

硒催化羰基化合成苯氨基甲酸酯和二苯甲烷二氨基甲酸酯
收稿日期: 2016-09-12
修回日期: 2016-10-12
网络出版日期: 2016-10-18
基金资助
河南省高校科技创新团队支持计划(No.15IRTSTHN003)、河南省高等学校青年骨干教师(No.2013GGJS-059)和河南师范大学青年骨干教师(No.2011-8)资助项目.
Selenium-Catalyzed Carbonylation to Phenylcarbamates and Methylene Diphenyl Dicarbamates
Received date: 2016-09-12
Revised date: 2016-10-12
Online published: 2016-10-18
Supported by
Project supported by the Program for Innovative Research Team in Science and Technology in University of Henan Province (No. 15IRTSTHN003), the Young Backbone Teachers Training Fund of Education Department of Henan Province (No. 2013GGJS-059) and the Young Backbone Teachers Training Fund of Henan Normal University (No. 2011-8).
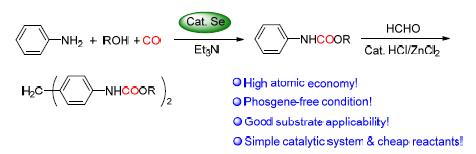
首先以廉价易得的非金属硒作催化剂,CO替代剧毒光气作羰基化试剂,O2作氧化剂,通过硒催化苯胺与醇“一锅煮”的氧化羰基化反应制得苯氨基甲酸酯;然后在HCl/ZnCl2催化下再与甲醛缩合制得二苯甲烷二氨基甲酸酯.两反应的目标产物收率从中等到良好,底物普遍适用性广.CO利用率及原子经济性高,且避免了剧毒光气的使用.催化剂硒具有相转移催化作用,便于回收且能循环使用.合成方法清洁、经济、高效.提出了可能的反应机理.
关键词: 硒催化; 羰基化; 苯氨基甲酸酯; 二苯甲烷二氨基甲酸酯
张晓鹏 , 荆唤芝 , 彭惟于 , 李亚芳 , 范学森 , 张贵生 . 硒催化羰基化合成苯氨基甲酸酯和二苯甲烷二氨基甲酸酯[J]. 有机化学, 2017 , 37(2) : 411 -417 . DOI: 10.6023/cjoc201609013
A clean, economical and efficient approach to phenylcarbamates and methylene diphenyl dicarbamates was reported. With cheap and easily available nonmetal selenium as the catalyst, carbon monoxide instead of virulent phosgene as the carbonylation reagent, oxygen as the oxidant, the selenium-catalyzed oxidative carbonylation reaction of aniline could proceed smoothly with alcohols to afford phenylcarbamates mostly in moderate to good yields. Then, catalyzed by HCl/ZnCl2, the condensation of formaldehyde with the generated phenylcarbamates gave methylene diphenyl dicarbamates in moderate to good yields. The applicability of the substrates was good. Catalyst selenium could be easily recovered due to its function of phase-transfer catalysis and could be recycled. High atomic economy, low cost, no emission of corrosive waste, and phosgene-free condition make this approach very promising. The possible reaction mechanisms were also proposed.

[1] (a) Jia C. Q.; Yang D. Y.; Che C. L.; Ma Y. Q.; Rui C. H.; Yan X. J.; Tan Z. H. Chem. J. Chin. Univ. 2016, 37, 892 (in Chinese).(贾长青, 杨冬燕, 车传亮, 马永强, 芮昌辉, 闫晓静, 覃兆海, 高等学校化学学报, 2016, 37, 892.)
(b) Wu, X. P.; Wang, L.; Xie, Z. H.; Yan, C.; Chen, G. N. Chem. Res. Chin. Univ. 2007, 23, 109.
(c) Gupte, S. P.; Shivarkar, A. B.; Chaudhari, R. V. Chem. Commun. 2001, 2620.
(d) Kodaka, R.; Sugano, T.; Katagi, T.; Takimoto, Y. J. Agric. Food Chem. 2003, 51, 7730.
(e) Srivastava, R.; Manju, M. D.; Srinivas, D.; Ratnasamy, P. Catal. Lett. 2004, 97, 41.
[2] (a) Tafesh, A. M.; Weiguny, J. Chem. Rev. 1996, 96, 2035.
(b) Paul, P. Coord. Chem. Rev. 2000, 203, 269.
(c) Baba, T.; Kobayashi, A.; Kawanami, Y.; Inazu, K.; Ishikawa, A.; Echizenn, T.; Murai, K.; Aso, S.; Inomata, M. Green Chem. 2005, 7, 159.
(d) Shi, F.; Deng, Y. Q.; Sima, T. L.; Yang, H. Z. J. Catal. 2001, 203, 525.
[3] (a) White, J. D.; Blakemore, P. R.; Milicevic, S. Org. Lett. 2002, 4, 1803.
