

1'-芳甲基二茂铁联烯的合成
收稿日期: 2015-02-08
修回日期: 2015-03-08
网络出版日期: 2015-03-13
基金资助
国家自然科学基金(No. 21262023)和内蒙古自治区杰出青年培育基金(No. 2014JQ02)资助项目
Synthesis of 1'-Arylmethyl-1-(1,2-dienyl)ferrocenes
Received date: 2015-02-08
Revised date: 2015-03-08
Online published: 2015-03-13
Supported by
Project supported by the National Natural Science Foundation of China (No. 21262023) and the Natural Science Foundation of Inner Mongolia of China (No. 2014JQ02).
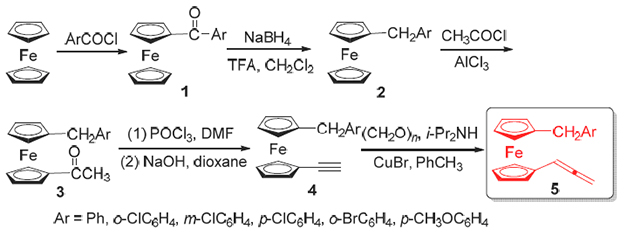
设计了新型1'-芳甲基二茂铁联烯衍生物的合成方法, 该方法以方便易得的二茂铁为底物, 通过傅氏酰基化、还原、乙酰化、乙炔化及改进的Crabbé反应等过程, 合成了六种目标化合物. 并通过核磁共振氢谱、碳谱及高分辨质谱对其结构进行了表征.
关键词: 二茂铁; 联烯; 1'-芳甲基二茂铁联烯; 合成
王晨君 , 王晗 , 马超 , 张开新 , 韩飞 , 李保国 , 陈树峰 . 1'-芳甲基二茂铁联烯的合成[J]. 有机化学, 2015 , 35(7) : 1544 -1550 . DOI: 10.6023/cjoc201502013
A convenient procedure for synthesizing 1'-arylmethyl-1-(1,2-dienyl)ferrocene derivatives was explored. Using commercially available ferrocene as the starting material, the six target compounds were obtained in five steps via Friedel-Crafts acylation, reduction, acetylation, acetylene formation and modified Crabbé reaction. The structures of the obtained allenes were characterized by 1H NMR and 13C NMR and HRMS.

Key words: ferrocene; allene; 1'-arylmethyl-1-(1,2-dienyl)ferrocene; synthesis
[1] Kealy, T. J.; Pauson, P. L. Nature 1951, 168, 1039.
[2] Št?pni?ka, P. Ferrocenes: Ligands, Materials and Biomolecules, Wiley-VCH, Chichester, U. K., 2008.
[3] Togni, A.; Hayashi, T. Ferrocenes, VCH, New York, 1995.
[4] Hildebrandt, A.; Lang, H. Organometallics 2013, 32, 5640.
[5] Fabre, B. Acc. Chem. Res. 2010, 43, 1509.
[6] Musgrave, R. A.; Russell, A. D.; Manners, I. Organometallics 2013, 32, 5654.
[7] Dai, L.; Tu, T.; You, S.; Deng, W.; Hou, X. Acc. Chem. Res. 2003, 36, 659.
[8] Atkinson, R. C. J.; Gibson, V. C.; Long, N. J. Chem. Soc. Rev. 2004, 33, 313.
[9] Schaarschmidt, D.; Lang, H. Organometallics 2013, 32, 5668.
[10] Moriuchi, T.; Hirao, T. Acc. Chem. Res. 2010, 43, 1040.
[11] Braga, S. S.; Silva, A. M. S. Organometallics 2013, 32, 5626.
[12] Hillard, E.; Vessières, A.; Jaouen, G. Top. Organomet. Chem. 2010, 32, 81.
[13] Ornelas, C. New J. Chem. 2011, 35, 1973.
[14] Brasholz, M.; Reissig, H.-U.; Zimmer, R. Acc. Chem. Res. 2009, 42, 45.
[15] Ma, S. Acc. Chem. Res. 2009, 42, 1679.
[16] Krause, N.; Winter, C. Chem. Rev. 2011, 111, 1994.
[17] Aubert, C.; Fensterbank, L.; Garcia, P.; Malacria, M.; Simonneau, A. Chem. Rev. 2011, 111, 1954.
[18] Yu, S.; Ma, S. Angew. Chem., Int. Ed. 2012, 51, 3074.
[19] Deng, Y.; Gu, Z.; Ma, S. Chin. J. Org. Chem. 2006, 26, 1468 (in Chinese). (邓友前, 顾振华, 麻生明, 有机化学, 2006, 26, 1468.)
[20] Brandsma, L. Synthesis of Acetylenes, Allenes and Cumulenes, Elsevier, Oxford, 2004.
[21] Krause, N.; Hoffmann-Roder, A. Tetrahedron 2004, 60, 11671.
[22] Brummond, K. M.; Deforrest, J. E. Synthesis 2007, 795.
[23] Ogasawara, M. Tetrahedron: Asymmetry 2009, 20, 259.
[24] Yu, S.; Ma, S. Chem. Commun. 2011, 47, 5384.
[25] Rona, P.; Crabbé, P. J. Am. Chem. Soc. 1969, 91, 3289.
[26] Kuang, J.; Ma, S. J. Org. Chem. 2009, 74, 1763.
[27] Chen, B.; Wang, N.; Fan, W.; Ma, S. Org. Biomol. Chem. 2012, 10, 8465.
[28] Kuang, J.; Ma, S. J. Am. Chem. Soc. 2010, 132, 1786.
[29] Ye, J.; Li, S.; Chen, B.; Fan, W.; Kuang, J.; Liu, J.; Liu, Y.; Miao, B.; Wan, B.; Wang, Y.; Xie, X.; Yu, Q.; Yuan, W.; Ma, S. Org. Lett. 2012, 14, 1346.
[30] Kuang, J.; Luo, H.; Ma, S. Adv. Synth. Catal. 2012, 354, 933.
[31] Tang, X.; Zhu, C.; Cao, T.; Kuang, J.; Lin, W.; Ni, S.; Zhang, J.; Ma, S. Nat. Commun. 2013, 4, 2450.
[32] Chen, S.; Shi, J.; Wang, B.; Zhao, H.; Li, B. Appl. Organomet. Chem. 2012, 26, 284.
[33] Chen, S.; Wang, B.; Yan, Q.; Shi, J.; Zhao, H.; Li, B. RSC Adv. 2013, 3, 1758.
/
| 〈 |
|
〉 |