

基于氨硼烷的直接转移氢化反应研究进展
收稿日期: 2014-12-31
修回日期: 2015-02-14
网络出版日期: 2015-02-28
基金资助
国家自然科学基金(Nos. 21407028, 21471037)以及广东省自然科学基金(No. 2014A030310449)资助项目.
Research Progress on Ammonia-Borane Based Direct Transfer Hydrogenation Reactions
Received date: 2014-12-31
Revised date: 2015-02-14
Online published: 2015-02-28
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21407028, 21471037) and the Natural Science Foundation of Guangdong Province (No. 2014A030310449).
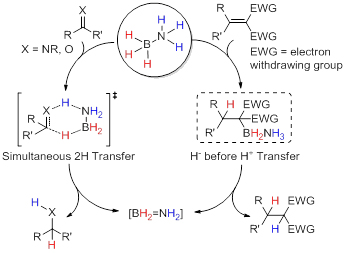
直接转移氢化反应是指氢原子在没有催化剂参与的情况下从氢供体转移到氢受体的过程——即所谓不含金属的反应. 该过程是机理研究中理想的基本步骤, 却通常因反应能垒太高而鲜少报道. 然而, 理论和实验研究都表明, 提高底物的极性可有效降低反应能垒, 意味着极性的氢供体和氢受体直接很可能发生这类反应. 氨硼烷是一种含氢量很高的知名化学储氢材料, 同时, 它也是一种同时含有正、负电性氢原子的理想极性氢供体. 事实上, 以氨硼烷为极性氢供体, 亚胺、极化烯烃以及醛酮等为极性氢受体, 直接转移氢化反应能在温和条件下发生. 主要对这些反应的底物范围以及机理上的异同进行了综述.
阳香华 , 谢珍茗 , 何军 , 余林 . 基于氨硼烷的直接转移氢化反应研究进展[J]. 有机化学, 2015 , 35(3) : 603 -609 . DOI: 10.6023/cjoc201412057
Direct transfer hydrogenation reaction is a H transfer process from a hydrogen donor to a hydrogen acceptor without the presence of a catalyst——the so-called metal-free reaction. It's an idea elementary process for mechanistic study, but was rarely reported due to very high energy barriers. However, it is revealed both theoretically and experimentally that increase in polarity of the substrates can efficiently lower the energy barrier, meaning that polar hydrogen donor and hydrogen acceptors might make such reactions happen. Ammonia borane is well-known as a chemical hydrogen storage material with very high H content. In the meanwhile, it's an ideal polar hydrogen donor containing both hydridic and protic H atoms. Applying ammonia borane as the polar hydrogen donor and some polar hydrogen acceptor molecules such as imines, polarized olefins, aldehydes and ketones, direct transfer hydrogenation reaction occurred under mild conditions. The reaction scope, similarity and differences in mechanism are reviewed herein.

[1] (a) Berke, H. ChemPhysChem 2010, 11, 1837. (b) Ujaque, G.; Lledós, A. J. Mol. Struct.: THEOCHEM 2009, 903, 123.
[2] (a) Meerwein, H.; Schmidt, R. Justus Liebigs Ann. Chem. 1925, 444, 221. (b) Ponndorf, W. Angew. Chem. 1926, 39, 138. (c) Verley, A. Bull. Soc. Chim. Fr. 1925, 37, 871.
[3] (a) Noyori, R.; Okhuma, T.; Kitamura, M.; Takaya, H.; Sayo, N.; Kumobayashi, H.; Akuragawa, S. J. Am. Chem. Soc. 1987, 109, 5856. (b) Noyori, R. In Organic Synthesis, Wiley-Interscience, John Wiley & Sons, New York, 1994, pp. 16~94 and references cited therein. (c) Blum, Y.; Czarkie, D.; Rahamim, Y.; Shvo, Y. Organometallics 1985, 4, 1459. (d) Shvo, Y.; Czarkie, D.; Rahamim, Y.; Chodosh, D. F. J. Am. Chem. Soc. 1986, 108, 7400. (e) Casey, C. P.; Strotman, N. A.; Beetner, S. E.; Johnson, J. B.; Priebe, D. C.; Vos, T. E.; Khodavandi, B.; Guzei, I. A. Organometallics 2006, 25, 1230.
[4] (a) Miller, D. J.; Smith, D. M.; Chan, B.; Radom, M. Mol. Phys. 2006, 104, 777. (b) Fernández, I.; Sierra, M. A.; Cossío, F. P. J. Org. Chem. 2007, 72, 1488.
[5] Feller, D. F.; Schmidt, M. W.; Ruedenberg, K. J. Am. Chem. Soc. 1982, 104, 960.
[6] McKee, M. L.; Stanbury, D. M. J. Am. Chem. Soc. 1992, 114, 3214.
