

光学活性γ-硝基酮与查尔酮的串联环化反应构建五手性中心的环己烷骨架
收稿日期: 2016-05-04
修回日期: 2016-05-31
网络出版日期: 2016-06-01
基金资助
南方科技大学2015年科研经费(No.FRG-SUSTC1501A-57)、国家自然科学基金(No.21302089)、杭州市科技发展计划(No.20130533B14)资助项目.
Tandem Cyclization Reaction between Optically Active γ-Nitro Ketone and Chalcone towards the Synthesis of Chiral Cyclohexane Skeletons Bearing Five Stereocenters
Received date: 2016-05-04
Revised date: 2016-05-31
Online published: 2016-06-01
Supported by
Project supported by the Research Grant in 2015 of the South University of Science and Technology of China (No. FRG-SUSTC1501A-57), the National Natural Science Foundation of China (No. 21302089) and the Science and Technology Development Program of Hangzhou (No. 20130533B14).
于璐 , 程玉宇 , 李柔 , 焦艳华 , 李鹏飞 . 光学活性γ-硝基酮与查尔酮的串联环化反应构建五手性中心的环己烷骨架[J]. 有机化学, 2016 , 36(7) : 1572 -1579 . DOI: 10.6023/cjoc201605006
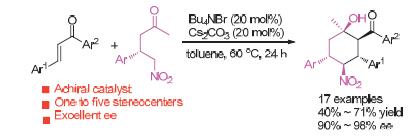
Chiral cyclohexane motif is widespread in the molecular structures of nature products and medicinal chemicals. And polysubstituted chiral cyclohexanes are important building blocks in organic synthesis. A tandem cyclization via asymmetric induction for the construction of polysubstituted chiral cyclohexane skeletons is described. In the presence of phase-transfer-catalyst, optically active γ-nitro ketone reacted with chalcone smoothly to afford cyclohexane skeletons bearing five consecutive stereocenters in 40%~71% yields with 90%~98% ee. It should be noted that the scope of chalcone is broad. Especially, the asymmetric induction is kept at a high level in each step of the tandem cyclization reaction.
Key words: nitro ketone; chalcone; tandem cyclization reaction; cyclohexane
[1] For selected reviews, see: (a) Tietze, L. F. Chem. Rev. 1996, 96, 115.
(b) Parsons, P. J.; Penkett, C. S.; Shell, A. J. Chem. Rev. 1996, 96, 195.
(c) Pellissier, H. Tetrahedron 2006, 62, 1619.
(d) Pellissier, H. Tetrahedron 2006, 62, 2143.
(e) Nicolaou, K. C.; Edmonds, D. J.; Bulger, P. G. Angew. Chem., Int. Ed. 2006, 45, 7134.
(f) Chapman, C. J.; Frost, C. G. Synthesis 2007, 1.
(g) Enders, D.; Grondal, C.; Hüttl, M. R. M. Angew. Chem., Int. Ed. 2007, 46, 1570.
(h) Nicolaou, K. C.; Chen, J. S. Chem. Soc. Rev. 2009, 38, 2993.
(i) Grondal, C.; Jeanty, M.; Enders, D. Nat. Chem. 2010, 2, 167.
(j) Ruiz, M.; López-Alvarado, P.; Giorgi, G., Menéndez, J. C. Chem. Soc. Rev. 2011, 40, 3445.
(k) Albrecht, ?.; Jiang, H.; Jørgensen, K. A. Angew. Chem., Int. Ed. 2011, 50, 8492.
(l) Pellissier, H. Adv. Synth. Catal. 2012, 354, 237.
(m) Pellissier, H. Chem. Rev. 2013, 113, 442.
(n) Volla, C. M. R.; Atodiresei, I.; Rueping, M. Chem. Rev. 2014, 114, 2390.
(o) Vetica, F.; de Figueiredo, R. M.; Orsini, M.; Tofani, D.; Gasperi, T. Synthesis 2015, 47, 2139.
[2] (a) Merino, P.; Marqués-López, E.; Tejero, T.; Herrera, R. P. Synthesis 2010, 1.
(b) Moyano, A.; Rios, R. Chem. Rev. 2011, 111, 4703.
(c) Pellissier, H. Tetrahedron 2012, 68, 2197.
