.
(g) Zhang, J.; Leitus, G.; Ben-David, Y.; Milstein, D. J. Am. Chem. Soc. 2005, 127, 10840.
[62] Liu, C.; Wang, J.; Meng, L.; Deng, Y.; Li, Y.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 5144.
[63] Luo, X.; Zhang, H.; Duan, H.; Liu, Q.; Zhu, L.; Zhang, T.; Lei, A. Org. Lett. 2007, 9, 4571.
[64] (a) Sigman, M. S.; Jensen, D. R. Acc. Chem. Res. 2006, 39, 221.
(b) Sheldon, R. A.; Arends, I. W. C. E.; ten Brink, G.-J.; Dijksman, A. Acc. Chem. Res. 2002, 35, 774.
(c) Schultz, M. J.; Sigman, M. S. Tetrahedron 2006, 62, 8227.
(d) Stahl, S. S. Angew. Chem., Int. Ed. 2004, 43, 3400.
(e) Kroutil, W.; Mang, H.; Edegger, K.; Faber, K. Adv. Synth. Catal. 2004, 346, 125.
(f) Mallat, T.; Baiker, A. Chem. Rev. 2004, 104, 3037.
[65] (a) Swamy, K. C. K.; Kumar, N. N. B.; Balaraman, E.; Kumar, K. V. P. P. Chem. Rev. 2009, 109, 2551.
(b) Otera, J. Esterification: Methods, Reactions, and Applications, Wiley-VCH, Weinheim, 2003.
[66] Liu, C.; Tang, S.; Zheng, L.; Liu, D.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2012, 51, 5662.
[67] Liu, C.; Tang, S.; Lei, A. Chem. Commun. 2013, 49, 1324.
[68] Muzart, J. Tetrahedron 2003, 59, 5789.
[69] Marquard, S. L.; Hartwig, J. F. Angew. Chem., Int. Ed. 2011, 50, 7119.
[70] (a) Widenhoefer, R. A.; Zhong, H. A.; Buchwald, S. L. J. Am. Chem. Soc. 1997, 119, 6787.
(b) Zuideveld, M. A.; Kamer, P. C. J.; van Leeuwen, P. W. N. M.; Klusener, P. A. A.; Stil, H. A.; Roobeek, C. F. J. Am. Chem. Soc. 1998, 120, 7977.
[71] (a) Massari, S.; Daelemans, D.; Barreca, M. L.; Knezevich, A.; Sabatini, S.; Cecchetti, V.; Marcello, A.; Pannecouque, C.; Tabarrini, O. J. Med. Chem. 2010, 53, 641.
(b) Black, L. A.; Cowart, M. D.; Gfesser, G. A.; Wakefield, B. D.; Altenbach, R. J.; Liu, H.; Zhao, C.; Hsieh, G. C. WO 2009085945, 2009 [Chem. Abstr. 2009, 151, 123980].
(c) Aiello, S.; Wells, G.; Stone, E. L.; Kadri, H.; Bazzi, R.; Bell, D. R.; Stevens, M. F. G.; Matthews, C. S.; Bradshaw, T. D.; Westwell, A. D. J. Med. Chem. 2008, 51, 5135.
[72] (a) Moghaddam, F. M.; Boeini, H. Z. Synlett 2005, 1612.
(b) Bose, D. S.; Idrees, M. Tetrahedron Lett. 2007, 48, 669.
(c) Bose, D. S.; Idrees, M. J. Org. Chem. 2006, 71, 8261.
[73] (a) Vera, M. D.; Pelletier, J. C. J. Comb. Chem. 2007, 9, 569.
(b) Evindar, G.; Batey, R. A. J. Org. Chem. 2006, 71, 1802.
(c) Benedi, C.; Bravo, F.; Uriz, P.; Fernandez, E.; Claver, C.; Castillon, S. Tetrahedron Lett. 2003, 44, 6073.
[74] Wang, H.; Wang, L.; Shang, J.; Li, X.; Wang, H.; Gui, J.; Lei, A. Chem. Commun. 2012, 48, 76.
[75] (a) Pozgan, F.; Dixneuf, P. H. Adv. Synth. Catal. 2009, 351, 1737.
(b) Tsai, A. S.; Bergman, R. G.; Ellman, J. A. J. Am. Chem. Soc. 2008, 130, 6316.
(c) Lebrasseur, N.; Larrosa, I. J. Am. Chem. Soc. 2008, 130, 2926.
[76] Yi, H.; Liu, Q.; Liu, J.; Zeng, Z.; Yang, Y.; Lei, A. ChemSusChem 2012, 5, 2143.
[77] (a) Baba, H.; Moriyama, K.; Togo, H. Tetrahedron Lett. 2011, 52, 4303.
(b) Kumar, V.; Sharma, A.; Sharma, M.; Sharma, U. K.; Sinha, A. K. Tetrahedron 2007, 63, 9718.
(c) Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287.
[78] Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215.
[79] (a) Li, Z.; Cao, L.; Li, C.-J. Angew. Chem., Int. Ed. 2007, 46, 6505.
(b) Borduas, N.; Powell, D. A. J. Org. Chem. 2008, 73, 7822.
(c) Li, Y.-Z.; Li, B.-J.; Lu, X.-Y.; Lin, S.; Shi, Z.-J. Angew. Chem., Int. Ed. 2009, 48, 3817.
(d) Correia, C. A.; Li, C.-J. Adv. Synth. Catal. 2010, 352, 1446.
(e) Liu, X.; Zhang, Y.; Wang, L.; Fu, H.; Jiang, Y.; Zhao, Y. J. Org. Chem. 2008, 73, 6207.
(f) Wang, Z.; Zhang, Y.; Fu, H.; Jiang, Y.; Zhao, Y. Org. Lett. 2008, 10, 1863.
(g) Fan, R.; Li, W.; Pu, D.; Zhang, L. Org. Lett. 2009, 11, 1425.
[80] Liu, L.; Floreancig, P. E. Org. Lett. 2010, 12, 4686.
/
| 〈 |
|
〉 |