

巴西木素类天然产物的合成研究进展
收稿日期: 2015-03-17
修回日期: 2015-05-03
网络出版日期: 2015-05-14
基金资助
长江学者和创新团队发展计划(No. IRT13095)、国家自然科学基金(Nos. 21462049, 21332007, U1402227)、云南省自然科学基金(Nos. 2013FA028, 2012FB113)、云南大学青年英才培育计划资助项目.
Research Progress on the Synthesis of Brazilin-Type Natural Products
Received date: 2015-03-17
Revised date: 2015-05-03
Online published: 2015-05-14
Supported by
Project supported by the Program for Changjiang Scholars and Innovative Research Team in University (No. IRT13095), the National Natural Science Foundation of China (Nos. 21462049, 21332007, U1402227), the Natural Science Foundation of Yunnan Province (Nos. 2013FA028, 2012FB113) and the Excellent Young Talents of Yunnan University.
巴西木素类型天然产物是从中药苏木(Caesalpinia sappan)中分离得到的一类化合物, 因其独特的结构和良好的生物活性而成为合成化学家关注的焦点. 目前, 已有许多关于这类天然产物合成的报道. 我们主要对巴西木素类型天然产物的合成研究进展进行了综述.
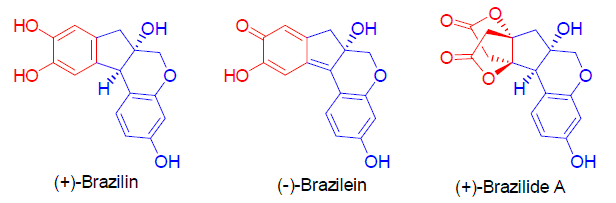
关键词: 巴西木素; Brazilide A; Brazilein; 合成
汪学全 , 刘卫 , 段素月 , 羊晓东 , 张洪彬 . 巴西木素类天然产物的合成研究进展[J]. 有机化学, 2015 , 35(8) : 1585 -1597 . DOI: 10.6023/cjoc201503025
Brazilin-type natural products were isolated from the heartwood of Caesalpinia sappan, a famous traditional Chinese medicine. These compounds have stimulated a great deal of interest on synthetic chemists due to their structural novelty and promising biological activities. Up to now, many methods towards the synthesis of these natural products have been developed. Progress on the synthesis of Brazilin-type natural products are reviewed in this paper.

Key words: brazilin; brazilide A; brazilein; synthesis
[1] Robinson, R. Bull. Soc. Chim. Fr. 1958, 125.
[2] Masahiro, N.; Seiji, N. Chem. Pharm. Bull. 1990, 38, 1490.
[3] Michio, N.; Hiroyuki, N.; Mariko, N. Chem. Pharm. Bull. 1987, 35, 3568.
[4] Yang, B. O.; Ke, C. Q.; He, Z. S.; Yang, Y. P.; Ye, Y. Tetrahedron Lett. 2002, 43, 1731.
[5] Kim, D. S.; Baek, N. I.; Oh, S. R.; Jung, K. Y.; Lee, I. S.; Lee, H. K. Phytochemistry 1997, 46, 177.
[6] (a) Puchtler, H.; Sweat, F. Histochemistry 1964, 4, 197. (b) Puchtler, H.; Meloan, S. N.; Waldrop, F. S. Histochemistry 1986, 85, 353.
[7] Kim, B. H.; Kim, S. J.; Jeong, E. J.; Sohn, J. H.; Jung, M.; Lee, H.; Kim, S. H. J. Agric. Food Chem. 2012, 60, 9882.
[8] (a)Kim, S. G.; Kim, Y. M.; Khil, L. Y.; Jeon, S. D.; So, D. S.; Moon, C. H.; Moon, C. K. Arch. Pharm. Res. 1998, 21, 140. (b) Hwang, G. S.; Kim, J. Y.; Chang, T. S.; Jeon, S. D.; So, D. S.; Moon, C. K. Arch. Pharm. Res. 1998, 21, 774.
[9] Hikino, H.; Taguchi, T.; Fujimura, H.; Hiramatsu, Y. Planta Med. 1977, 31, 214.
[10] Xu, H. X.; Lee, S. F. Phytother. Res. 2004, 18, 647.
[11] Moon, C. K.; Park, K. S.; Kim, S. G.; Won, H. S.; Chung, J. H. Drug Chem. Taxicol. 1992, 15, 81.
