

苯并[b][1,4]二氮杂衍生物的合成及其应用新进展
收稿日期: 2015-10-08
修回日期: 2015-11-19
网络出版日期: 2015-12-15
基金资助
国家自然科学基金(No.21276064)资助项目.
Progress in Synthesis and Application of Benzo[b][1,4]diazepine Derivatives
Received date: 2015-10-08
Revised date: 2015-11-19
Online published: 2015-12-15
Supported by
Project supported by the National Natural Science Foundation of China (No. 21276064).
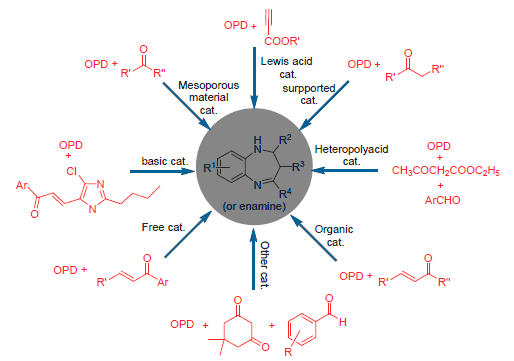
苯并[b][1,4]二氮杂衍生物是一类含有两个氮杂原子的苯并七元杂环化合物,以其特殊的结构特点和广泛的生理及药理活性而备受关注,因此其合成和活性研究也是有机化学及药学领域的研究热点.就近10年来苯并[b][1,4]二氮杂衍生物在抑菌、抗神经炎和抗癌活性等方面的应用进展进行了归纳总结;综述了其不同催化体系下合成及机理的最新研究进展,同时对本课题组的研究成果做了阐述,并对其合成及应用前景进行了展望.
关键词: 苯并[b][1,4]二氮杂; 合成; 应用; 催化剂; 研究进展
尹刘燕 , 王兰芝 , 李晓庆 , 安迎双 . 苯并[b][1,4]二氮杂衍生物的合成及其应用新进展[J]. 有机化学, 2016 , 36(4) : 711 -723 . DOI: 10.6023/cjoc201510002
Benzo[b][1,4]diazepine derivatives is a class of benzo and seven-number heterocyclic compounds containing double nitrogen atoms. It was concerned for its special structural features, strong physiological and pharmacological activity. Therefore, the research on synthetic method and biological activity of benzo[b][1,4]diazepine has become one of the hot issues in the chemical and pharmaceutical areas. Their application in the antimicrobial, anti-neuroinflammatory, anticancer activity and so on in the recently ten years is reviewed. Besides, the progress in synthesis of the benzo[b][1,4]diazepine compounds at different types of catalysts and its synthetic reaction mechanisms are summarized. Meanwhile, the recent works of authors' research group are discussed. Their synthesis and application prospects are also expected.

Key words: benzo[b][1,4]diazepine; synthesis; application; catalyst; progress
[1] (a) Pan, X. Q.; Zou, J. P.; Huang, Z. H.; Zhang, W. Tetrahedron Lett. 2008, 49, 5302.
(b) Li, X. Q.; Wang S. S.; Cheng, S. Y.; Wang, L. Z. J. Hebei Normal Univ. 2013, 37, 317 (in Chinese). (李晓庆, 王莎莎, 程素艳, 王兰芝, 河北师范大学学报, 2013, 37, 317.)
[2] Jiang, Y. J.; Cai, J. J.; Zou, J. P.; Zhang, W. Tetrahedron Lett. 2010, 51, 471.
[3] Wang, L.; Sullivan, G. M.; Hexamer, L. A.; Hasvold, L. A.; Thalji, R.; Przytulinska, M.; Tao, Z. F.; Li, G. Q.; Chen, Z. H.; Xiao, Z.; Gu, W. Z.; Xue, J.; Bui, M. H.; Merta, P.; Kovar, P.; Bouska, J. J.; Zhang, H. Y.; Park, C.; Stewart, K. D.; Sham, H. L.; Sowin, T. J.; Rosenberg, S. H.; Lin, N. H. J. Med. Chem. 2007, 50, 4162.
[4] Corral, C.; Lissavetzky, J.; Valdolmillos, A. M. J. Heterocycl. Chem. 1985, 22, 1349.
[5] Bunin, B. A.; Ellman, J. A. J. Am. Chem. Soc. 1992, 114, 10997.
[6] Reddy, K. V. V.; Rao, P. S.; Ashok, D. Synth. Commun. 2000, 30, 1825.
[7] Bandgar, B. P.; Patil, A. V.; Chavan, O. S. J. Mol. Catal. A: Chem. 2006, 256, 99.
[8] Zhao, H. Y. Ph. D. Dissertation, Doctoral Dissertation of Peking Union Medical College & Chinese Academy of Medical Sciences, Beijing, 2007 (in Chinese). (赵海燕, 博士论文, 中国医学科学院&中国协和医科大学药物研究所, 北京, 2007.)
[9] Wüller, W. E.; Groh, B.; Bub, O.; Hofmann, H. P.; Kreiskott, H. Pharmacopsychiatry 1986, 19, 314.
