

α-山道年光催化重排衍生物的制备及细胞毒活性研究
收稿日期: 2015-05-11
修回日期: 2015-06-08
网络出版日期: 2015-06-12
基金资助
国家自然科学基金(Nos. 21372056, 21202027)资助项目.
Photocatalysis Synthesis and Cytotoxicity of α-Santonin Rearrangement Derivatives
Received date: 2015-05-11
Revised date: 2015-06-08
Online published: 2015-06-12
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21372056, 21202027).
戴笑丽 , 汤建 , 章凌琼 , 陶连芝 , 欧阳臻 , 王民 . α-山道年光催化重排衍生物的制备及细胞毒活性研究[J]. 有机化学, 2015 , 35(10) : 2142 -2149 . DOI: 10.6023/cjoc201505020
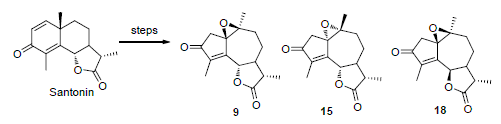
20 guaianolide derivatives 1~20 and 6-epi-santonin were synthesized from α-santonin through rearrangement, oxidation, and reduction, hydrolysis. The structures of all compounds were characterized by 1H NMR, 13C NMR and MS. The cell proliferation inhibiting activities of the target compounds were evaluated against A549 and HepG2 cell lines by thiazolyl blueterazolium bromide (MTT) method. The results showed that the epoxy guaiane 9 had obvious inhibitory effect on A549 and HepG2 cell lines with IC50 values of (14.5±0.1), (10.6±0.3) μmol/L while epoxy guaiane 15 showed IC50 values of (6.4±1.3) and (8.1±0.7) μmol/L.
Key words: α-santonin; guaiane; derivative; cytotoxicity
[1] Wu, L. J. Textbook of Medicinal Chemistry of Natural Products, People's Medical Publishing House, Beijing, 2007, pp. 227~232 (in Chinese). (吴立军, 天然药物化学教材, 人民卫生出版社, 北京, 2007, pp. 227~232.)
[2] Saxena, V. K.; Mondal, S. K. Phytochemistry 1994, 35, 1080.
[3] Qian, M. K.; Zhu, D. Y. Pharm. Bull. 1979, 17, 277 (in Chinese ). (钱名堃, 朱大元, 药学通报, 1979, 17, 277.)
[4] Bruno, M.; Rosselli, S.; Maggio, A.; Raccuglia, R. A.; Bastow, K. F.; Lee, K. H. J. Nat. Prod. 2005, 68, 1042.
[5] René, C.; Anke, H.; Bianka, S.; Stefan, S.; Alexander, B.; Ralph, K.; Dieter, S. Pharm. Chem. Life Sci. 2012, 345, 215.
[6] Francisco, A. M.; Alejandro, S.; Azusa, Y.; Rosa, M., V.; Frank, R. F.; José, M. G. M. J. Nat. Prod. 2012, 75, 1967.
[7] Park, K. H.; Yang, M. S.; Park, M. K.; Kim, S. C.; Yang, C. H.; Park, S. J.; Lee, J. R. Fitoterapia 2009, 80, 54.
[8] Usuki, T.; Sato, M.; Hara, S.; Yoshimoto, Y.; Kondo, R.; Zimmermann, S.; Kaiser, M.; Brun, R.; Hamburger, M.; Adams, M. Bioorg. Med. Chem. Lett. 2014, 24, 794.
[9] Zhang, W. H.; Luo, S. J.; Fang, F.; Chen, C. S.; Hu, H. W.; Jia, X. S.; Zhai, H. B. J. Am. Chem. Soc. 2005, 127, 18.
[10] Li, C.; Dong, T.; Li, Q.; Lei, X. Angew. Chem., Int. Ed. 2014, 53, 12111.
[11] Li, C.; Yu, X. L.; Lei, X. G. Org. Lett. 2010, 12, 4284.
[12] Xia, W.-J.; Sun, L.-D.; Shi, L.; Zhang, S.-Y.; Tu, Y.-Q. Chin. J. Chem. 2004, 22, 377.
[13] Li, D. R.; Xia, W. J.; Shi, L.; Tu, Y. Q. Synthesis 2003, 41.
[14] Xia, W. J.; Tu, Y. Q.; Li, D. R. Synth. Commun. 2001, 31, 1613.
[15] Xia, D.; Du Y.; Yi, Z.; Song, H.; Qin, Y. Chem.-Eur. J. 2013, 19, 4423.
[16] Bargues, V.; Blay, G.; Cardona, L.; Garcia, B.; Pedro, J. R. Tetrahedron 1998, 54, 1845.
[17] Arantes, F. F. P.; Barbosa, L. C. A.; Alvarenga, E. S.; Demuner, A. J.; Bezerra, D. P.; Ferreira, J. R. O.; Costa-Lotufo, L. V.; Pessoa, C.; Moraes, M. O. Eur. J. Med. Chem. 2009, 44, 3739.
[18] Andrew, E. G.; Mark, T. E. J. Org. Chem. 1989, 54, 1468.
[19] Peter, M.; Simon, B.; Roland, F. J. Am. Chem. Soc. 1993, 115, 12595.
[20] Blay, G.; Cardona, L.; Garcia, B.; Lahoz, L.; Pedro, J. R. J. Org. Chem. 2001, 66, 7700.
[21] Makiyi, E. F.; Frade, R. F. M.; Lebl, T.; Jaffray, E. G.; Cobb, S. E.; Harvey, A. L.; Slawin, A. M. Z.; Hay, R. T.; Westwood, N. J. Eur. J. Org. Chem. 2009, 5711.
[22] Ando, M.; Akahane, A.; Yamaoka, H.; Takase, K. J. Org. Chem. 1982, 47, 3909.
/
| 〈 |
|
〉 |