[12] Lee, Y. R.; Noh, E. M.; Han, J. H.; Kim, J. M.; Hwang, J. K.; Hwang, B. M.; Chung, E. Y.; Kim, B. S.; Lee, S. H.; Lee, S. J.; Kim, J. S. Eur. J. Pharmacol. 2012, 674, 80.
[13] Wang, X.; Zhao, H. X.; Bai, H. Acta Chin. Med. Pharmacol. 2013, 41, 164 (in Chinese). (王鑫, 赵焕新, 白虹, 中医药学报, 2013, 41, 164.)
[14] (a) Zhao, Y. N.; Pan, Y.; Tao, J. L.; Xing, D. M.; Du, L. J. Pharmacology 2006, 76, 76. (b) Zhong, X.; Wu, B.; Pan, Y. J.; Zheng, S. Neoplasma 2009, 56, 387. (c) Shen, J.; Zhang, H.; Lin, H.; Su, H.; Xing, D.; Du, L. Eur. J. Pharmacol. 2007, 558, 88. (d) Ye, M.; Xie, W. D.; Lei, F.; Meng, Z.; Zhao, Y. N.; Su, H.; Du, L. J. Int. Immunopharmacol. 2006, 6, 426. (e) Shen, J.; Yip, S.; Wang, Z. X.; Wang, W.; Xing, D. M.; Du, L. J. Eur. J. Pharmacol. 2008, 580, 366.
[15] Hill, R. A.; Parker, M. C. Nat. Prod. Rep. 2002, 19, ⅲ.
[16] Heller, W.; Tamm, C. Progress in the Chemistry of Organic Natural Products, Vol. 40, Eds.: Herz, H.; Criesenbach, H.; Kirby, W, Springer-Verlag, Inc., New Work, 1980, p.105.
[17] Saitoh, T.; Sakashita, S.; Nakata, H.; Shimokawa, T.; Kinjo, J.; Yamahara, J.; Yamasakim, M.; Nohara, T. Chem. Pharm. Bull. 1986, 34, 2506.
[18] (a) Kirkiacharian, B. S. C. R. Acad. Ser. C 1972, 274, 2096. (b) Kirkiacharian, B. S.; Billet, D.; Durgeat, M.; Heitz, S.; Adjangba, M. K. Bull. Soc. Chim. Fr. 1975, 1770.
[19] Yen, C. T.; Kyoko, N. G.; Hwang, T. L.; Wu, P. C.; Morris-Natschke, S. L.; Lai, W. C.; Bastowd, K. F.; Chang, F. R.; Wu, Y. C.; Lee, K. H. Bioorg. Med. Chem. Lett. 2010, 20, 1037.
[20] (a) Davis, F. A.; Chen, B. C. J. Org. Chem. 1993, 58, 1751. (b) Davis, F. A.; Chen, B. C. Tetrahedron Lett. 1990, 31, 6823.
[21] Arnoldi, A.; Bassoli, A.; Borgonovo, G.; Merlini, L. J. Chem. Soc., Perkin Trans. 1 1995, 2447.
[22] Ishii, H.; Koyama, H.; Hagiwara, K.; Miura, T.; Xue, G. G.; Hashimoto, Y.; Kitahara, G.; Aida, Y.; Suzuki, M. Bioorg. Med. Chem. Lett. 2012, 22, 1469.
[23] Javed, U.; Karim, M.; Jahng, K. C.; Park, J. G.; Jahng, Y. D. Tetrahedron: Asymmetry 2014, 25, 1270.
[24] Huang, Y. D.; Zhang, J. S.; Pettus, T. R. R. Org. Lett. 2005, 7, 5841.
[25] Lin, C. C.; Teng, T. M.; Tsai, C. C.; Liao, H. Y.; Liu, R. S. J. Am. Chem. Soc. 2008, 130, 16417.
[26] Pan, C. X.; Zeng, X. H.; Guan, Y. F.; Jiang, X. L.; Li, L.; Zhang, H. B. Synlett 2011, 425.
[27] Wang, X. Q.; Zhang, H. B.; Yang, X. D.; Zhao, J. F.; Pan, C. X. Chem. Commun. 2013, 49, 5405.
[28] Li, L. Q.; Li, M. M.; Wang, K.; Qin, H. B. Tetrahedron Lett. 2013, 54, 6029.
[29] Yadav, J. S.; Mishra, A. K.; Das, S. Tetrahedron 2014, 70, 7560.
[30] Jung, Y.; Kim, I. J. Org. Chem. 2015, 80, 2001.
/
| 〈 |
|
〉 |