(b) Feroci, M.; Casadei, M. A.; Orsini, M.; Palombi, L.; Ines, A. J. Org. Chem. 2003, 68, 1548.
(c) Deleon, R. G.; Kobayashi, A.; Yamauchi, T.; Ooishi, J.; Baba, T.; Sasaki, M.; Hiarata, F. Appl. Catal., A 2002, 225, 43.
[4] (a) Crosby, D. G.; Niemann, C. J. Am. Chem. Soc. 1954, 76, 4458.
(b) Zhang, A.; Kuwahara, Y.; Hotta, Y.; Tsuda, A. Asian J. Org. Chem. 2013, 2, 572.
[5] (a) Uhlig, N.; Li, C. J. Chem.-Eur. J. 2014, 20, 12066.
(b) Seth, K.; Nautiyal, M.; Purohit, P.; Parikh, N.; Chakraborti, A. K. Chem. Commun. 2015, 51, 191.
(c) Izawa, Y.; Ishiguro, K.; Tomioka, H. Bull. Chem. Soc. Jpn. 1983, 56, 951.
(d) Padiya, K. J.; Gavade, S.; Kardile, B.; Tiwari, M.; Bajare, S.; Mane, M.; Gaware, V.; Varghese, S.; Harel, D.; Kurhade, S. Org. Lett., 2012, 14, 2814.
(e) Kang, W. K.; Wang, G. Y.; Yao, J. Chem. Res. Chin. Univ. 2006, 22, 669.
[6] Ragaini, F.; Gasperini, M.; Cenini, S. Adv. Synth. Catal. 2004, 346, 63.
[7] Yang, Y.; Lu, S. W. Chin. J. Catal. 1999, 20, 224 (in Chinese).(杨瑛, 陆世维, 催化学报, 1999, 20, 224.)
[8] (a) Shi, F.; Sima, T. L.; Deng, Y. Q. Chem. J. Chin. Univ. 2000, 21, 1566 (in Chinese).(石峰, 司马天龙, 邓友全, 高等学校化学学报, 2000, 21, 1566.)
(b) Mei, F. M.; Chen, L. J.; Li, G. X. Appl. Organomet. Chem. 2010, 24, 86.
(c) Shi, F.; Deng, Y. Q. Chem. Commun. 2001, 443.
[9] Stock, C.; Brueckner, R. Adv. Synth. Catal. 2012, 354, 2309.
[10] (a) Pei, Y. X.; Li, H. Q.; Liu, H. T.; Zhang, Q. H.; Zhang, Y. Chem. Res. Chin. Univ. 2010, 26, 550.
(b) Kim, S. D.; Lee, K. H. J. Mol. Catal. 1993, 78, 237.
[11] (a) Zhang, X. P.; Miao, J. H.; Sun, Y. B. Chin. J. Org. Chem. 2009, 29, 1555 (in Chinese).(张晓鹏, 苗江欢, 孙玉标, 有机化学, 2009, 29, 1555.)
(b) Sonoda, N. Pure Appl. Chem. 1993, 65, 699.
[12] Zhang, X. P.; Li, D. S.; Ma, X. J.; Wang, Y.; Zhang, G. S. Synthesis 2013, 45, 1357.
[13] Kang, L. J.; Zhao, X. Q.; An, H. L.; Wan, Y. J. Acta Pet. Sin. 2013, 29, 249 (in Chinese).(康丽娟, 赵新强, 安华良, 王延吉, 石油学报, 2013, 29, 249.)
[14] Kianmehr, E.; Baghersad, M. H. Adv. Synth. Catal. 2011, 353, 2599.
[15] George, D. K.; Moore, D. H.; Brian, W. P.; Garman, J. A. J. Agric. Food Chem. 1954, 2, 356.
[16] Whitmore, F. C.; Popkin, A. H.; Whitaker, J. S.; Mattil, K. F.; Zech, J. D. J. Am. Chem. Soc. 1938, 60, 2462.
[17] Schicktanz, S. T.; Etienne, A. D.; Steele, W. I. Ind. Eng. Chem. Anal. Ed. 1939, 11, 420.
[18] Bayer, F. GB 775723, 1957 [Chem. Abstr. 1957, 51, 90827].
[19] Brockway, C. E. US 2806051, 1957 [Chem. Abstr. 1957, 52, 15931].
[20] Murphy, W. S.; Raman, K. P. J. Chem. Soc., Perkin Trans. 1 1981, 447.
[21] Matthews, K. H.; Mclennaghan, A.; Pethrick, R. A. Brit. Polym. J. 1987, 19, 165.
[22] Tereshatov, V. V.; Senichev, V. Y. J. Appl. Polym. Sci. 2015, 41481.
[23] Chapman,T. M. J. Appl. Polym. Sci. Part A: Polym. Chem. 1989, 27, 1993.
[24] Lu, Q. W.; Hoye, T. R.; Macosko, C. W. Thermoplastic Polyurethane Blends 2002, 2310.
/
| 〈 |
|
〉 |