

侧链含不饱和基团取代的硫代双烯型镍络合物的合成及其性能
收稿日期: 2015-03-04
修回日期: 2015-05-05
网络出版日期: 2015-05-14
Synthesis and Properties of Extended Bis-1,2-dithiolene Nickel Complexes Substituted with Unsaturated Group
Received date: 2015-03-04
Revised date: 2015-05-05
Online published: 2015-05-14
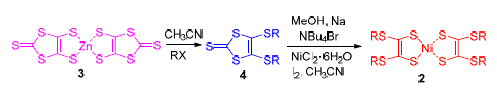
合成了3个侧链含不饱和基团取代的硫代双烯型镍络合物, 即双[4,5-二(4'-叔丁基苄基)硫-1,3-二硫杂环戊烯]镍(2a)、双[4,5-二(烯丙基)硫-1,3-二硫杂环戊烯]镍(2b)和双[4,5-二(3'-苯基-烯丙基)硫-1,3-二硫杂环戊烯]镍(2c)以及作为参照品的一个侧链含饱和基团的硫代双烯型镍络合物, 即双[4,5-二(癸基)硫-1,3-二硫杂环戊烯]镍(2d). 它们的结构经1H NMR和质谱得到了表征. 这些化合物在甲苯中有较好的溶解性, 最大吸收波长(λmax)在1000 nm左右, 而且取代基对它们最大吸收波长的位移影响很小. 它们在固态聚甲基丙烯酸甲酯(PMMA)中的λmax也在1000 nm左右, 且浓度较低时, 吸收曲线未见有宽化的迹象. 固态循环伏安的测试结果表明, 苯基的引入对提高这类化合物的氧化电位有影响. 热重分析(TGA)的结果表明, 这类化合物失重5%的温度都在200 ℃以上. 氙灯(300 W)照射100 min, 它们的吸光度在甲苯中下降不到10%, 在固态的PMMA中下降不到2%, 苯基的引入有助于提高这类化合物在溶液和固态PMMA中的光化学稳定性.
孙健 , 金哲明 , 王成云 , 沈永嘉 . 侧链含不饱和基团取代的硫代双烯型镍络合物的合成及其性能[J]. 有机化学, 2015 , 35(9) : 1948 -1954 . DOI: 10.6023/cjoc201503005
Three bis-1,2-dithiolene nickel complexes with unsaturated substituted groups in the terminals, bis[4,5-di(4'-t-butylbenzyl)thio-1,3-dithion]nickel (2a), bis[4,5-dially-thio-1,3-dithion]nickel (2b) and bis[4,5-di(3'-phenyl-ally)thio-1,3-di-thion]nickel (2c) together with a bis-1,2-dithiolene nickel complexes with saturated substituted groups in the terminals, bis[4,5-didecylthio-1,3-dithion]nickel (2d), as a reference, were synthesized and characterized by 1H NMR and MS. They were easily soluble in toluene with the maximum absorption wavelengths (λmax) around 1000 nm, the effects of substituent groups in the terminals on λmax were not so significant. Their λmax in solid polymethyl methacrylate (PMMA) were around 1000 nm as well, and no broadening absorption bands could be observed in lower concentration. The solid cyclic voltammetry (CV) curves showed that the oxidation potentials would be increased by introduction of phenyl group in the terminals, thus their electrochemical stability could be improved. The results of thermal gravimetric analysis (TGA) indicated that the temperature of 5% weight-loss was higher than 200 ℃ for these compounds. Irradiated by xenon lamp (300 W) for 100 min, their absorbance was decreased less than 100% in toluene and 2% in solid polymethyl methacrylate (PMMA), respectively. The introduction of phenyl group in the terminals could improve the photo-stability for these molecules both in solution and solid PMMA.

[1] Men, J. F.; Cheng, H. F.; Chen, Z. H.; Chu, Z. Y. Mater. Rev. 2008, 22, 3 (in Chinese). (门金凤, 程海峰, 陈朝辉, 楚增勇, 材料导报, 2008, 22, 3.)
[2] Marshall, K. L.; Painte, G.; Lotito, K. A. Mol. Cryst. Liquid Cryst. 2006, 47, 449.
[3] Aragoni, C.; Arca, M.; Demartin, F.; Devillanova, F. A.; Garau, A.; Isaia, F.; Lelj, F.; Lippolis, V.; Verani, G. J. Am. Chem. Soc. 1991, 121, 7098.
[4] Mueller-Westerhoff, U. T.; Vance, B.; Yoon, D. I. Tetrahedron 1991, 47, 909.
[5] Espine, P. Coord. Chem. Rev. 1992, 117, 215.
[6] Fabian, J.; Nakazumi, H.; Matsuoka, M. Chem. Rev. 1992, 92(6), 1200.
[7] Coe, B. J. Compr. Coord. Chem. 2004, 9, 621.
[8] Cho, J. Y.; Domercq, B.; Jones, S. C.; Yu, J.; Zhang, X.; An, Z.; Bishop, M.; Barlow, S.; Marder, S. R.; Kippelen, B. J. Mater. Chem. 2007, 17, 2642.
[9] Stiefel, E. I. Karlin, K. D. Dithiolene Chemistry, Progress in Inorganic Chemistry, John Wiley Sons, 2004, 52, 399.
[10] Deplano, P.; Pilia, L.; Espa, D.; Mercuri, M. L.; Serpe, A. Coord. Chem. Rev. 2010, 254, 1434.
[11] Robertson, N.; Cronin, L. Coord. Chem. Rev. 2002, 227, 93.
[12] Chen, H.; Cheng, Z. S. J. East China Institute Chem. Technol. 1992, 18(5), 628 (in Chinese). (陈红, 程铸生, 华东化工学院学报, 1992, 18(5), 628.)
[13] Men, J. F.; Cheng, H. F.; Chen, Z. H.; Chu, Z. Y.; Zheng, W. W.; Wang, Q. Infrared Technol. 2008, 30, 150 (in Chinese).(门金凤, 程海峰, 陈朝辉, 楚增勇, 郑文伟, 王茜, 红外技术, 2008, 30, 150.)
[14] Dai, F.; Fan, W. H.; Wu, J. H.; Wang, L. X.; Zhang, Q. T. Laser Infrared 2011, 41, 1131 (in Chinese). (戴峰, 樊卫华, 吴金华, 王丽熙, 张其土, 激光与红外, 2011, 41(10), 1131.)
[15] Gao, J. X.; Miao, Q. Q.; Wang, Z. Q.; Gao, Y. R.; Ma, Y. L. Fine Chem. 2011, 28, 909 (in Chinese). (高俊雄, 苗青青, 王泽清, 高玉荣, 马廷丽, 精细化工, 2011, 28, 909.)
[16] Charlton, A.; Hill, C. A. S.; Underhill, A. E.; Malik, K. M. A.; Hursthouse, M. B.; Karaulovb, A. I.; Meller, J. J. Mater. Chem. 1994, 4(12), 1861.
[17] Robertson, N.; Cronin, L. Coord. Chem. Rev. 2002, 227, 93.
[18] Svenstrup, N.; Becher, J. Synthesis 1995, 215.
[19] Bui, T. T.; Vuong, M. H. New J. Chem. 2012, 36, 2033.
[20] Huang, Q.; Wang, L. X.; Zhang, Q. S. Laser Infrared 2008, 38(5), 421 (in Chinese). (黄强, 王丽熙, 张其土, 激光与红外, 2008, 38(5), 421.)
/
| 〈 |
|
〉 |