[10] Kruse, H. Drug Dev. Res. 1982, 2, 145.
[11] Nicholson, A. N.; Stone, B. M.; Clarke, C. H. Br. J. Clin. Pharmacol. 1977, 4, 567.
[12] Al-Hiari, Y. M.; Abu-Dahab, R.; El-Abadelah, M. M. Molecules 2008, 13, 2880.
[13] Konda, S. G.; Shaikh, B. M.; Chavan, S. A.; Dawane, B. S. Chin. Chem. Lett. 2011, 22, 65.
[14] Joshi, Y. C.; Saingar, S.; Kavita; Joshi, P.; Kumar, R. J. Korean Chem. Soc. 2011, 55, 638.
[15] Palanisamy, P.; Jenniefer, S. J.; Muthiah, P. T.; Kumaresan, S. RSC Adv. 2013, 3, 19300.
[16] Thakrar, S.; Bavishi, A.; Radadiya, A.; Parekh, S.; Bhavsar, D.; Vala, H.; Pandya, N.; Shah, A. J. Heterocycl. Chem. 2013, 50, E73.
[17] Wang, L. Z.; Hua, Z. X.; Wang, S. S. Chin. J. Org. Chem. 2013, 33, 2376 (in Chinese). (王兰芝, 花中霞, 王莎莎, 有机化学, 2013, 33, 2376.)
[18] Wang, L. Z.; Li, X. Q.; An, Y. S. Org. Biomol. Chem. 2015, 13, 5497.
[19] Ha, S. K.; Shobha, D.; Moon, E.; Chari, M. A.; Mukkanti, K.; Ki, S. H.; Ahn, K. H.; Kim, S. Y. Bioorg. Med. Chem. Lett. 2010, 20, 3969.
[20] Chen, Y. B.; Le, V.; Xu, X. Y.; Shao, X. S.; Liu, J. W.; Li, Z. Bioorg. Med. Chem. Lett. 2014, 24, 3948.
[21] Ekonomopoulou, M. T.; Tsoleridis, C. A.; Argyraki, M.; Polatoglou, E. Genet. Test. Mol. Biomarkers 2010, 14, 377.
[22] Eleftheriadis, N.; Neochoritis, C. G.; Tsoleridis, C. A.; Julia, S. S.; Zafiroula, I. K. Eur. J. Med. Chem. 2013, 67, 302.
[23] Herbert, J. A. L.; Suschitzky, H. J. Chem. Soc., Perkin. Trans. 1 1974, 2657.
[24] Jung, D. I.; Choi, T. W.; Kim, Y. Y.; Kim, I. S.; Park, Y. M.; Lee, Y. G.; Jung, D. H. Synth. Commun. 1999, 29, 1941.
[25] Balakrishna, M. S.; Kaboudin. B. Tetrahedron Lett. 2001, 42, 1127.
[26] Curini, M.; Epifano, F.; Marcotullio, M. C.; Rosati, O. Tetrahedron Lett. 2001, 42, 3193.
[27] Minothora, P.; Julia, S. S.; Constantinos, A. T. Tetrahedron Lett. 2002, 43, 1755.
[28] Yang, H. Q.; Zhao, S. H.; Liu, Y. Y. Chemistry 2015, 78, 505 (in Chinese). (杨会琴, 赵士豪, 刘月英, 化学通报, 2015, 78, 505.)
[29] Liu, K.; Chen, Y.; Wang, X. J.; Zhang, H. Org.-Fluorine Ind. 2013, 3, 58 (in Chinese). (刘康, 陈越, 王学军, 张恒, 有机氟工业, 2013, 3, 58.)
[30] Nardi, M.; Cozza, A.; Maiuolo, L.; Oliveriob, M.; Procopio, A. Tetrahedron Lett. 2011, 52, 4827.
[31] Wu, H. S.; Yang, J.; Wang, L. Chin. J. Chem. 2011, 29, 1721.
[32] Maiti, G.; Utpal, K.; Karmakar, R.; Bhattacharya, R. N. Tetrahedron Lett. 2012, 53, 1460.
[33] Nagawadea, R. R.; Shinde, D. B. Mendeleev Commun. 2006, 16, 113.
[34] Pawar, S. S.; Shingare, M. S.; Thore, S. N. Chin. Chem. Lett. 2009, 20, 32.
[35] Heravi, M. M.; Zadsirjan, V.; Behbahani, F. K.; Oskooie, H. A. J. Mol. Catal. A: Chem. 2006, 259, 201.
[36] Pandit, S. S.; Vikhe, B. D.; Shelke, G. J. Chem. Sci. 2007, 119, 295.
[37] Rekha, M.; Hamza, A.; Venugopal, B. R.; Nagaraju, N. Chin. J. Catal. 2012, 33, 439.
[38] Chatterjee, N.; Sarkar, S.; Pal, R.; Sen, A. K. Tetrahedron Lett. 2014, 55, 2261.
[39] Guo, P.; Zeng, X. M.; Chen, S.; Luo, M. M. J. Organomet. Chem. 2014, 751, 438.
