

一种合成二氢黄酮醇衍生物的简易方法
收稿日期: 2016-05-30
修回日期: 2016-06-25
网络出版日期: 2016-07-15
基金资助
国家自然科学基金(No.31370378)资助项目.
A Facile Method to Prepare Dihydroflavonol Derivatives
Received date: 2016-05-30
Revised date: 2016-06-25
Online published: 2016-07-15
Supported by
Project supported by the National Natural Science Foundation of China (No.31370378).
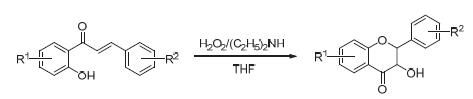
以2'-羟基查尔酮衍生物为原料,在低温条件下,以四氢呋喃为溶剂,在H2O2/(C2H5)2NH体系中,高收率地实现一系列二氢黄酮醇类衍生物的合成.考查了反应温度、反应溶剂、碱添加剂的种类和比例以及氧化剂的种类和比例对该反应的影响,确定了最优反应条件.此合成方法具有操作简单、条件温和及后处理方便等特点.此外,所合成产物的结构经核磁共振谱表征确证.
关键词: 2'-羟基查尔酮衍生物; 二氢黄酮醇类衍生物; 简易的方法
周宗宝 , 王红 , 陶凯奇 , 叶晓川 , 胡春玲 . 一种合成二氢黄酮醇衍生物的简易方法[J]. 有机化学, 2016 , 36(11) : 2752 -2756 . DOI: 10.6023/cjoc201605044
A series of dihydroflavonol derivatives were synthesized by the reaction of 2'-hydroxyl chalcone derivatives with H2O2/(CH3CH2)2NH system in anhydrous tetrahydrofuran under low temperature. The influences of reaction temperature, re-action solvents, the kinds and proportion of alkaline additive and oxidants on this reaction were investigated, and the optimal reaction conditions were determined. This method has the advantages of simple operation, mild conditions and convenient post-treatment. Besides, all synthetic products were fully characterized by NMR spectra.

[1] Hiroyuki, H.; Yasutomo, M.; Sachiko, S.; Hitoshi, M.; Yukiyoshi, T.; Kenji, M.; Osamu, T.; Chou, W.-H. Biosci. Biotechnol. Biochem. 1996, 60, 945.
[2] Sugihara, N.; Arakawa, T.; Ohnishi, M.; Furuno, K. Free Radical Biol. Med. 1999, 27, 1313.
[3] Matsumoto, T.; Tahara, S. Nippon Nogeikagaku Kaishi. 2001, 75, 659.
[4] Devi, M. A.; Das, N. P. Cancer Lett. 1993, 69, 191.
[5] Kandaswami, C.; Perkins, E.; Drzewiecki, G.; Soloniuk, D. S.; Middleton, E. J. Anti-Cancer Drugs. 1992, 3, 525.
[6] Mecocci, P.; Polidori, M. C.; Praticó, D. Biochim. Biophys. Acta. 2012, 18, 631.
[7] Sato, M.; Murakami, K.; Uno, M.; Nakagawa, Y.; Katayama, S.; Akagi, K. I.; Masuda, Y.; Takegoshi, K.; Irie, K. J. Biol. Chem. 2013, 288, 23213.
[8] Shi, L.; Feng, X. E.; Cui, J. R.; Fang, L. H.; Du, G. H.; Li, Q. S. Bioorg. Med. Chem. Lett. 2010, 20, 5466.
[9] Pandurangan, N.; Bose, C.; Banerji, A. Bioorg. Med. Chem. Lett. 2011, 21, 5328.
[10] Mei, Q. G.; Yuan, W. C.; Wang, C. Chin. J. Org. Chem. 2015, 35, 70 (in Chinese). (梅青刚, 袁伟成, 王淳, 有机化学, 2015, 35, 70.)
[11] Brown, N. B.; Macbride, J. A. H. J. Chem. Soc. 1964, 3822.
[12] Shabnam, S.; Makrandl, J. K.; Grover, S. K. Synthesis 1985, 11, 110.
[13] Loser, R.; Chlupacova, M.; Marecek, A.; Opletalova, V.; Gutschow, M. Helv. Chim. Acta. 2004, 87, 2597.
[14] Oyamada, T.; Baba, H.; Koyama, T. Bull. Yamagata Univ., Eng. 1967, 9, 351.
[15] Tanaka, K.; Sugino, T. Green Chem. 2001, 3, 133.
[16] Cummins, B.; Donnelly, D. M. X.; Eades, J. F.; Fletcher, H.; Cinnéide, F. O.; Philbin, E. M.; Swirski, J.; Wheeler, T. S.; Wilson, R. K. Tetrahedron 1963, 19, 499.
[17] Patonay, T.; Tóth, G.; Adam, W. Tetrahedron Lett. 1993, 34, 5055.
[18] Algar, J.; Flynn, J. P. Proc. R. Ir. Acad. 1934, B42, 1.
[19] Oyamada, T. J. Chem. Soc. Jpn. 1934, 55, 1256.
[20] Geissman, T. A.; Fukushima, D. K. J. Am. Chem. Soc. 1948, 70, 1686.
[21] Hart, H.; Verma, M.; Wang, I. J. Org. Chem. 1973, 38, 3418.
[22] Tanaka, H.; Stohlmeyer, M. M.; Wandless, T. J.; Taylor, L. P. Tetrahedron Lett. 2000, 41, 9735.
[23] Miao, C. X.; Hong, Y. Y.; Deng, Y. H. Chem. Reag. 2008, 30, 63 (in Chinese). (苗彩霞, 洪镛裕, 邓优华, 化学试剂, 2008, 30, 63.)
[24] Miao, T. G.; Wang, Q. A.; Fang, W. Q. Chin. J. Org. Chem. 2006, 26, 685 (in Chinese). (廖头根, 汪秋安, 方伟琴, 有机化学, 2006, 26, 685.)
[25] Cabrera, M.; Simoens, M.; Falchi, G.; Lavaggi, L.; Piroo, E.; Eduardo, E.-E. Bioorg. Med. Chem. 2007, 15, 3356.
[26] Mari, J.-C.; Xiong, Y.; Geanna, K. M.; Adam, R. Y.; Tohru T.; Nina, B.; Scott, E. S.; John, A. P. J. Org. Chem. 2010, 75, 4584.
[27] Su, S.; Acquilano, D. E.; Arumugasamy, J.; Beeler, A. B.; Eastwood, E. L.; Giguere, J. R.; Lan, P.; Lei, X.; Min, G. K.; Yeager, A. R.; Zhou, Y.; Panek, J. S.; Snyder, J. K.; Schaus, S. E.; Porco, J. A. Jr. Org. Lett. 2005, 7, 2751.
/
| 〈 |
|
〉 |