

展毛大渡乌头全草中的新二萜生物碱
收稿日期: 2016-04-26
修回日期: 2016-06-21
网络出版日期: 2016-07-13
基金资助
国家自然科学基金(No.81274193)、四川省小分子天然药物创新团队(No.15TD0048)、中央高校基本科研业务费专项(No.2682015YXZT03)和西南交通大学2015年研究生创新实验实践(No.YC201510102)资助项目.
New Ditetpenoid Alkaloid from the Whole Plant of Aconitum franchetii var. villosulum
Received date: 2016-04-26
Revised date: 2016-06-21
Online published: 2016-07-13
Supported by
Project supported by the National Natural Science Foundation of China (No.81274193),the Small Molecule Natural Medicine Innovation Team (No.15TD0048),the Fundamental Research Funds for Central Universities (No.2682015YXZT03) and the Southwest Jiaotong University Graduate Students Creative Experiment (No.YC201510102).
从展毛大渡乌头(Aconitum franchetii var.villosulum)中分离得到11个C19-二萜生物碱,经过HR-ESIMS,1D和2D NMR等波谱技术确定了它们的结构.其中一个为新的乌头碱型二萜生物碱,villosutine(1).其余10个为已知化合物,包含4个乌头碱型二萜生物碱、1个热解型二萜生物碱和5个大渡乌碱型二萜生物碱.
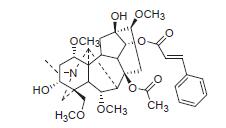
关键词: 乌头属; 展毛大渡乌头; 二萜生物碱; villosutine
徐文亮 , 单连海 , 黄帅 , 李遂焰 , 周先礼 . 展毛大渡乌头全草中的新二萜生物碱[J]. 有机化学, 2016 , 36(11) : 2739 -2742 . DOI: 10.6023/cjoc201604055
A new aconitine-type diterpenoid alkaloid, villosutine (1), was isolated from the whole plant of Aconitum franchetii var. villosulum, together with four other known aconitine-type diterpenoid alkaloids, one known pyro-type diterpenoid alkaloid and five known franchetine-type diterpenoid alkaloids. Their structures were elucidated on the basis of extensive spectroscopic analy-sis, including HR-ESIMS, 1D and 2D NMR experiments.

[1] Xiao, P.-G.; Wang, F.-P.; Gao, F.; Yan, L.-P.; Chen, D.-L.; Liu, Y. Acta Phytotaxon. Sin. 2006, 44, 1 (in Chinese). (肖培根, 王峰鹏, 高峰, 闫路平, 陈东林, 刘勇, 植物分类学报, 2006, 44, 1.)
[2] Institute of Botany, Chinese Academy of Sciences and Institute of Materia Medica, Chinese Academy of Medical Sciences Flora Reipublicae Populais Sinica, Vol. 27, Science Press, Beijing, 1979, p. 211 (in Chinese). (中国科学院植物所, 中国医学科学院药物所, 中国植物志, Vol. 27, 科学出版社, 北京, 1979, p. 211.)
[3] Pelletier, S. W.; Mody, N. V.; Sawhney, R. S.; Bhattacharyya, J. Heterocycles 1977, 7, 327.
[4] Gabrieldela, F.; Rafael, D. A.; Tomas, O. Heterocycles 1989, 29, 205.
[5] Boido, V.; Edwards, O. E.; Handa, K. L.; Kolt, R. J. Can. J. Chem. 1984, 62, 778.
[6] Pelletier, S. W.; Mody, N. V.; Puri, H. S. J. Chem. Soc., Chem. Commun. 1977, 1, 12
[7] Wang, F.-P.; Li, Z.-B.; Dai, X.-P.; Peng, C.-S. Phytochemstry 1997, 45, 1539.
[8] Peng, C.-S.; Dai, X.-P.; Chen, D.-L.; Wang, F.-P. Nat. Prod. Res. Dev. 1999, 11, 23 (in Chinese). (彭崇胜, 代雪平, 陈东林, 王锋鹏, 天然产物研究与开发, 1999, 11, 23.)
[9] Chen, D.-L.; Lin, L.-Y.; Chen, Q.-H.; Jian, X.-X.; Wang, F.-P. J. Asian Nat. Prod. Res. 2003, 5, 209.
[10] Chen, D.-L.; Chen, Q.-H.; Jian,X.-X.; Wang,F.-P. Nat. Prod. Res. Dev. 2002, 14, 6 (in Chinese). (陈东林, 陈巧红, 简锡贤, 王峰鹏, 天然产物研究与开发, 2002, 14, 6.)
[11] Zhang, F.; Peng, S.-L.; Luo, F.; Ding, L. Chin. Chem. Lett. 2005, 16, 1046.
[12] Chen, L.; Shan, L.-H.; Zhang, J.-F.; Xu,W.-L.; Wu, M.-Y.; Huang, S.; Zhou, X.-L. Nat. Prod. Commun. 2015, 10, 2063.
[13] Wang, F.-P. Chin. J. Org. Chem. 1982, 2, 161 (in Chinese). (王锋鹏, 有机化学, 1982, 2, 161.)
/
| 〈 |
|
〉 |