[40] Bandgar, B. P.; Patl, A. V.; Chavan, O. S. J. Mol. Catal. A: Chem. 2006, 256, 99.
[41] Fazaeli, R.; Aliyan, H. Appl. Catal. A 2007, 331, 78.
[42] Mohammad, A. A.; Iraj, M. B.; Zaghaghi, Z. Catal. Commun. 2008, 9, 2496.
[43] Sharma, R. K.; Gulati, S.; Pandey, A. Inorg. Chim. Acta 2013, 397, 21.
[44] Maleki, A.; Kamalzare, M. Tetrahedron Lett. 2014, 55, 6931.
[45] An, Y. S.; Li, X. Q.; An, X. R.; Wang, L. Z. Monatsh. Chem. 2015, 146, 165.
[46] Wang, D. S.; Yan, L.; Wang, X. L. J. Mol. Catal. 2012, 26, 366 (in Chinese). (王德胜, 闫亮, 王晓来, 分子催化, 2012, 26, 366.)
[47] Heravia, M. M.; Derikvand, F.; Ranjbar, L.; Bamoharram, F. F. J. Mol. Catal. A: Chem. 2007, 261, 156.
[48] Pasquale, G. A.; Ruiz, D. M.; Jios, J. L.; Autinoa, J. C.; Romanelli, G. P. Tetrahedron Lett. 2013, 54, 6574.
[49] Li, X. Q.; Wang, L. Z. Chin. Chem. Lett. 2014, 25, 327.
[50] Das, B.; Ramu, R.; Ravikanth, B.; Reddy, V. S. J. Mol. Catal. A: Chem. 2006, 246, 76.
[51] Kuo, C. W.; More, S. V.; Yao, C. F. Tetrahedron Lett. 2006, 47, 8523.
[52] Sangshetti, J. N.; Kokare, N. D.; Shinde, D. B. Chin. Chem. Lett. 2007, 18, 1305.
[53] Murai, K.; Nakatani, R.; Kita, Y.; Fujioka, H. Tetrahedron 2008, 64, 11034.
[54] Lal, M.; Basha, R. S.; Sarkar, S.; Khan, A. T. Tetrahedron Lett. 2013, 54, 4264.
[55] Yadav, G. D.; Yadav, A. R. Ind. Eng. Chem. Res. 2013, 52, 17812.
[56] Sarkate, A. P.; Sangshetti, J. N.; Dharbale, N. B.; Sarkate, A. P.; Wakte, P. S.; Shinde, D. B. J. Chil. Chem. Soc. 2013, 58, 2200.
[57] Borodina, E. A.; Orlova, N. A.; Kargapolova, I. Y.; Gatilov, Y. V. J. Fluorine Chem. 2014, 162, 66.
[58] Tajbakhsh, M.; Hetavi, M. M.; Mohajerani, B.; Ahmadi, A. N. J. Mol. Catal. A: Chem. 2006, 247, 213.
[59] Vijayasankar, A. V.; Deepa, S.; Venugopal, B. R.; Nagaraju, N. Chin. J. Catal. 2010, 31, 1321.
[60] Shobha, D.; Chari, M. A.; Selvan, S. T.; Oveisib, H.; Manob, A.; Mukkantia, K.; Vinu, A. Microporous Mesoporous Mater. 2010, 129, 112.
[61] Jeganathan, M.; Pitchumani, K. ACS Sustainable Chem. Eng. 2014, 2, 1169.
[62] Yi, W. B.; Cai, C. J. Fluorine Chem. 2009, 130, 1054.
[63] Kurane, R.; Jadhav, J.; Khanapure, S.; Salunkhea, R.; Rashinkar, G. Green Chem. 2013, 15, 1849.
[64] Prakash, G. K. S.; Paknia, F.; Narayan, A.; Mathew, T.; Olah, G. A. J. Fluorine Chem. 2013, 152, 99.
[65] Jadhav, A. H.; Chinnappan, A.; Patil, R. H.; Kostjukb, S. V.; Kim, H. Chem. Eng. J. 2014, 240, 228.
[66] Naeimi, H.; Foroughi, H. Chin. J. Catal. 2015, 36, 734.
[67] Radatz, C. S.; Silva, R. B.; Perin, G.; Lenardão, E. J.; Jacob, R. G.; Alves, D. Tetrahedron Lett. 2011, 52, 4132.
[68] Xu, J. C.; Wei, J. M.; Bian, L. L.; Zhang, J. P.; Chen, J.; Deng, H. M.; Wu, X. Y.; Zhang, H.; Gao, W. Q. Chem. Commun. 2011, 47, 3607.
[69] Christophe, C.; Langlois, B. R.; Billard, T. J. Fluorine Chem. 2013, 155, 118.
[70] Solan, A.; Nisanci, B.; Belcher, M.; Young, J.; Schafer, C.; Wheeler, K. A.; Torok, B.; Dembinski, R. Green Chem. 2014, 16, 1120.
[71] Wang, S. S.; Wang, L. Z. Chem. Res. Appl. 2014, 26, 376 (in Chinese).
/
| 〈 |
|
〉 |