

有机合成中腈的去对称化生物转化反应研究进展
收稿日期: 2016-05-09
修回日期: 2016-06-11
网络出版日期: 2016-06-20
基金资助
国家自然科学基金(No.21502202)资助项目.
Biocatalytic Desymmetrization of Dinitriles in Organic Synthesis
Received date: 2016-05-09
Revised date: 2016-06-11
Online published: 2016-06-20
Supported by
Project supported by the National Natural Science Foundation of China (No.21502202).
敖宇飞 , 王其强 , 王德先 . 有机合成中腈的去对称化生物转化反应研究进展[J]. 有机化学, 2016 , 36(10) : 2333 -2343 . DOI: 10.6023/cjoc201605009
In comparison with the chemical hydration and hydrolysis of nitriles, which usually involves harsh reaction condi-tions and low selectivity, biocatalytic desymmetrizations of prochiral or meso nitriles are highly efficient, highly enantioselective and environmentally benign. Therefore, biocatalysis and biotransformation has offered an attractive and unique protocol for the enantioselective synthesis of polyfunctionalized organic compounds that are not readily obtainable by other methods. This review summarizes the biocatalytic desymmetrization of prochiral nitriles including glutaronitriles, malonitriles and meso cyclic dinitriles during the past two decades.
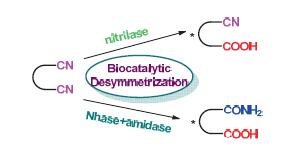
Key words: desymmetrization; biotransformations; dinitriles
[1] Faber, K. Biotransformations in Organic Chemistry, 6th ed., Springer, Berlin, 2011.
[2] Rappoport, Z.; Patai, S. The Chemistry of Functional Groups, The Chemistry of the Cyano Group, Wiley, London, 1970.
[3] (a) Evgred, D.; Harnett, S. Cyanide Compounds in Biology (Ciba Foundation Symposium 140), Wiley, Chichester, 1998. (b) Jallageas, J.-C.; Arnaud, A.; Galzy, P. Adv. Biochem. Eng. 1980, 12, 1. (c) Legras, J.-L.; Chuzel, G.; Arnaud, A.; Galzy, P. World J. Microbiol. Biotechnol. 1990, 6, 83.
[4] (a) Harper, D. B. Biochem. Soc. Trans. 1976, 4, 502. (b) Harper, D. B. Biochem. J. 1977, 165, 309. (c) Kobayashi, M.; Shimizu, S. FEBS Microbiol Lett. 1994, 120, 217.
[5] (a) Asano, Y.; Tani, Y.; Yamada, H. Agric. Biol. Chem. 1980, 44, 2251. (b) Asano, Y.; Tachibana, Y.; Tani, Y.; Yamada, H. Agric. Biol. Chem. 1982, 46, 1175.
[6] (a) Brenner, C. Curr. Opin. Struct. Biol. 2002, 12, 775. (b) Liu, Z. -Q.; Dong, L. -Z.; Cheng, F.; Xue, Y. -P.; Wang, Y. -S.; Ding, J. -N.; Zheng, Y. -G.; Shen, Y. -C. J. Agric. Food Chem. 2011, 59, 11560.
[7] (a) Mascharak, P. K. Coord. Chem. Rev. 2002, 225, 201. (b) Song, L. Y.; Wang, M. Z.; Shi, J. J.; Xue, Z. Q.; Wang, M. -X.; Qian, S. J. Biochem. Biophy. Res. Commun. 2007, 362, 319.
[8] (a) Fournand, D.; Arnaud, A. J. Appl. Microbiol. 2001, 91, 381. (b) Ohtaki, A.; Murata, K.; Sato, Y.; Noguchi, K.; Miyatake, H.; Dohmae, N.; Yamada, K.; Yohda, M.; Odaka, M. Biochim. Biophys. Acta 2010, 1804, 184.
[9] (a) Sugai, T.; Yamazaki, T.; Yokoyama, M.; Ohta, H. Biosci. Biotechnol. Biochem. 1997, 61, 1419. (b) Martínková, L.; K?en, V. Biocatal. Biotrans. 2002, 20, 73. (c) Banerjee, A.; Sharma, R. Banerjee, U. C. Appl. Microbiol. Biotechnol. 2002, 60, 33. (d) Wang, M.-X. Top. Catal. 2005, 35, 117. (e) Martínková, L.; Uhnáková, B.; Pátek, M.; Nešvera, J.; K?en, V. Rhodococcus. Environ. Int. 2009, 35, 162. (f) Wang, M.-X. Chimia 2009, 63, 331. (g) Prasad, S.; Bhalla, T. C. Biotechnol. Adv. 2010, 28, 725; (h) Velankar, H.; Clarke, K. G.; du Preez, R.; Cowan, D. A.; Burton, S. G. Trends Biotechnol. 2010, 28, 561. (i) Wang, M.-X. Top. Organomet. Chem. 2011, 36, 105. (j) Ramteke, P. W.; Maurice, N. G.; Joseph, B.; Wadher, B. J. Biotechnol. Appl. Biochem. 2013, 60, 459. (k) Wang, M.-X. Acc. Chem. Res. 2015, 48, 602.
[10] (a) Garcia-Urdiales, E.; Alfonso, I.; Gotor, V. Chem. Rev. 2005, 105, 313. (b) Palomo, J. M.; Cabrera, Z. Curr. Org. Synth. 2012, 9, 791.
[11] Kakya, H.; Sakai, N.; Yokoyama, M.; Sugai, T.; Ohta, H. Chem. Lett. 1991, 1823.