[7] (a) Hamilton, C. W.; Baker, R. T.; Staubitz, A.; Manners, I. Chem. Soc. Rev. 2009, 38, 279. (b) Marder, T. B. Angew. Chem., Int. Ed. 2007, 46, 8116. (c) Gutowska, A.; Li, L.; Shin, Y.; Wang, M.; Li, X. S.; Linehan, J. C.; Smith, R. S.; Kay, B. D.; Schmid, B.; Shaw, W.; Gutowski, M.; Autrey, T. Angew. Chem., Int. Ed. 2005, 44, 3578. (d) Grochala, W.; Edwards, P. P. Chem. Rev. 2004, 104, 1283.
[8] (a) Zhang, L.; Tu, Q.; Chen, X.; Liu, P. Prog. Chem. 2014, 26, 749. (b) Yang, L.; Cao, N.; Du, C.; Dai, H.; Hu, K.; Luo, W.; Cheng, G. Mater. Lett. 2014, 115, 113. (c) Li, H.; Yang, Q,; Chen, X.; Shore, S. G. J. Organomet. Chem. 2014, 751, 60. (d) Kim, Y.; Baek, H.; Lee, J. H.; Yeo, S.; Kim, K.; Hwang, S. J.; Eun, B.; Nam, S. W.; Lim, T. H.; Yoon, C. W. Phys. Chem. Chem. Phys. 2013, 15, 19584. (e) Hartmann, C. E.; Jurcik, V.; Songis, O.; Cazin, C. S. Chem. Commun. 2013, 49, 10629. (f) Marder, T. Angew. Chem., Int. Ed. 2007, 46, 8116. (k) Denney, M. C.; Pons, V.; Hebden, T. J.; Heinekey, D. M.; Goldberg, K. I. J. Am. Chem. Soc. 2006, 128, 12048.
[9] (a) Parvanov, V. M.; Schenter, G. K.; Hess, N. J.; Daemen, L. L.; Hartl, M.; Stowe, A. C.; Camaionia, D. M.; Autrey, T. Dalton Trans. 2008, 33, 4514. (b) Rasul, G. K. G.; Prakash, S.; Olah, G. A. THEOCHEM 2007, 818, 65.
[10] (a) Yurderi, M.; Bulut, A.; Zahmakiran, M.; Gulcan, M.; Ozkar, S. Appl. Catal. B, Environ. 2014, 160, 534. (b) Dong, H.; Berke, H. J. Organomet. Chem. 2011, 696, 1803. (c) Jiang, Y.; Blacque, O.; Fox, T.; Frech, C. M.; Berke, H. Organometallics 2009, 28, 5493. (d) Jiang, Y.; Berke, H. Chem. Commun. 2007, 3571.
[11] Hartmann, C. E.; Jurcik, V.; Songis, O.; Cazin, C. S. Chem. Commun. 2013, 49, 1005.
[12] Yang, X.; Zhao, L.; Fox, T.; Wang, Z.-X.; Berke, H. Angew. Chem., Int. Ed. 2010, 49, 2058.
[13] Morse, P. M. Phys. Rev. 1929, 34, 57.
[14] (a) Hammett, L. P. J. Am. Chem. Soc. 1937, 59, 96. (b) Hammett, L. P. Chem. Rev. 1935, 17, 125.
[15] (a) Yang, X.; Fox, T.; Berke, H. Chem. Commun. 2011, 47, 2053. (b) Yang, X.; Fox, T.; Berke, H. Org. Biomol. Chem. 2012, 10, 852.
[16] (a) Smythe N. C.; Gordon, J. C. Eur. J. Inorg. Chem. 2010, 509. (b) Pons, V.; Baker, R. T.; Szymczak, N. K.; Heldebrant, D. J.; Linehan, J. C.; Matus, M. H.; Grant D. J.; Dixon, D. A. Chem. Commun. 2008, 6597. (c) Shrestha, R. P.; Diyabalanage, H. V. K.; Semelsberger, T. A.; Ott K. C.; Burrell, A. K. Int. J. Hydrogen Energy 2009, 34, 2616.
[17] Yang, X.; Fox, T.; Berke, H. Tetrahedron 2011, 7121.
[18] Brown, H. C.; Schlesinger, H. I.; Burg, A. B. J. Am. Chem. Soc. 1939, 61, 673.
[19] (a) Brown, H. C.; Wang, K, K.; Chandrasekharan, J. J. Am. Chem. Soc. 1983, 105, 2343. (b) Kudo, T.; Higashide, T.; Ikedate, S.; Yamataka, H. J. Org. Chem. 2005, 70, 5157.
[20] Xu, W.; Fan, H.; Wu, G.; Chen, P. New J. Chem. 2012, 36, 1496.
[21] Xu, W.; Wu, G.; Yao, W.; Fan, H.; Wu, J.; Chen, P. Chem. Eur. J. 2012, 18, 13885.
[22] Wang, X.; Yao, W.; Zhou, D.; Fan, H. Mol. Phys. 2013, 111, 3014.
/
| 〈 |
|
〉 |