(d) Xie, P.; Huang, Y. Eur. J. Org. Chem. 2013, 6213.
(e) Yang, X.; Wang, J.; Li, P. Org. Biomol. Chem. 2014, 12, 2499.
[3] (a) Posner, G. H. Chem. Rev. 1986, 86, 831.
(b) Schultz, A. G. Acc. Chem. Res. 1990, 23, 207.
(c) Li J. K. In Name Reaction for Carbocyclic Ring Formarions, Wiley, Hoboken, NJ, 2010, p. 197~142.
(d) Goudedranche, S.; Raimondi, W.; Bugaut, X.; Constantieux, T.; Bonne, D.; Rodriguez, J. Synthesis 2013, 45, 1909.
[4] (a) Enders, D.; Hüttl, M. R. M.; Grondal, C.; Raabe, G. Nature 2006, 441, 861.
(b) Enders, D.; Hüttl, M. R. M.; Runsink, J.; Raabe, G.; Wendt, B. Angew. Chem., Int. Ed. 2007, 46, 467.
(c) Enders, D.; Hüttl, M. R. M.; Raabe, G.; Bats, J. W. Adv. Synth. Catal. 2008, 350, 267.
(d) Ishikawa, H.; Suzuki, T.; Orita, H.; Uchimaru, T.; Hayashi, Y. Chem. Eur. J. 2010, 16, 12616.
(e) Anwar, S.; Chang, H.-J.; Chen, K. Org. Lett. 2011, 13, 2200.
(f) Ma, G.; Lin, S.; Ibrahem, I.; Kubik, G.; Liu, L.; Sun, J.; Córdova, A. Adv. Synth. Catal. 2012, 354, 2865.
[5] (a) Han, B.; Xiao, Y.-C.; He, Z.-Q.; Chen, Y.-C. Org. Lett. 2009, 11, 4660.
(b) Wang, Y.; Yu, D.-F.; Liu, Y.-Z.; Wei, H.; Luo, Y.-C.; Dixon, D. J.; Xu, P.-F. Chem. Eur. J. 2010, 16, 3922.
[6] (a) Ishikawa, H.; Sawano, S.; Yasui, Y.; Shibata, Y.; Hayashi, Y. Angew. Chem., Int. Ed. 2011, 50, 3774.
(b) Hahn, R.; Raabe, G.; Enders, D. Org. Lett. 2014, 16, 3636.
[7] (a) Mao, Z.; Jia, Y.; Xu, Z.; Wang, R. Adv. Synth. Catal. 2012, 354, 1401.
(b) Enders, D.; Hahn, R.; Atodiresei, I. Adv. Synth. Catal. 2013, 355, 1126.
(c) Chauhan, P.; Mahajan, S.; Loh, C. C. J.; Raabe, G.; Enders, D. Org. Lett. 2014, 16, 2954.
(d) Chauhan, P.; Urbanietz, G.; Raabe, G.; Enders, D. Chem. Commun. 2014, 50, 6853.
(e) Zhou, R.; Wu, Q.; Guo, M.; Huang, W.; He, X.; Yang, L.; Peng, F.; He, G.; Han, B. Chem. Commun. 2015, 51, 13113.
[8] Xie, X.; Peng, C.; He, G.; Leng, H.-J.; Wang, B.; Huang, W.; Han, B. Chem. Commun. 2012, 48, 10487.
[9] (a) Varga, S.; Jakab, G.; Drahos, L.; Holczbauer, T.; Czugler, M.; Soós, T. Org. Lett. 2011, 13, 5416.
(b) Sun, J.; Jiang, C.; Zhou, Z. Eur. J. Org. Chem. 2016, 1165.
[10] Duan, J.-D.; Cheng, J.; Li, P.-F. Org. Chem. Front. 2015, 2, 1048.
[11] Yu, L.; Yang, Z.; Peng, J.-Z.; Li, P.-F. Eur. J. Org. Chem. 2016, 535.
[12] Yu, L.; Yang, Q.-J.; Li, P.-F. Eur. J. Org. Chem. 2014, 7499.
[13] Huang, H.; Jacobsen, E. N. J. Am. Chem. Soc. 2006, 128, 7170.
/
| 〈 |
|
〉 |