[12] (a) Crosby, J. A.; Parratt, J. S.; Turner, N. J. Tetrahedron:Asymmetry 1992, 3, 1547. (b) Beard, T.; Cohen, M. A.; Parratt, J. S.; Turner, N. J.; Crosby, J.; Moilliet, N. J. Tetrahedron:Asymmetry 1993, 4, 1085.
[13] (a) Wang, M.-X.; Liu, C.-S.; Li, J.-S. Meth-Cohn, O. Tetrahedron Lett. 2000, 41, 8549. (b) Wang, M.-X.; Liu, C.-S.; Li, J.-S. Tetrahedron:Asymmetry 2002, 12, 3367.
[14] Vink, M. K. S.; Schortinghuis, C. A.; Luten, J.; van Maarseveen, J. H.; Schoemaker, H. E.; Hiemstra, H.; Rutjes, F. P. J. T. J. Org. Chem. 2002, 67, 7869.
[15] (a) Santis, D. G.; Zhu, Z. L.; Greenberg, W. A.; Wong, K.; Chaplin, J.; Hanson, S. R.; Farwell, B.; Nicholson, L. W.; Rand, C. L.; Weiner, D. P.; Robertson, D. E.; Burk, M. J. J. Am. Chem. Soc. 2002, 124, 9024. (b) Santis, D. G.; Wong, K.; Farwell, B.; Chatman, K.; Zhu, Z. L.; Tomlinson, G.; Huang, H.; Tan, X.; Bibbs, L.; Chen, P.; Kretz, K.; Burk, M. J. J. Am. Chem. Soc. 2003, 125, 11476.
[16] Bergeron, S.; Chaplin, D. A.; Edwards, J. H.; Ellis, B. S. W.; Hill, C. L.; Karen, H.-T.; Knight, J. R.; Mahoney, T.; Osborne, A. P.; Ruecroft, G. Org. Process Res. Dev. 2006, 10, 661.
[17] Kinfe, H. H.; Chhiba, V.; Frederick, J.; Bode, M. L.; Mathiba, K.; Steenkamp, P. A.; Brady, D. J. Mol. Catal. B:Enzym. 2009, 59. 231.
[18] Xu, M. Z.; Ren, J.; Gong, J. S.; Dong, W. Y.; Wu, Q. Q.; Xu, Z. H.; Zhu, D. M. Chin. J. Biotechnol. 2013, 29, 31(in Chinese). (许美珍, 任杰, 龚劲松, 董文玥, 吴洽庆, 许正宏, 朱敦明, 生物工程学报, 2013, 29, 31.)
[19] Duan, Y. T.; Yao, P. Y.; Ren, J.; Han, C.; Li, Q.; Yuan, J.; Feng, J. H.; Wu, Q. Q.; Zhu, D. M. Sci. Chin. Chem. 2014, 57, 1164.
[20] Nojiri, M.; Uekita, K.; Ohnuki, M.; Taoka, N.; Yasohara, Y. J. Appl. Microbiol. 2013, 115, 1127.
[21] Yokoyama, M.; Sugai, T.; Ohta, H. Tetrahedron:Asymmetry 1993, 4, 1081.
[22] Yokoyama, M.; Kashiwagi, M.; Iwasaki, M.; Fuhshuku, K.; Ohta, H.; Sugai, T. Tetrahedron:Asymmetry 2004, 15, 2817.
[23] (a) Wu, Z. -L.; Li, Z. -Y. Chem. Commun. 2003, 386. (b) Wu, Z. -L.; Li, Z. -Y. J. Org. Chem. 2003, 68, 2479. (c) Wu, Z. -L.; Li, Z. -Y. Tetrahedron:Asymmetry 2003, 14, 2133.
[24] Vink, M. K. S.; Wijtmans, R.; Reisinger, C.; Berg, R. J. F.; Schortinghuis, C. A.; Schwab, H.; Schoemaker, H. E.; Rutjes, F. P. J. T. Biotechnol. J. 2006, 1, 569.
[25] Zhang, L.-B.; Wang, D.-X.; Wang, M.-X. Tetrahedron 2011, 67, 5604.
[26] Zhang, L.-B.; Wang, D.-X.; Zhao, L.; Wang, M.-X. J. Org. Chem. 2012, 77, 5584.
[27] Matoishi, K.; Sano, A.; Imai, N.; Yamazaki, T.; Yokoyama, M.; Sugai, T.; Ohta, H. Tetrahedron:Asymmetry 1998, 9, 1097.
[28] (a) Chen, P.; Gao, M.; Wang, D.-X.; Zhao, L.; Wang, M.-X. Chem. Common. 2012, 48, 3482. (b) Chen, P.; Gao, M.; Wang, D.-X.; Zhao, L.; Wang, M.-X. J. Org. Chem. 2012, 77, 4063.
[29] Ao, Y.-F.; Wang, D.-X.; Zhao, L.; Wang, M.-X. Chem. Asian J. 2015, 10, 938.
[30] (a) Kielbasinski, P.; Rachwalski, M.; Mikolajczyk, M.; Szyrej, M.; Wieczorek, M. W.; Wijtmans, R.; Rutjes, F. P. J. T. Adv. Synth. Catal. 2007, 349,1387. (b) Kielbasinski, P.; Rachwalski, M.; Kwiatkowska, M.; Mikolajczyk, M.; Wieczorek, M. W.; Szyrej, M.; Sieron, L.; Rutjes, F. P. J. T. Tetrahedron:Asymmetry 2007, 18, 2108.
[31] Fernandes, B. C. M.; Mateo, C.; Kiziak, C.; Chmura, A.; Wacker, J.; Rantwijk, F. V.; Stolz, A.; Sheldon, R. A. Adv. Synth. Catal. 2006, 348, 2597.
/
| 〈 |
|
〉 |