

一种咪唑并吩嗪内酰胺反应型识别氰离子的荧光探针
收稿日期: 2016-11-03
修回日期: 2016-12-29
网络出版日期: 2017-01-17
基金资助
国家自然科学基金(Nos.21662031,21661028,21574104,21262032)资助项目.
A Novel Imidazophenazine Lactam Reaction Type Recognition Cyanide Ion Fluorescence Probe
Received date: 2016-11-03
Revised date: 2016-12-29
Online published: 2017-01-17
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21662031, 21661028, 21574104, 21262032).
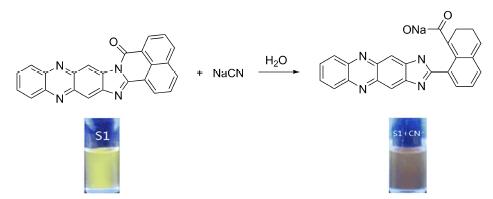
设计合成了一种新型咪唑并吩嗪内酰胺荧光传感器分子(S1),通过核磁共振氢谱、碳谱和高分辨质谱等手段对其结构进行了表征,并测定了S1在二甲基亚砜(DMSO)溶液中的荧光光谱,其最大荧光发射波长为524 nm. S1的DMSO溶液具有亮黄色荧光. 当在S1的DMSO溶液中分别加入F-,Cl-,Br-,I-,AcO-,H2PO4-,HSO4-,ClO4-,和SCN-等阴离子后,发现只有CN-的加入使S1的荧光光谱出现明显的下降并发生红移. 其溶液荧光颜色由黄色变为橘红色,说明S1对CN-具有良好的专一选择性. 抗干扰实验结果表明,这一识别过程不受其它阴离子干扰. 通过计算得到,S1对CN-的荧光光谱最低检测限为9.96×10-7 mol/L,这一数值低于世界卫生组织所规定的饮用水中氰离子含量. 机理研究表明,S1是一种反应型识别CN-的荧光传感器. 此外,将S1负载于固态硅胶之上,制备成固体CN-识别材料,并成功用于对固体NaCN的识别和对CN-水溶液的检测.
李翔 , 林奇 , 曲文娟 , 李乔 , 程晓斌 , 李文婷 , 张有明 , 姚虹 , 魏太保 . 一种咪唑并吩嗪内酰胺反应型识别氰离子的荧光探针[J]. 有机化学, 2017 , 37(4) : 889 -895 . DOI: 10.6023/cjoc201611008
A novel imidazophenazine lactam fluorescence chemosensor (S1) was designed, synthesized and characterized by 1H NMR, 13C NMR and HRMS techniques. Fluorescence spectra of S1 in dimethyl sulfoxide (DMSO) solution were measured. Its maximum emission wavelength was 524 nm. The DMSO solution of S1 has bright yellow fluorescence. Respectively, to the S1 solution add F-, Cl-, Br-, I-, AcO-, H2PO4-, HSO4-, ClO4-, and SCN-, the fluorescent color of the solution didn't change. Only the CN- addition, the fluorescent color of the S1 solution changed from yellow to orange-red, indicating that S1 has good specific selectivity for CN-. The results of anti-disturbance experiment demonstrated that S1 detect CN- without interference from other anions. By calculation, the linear of the fluorescence of the sensor for CN- is 9.96×10-7. This value is lower than the World Health Organization (WHO) provisions of the cyanide content of drinking water. Mechanism studies show that S1 is a fluorescence sensor by reactive recognition CN-. In addition, the application of sensor S1 supported on solid silica gel was used as a solid material to detecting solid NaCN and CN- in pure water.

Key words: phenazine derivative; fluorescence sensor; detecting anions
[1] Chen, J.-J.; Chen, W.; He, H.; Li, D.-B.; Li, W.-W.; Xiong, L.; Yu, H.-Q. Environ. Sci. Technol. 2013, 47, 1033.
[2] Ahuja, E. G.; Janning, P.; Mentel, M.; Graebsch, A.; Breinbauer, R.; Hiller, W.; Costisella, B.; Thomashow, L. S.; Mavrodi, D. V.; Blankenfeldt, W. J. Am. Chem. Soc. 2008, 130, 17053.
[3] Yang, L.; Li, X.; Qu, Y.; Qu, W.-S.; Zhang, X.; Hang, Y.-D.; Agren, H.; Hua, J.-L. Sen. Actuators, B 2014, 203, 833.
[4] Gu, P.-Y.; Zhao, Y.-B.; He, J.-H.; Zhang, J.; Wang, C.-Y.; Xu, Q.-F.; Lu, J.-M.; Sun, X.-W.; Zhang, Q.-C. J. Org. Chem. 2015, 80, 3030.
[5] Chen, W.; Su, J.-H,; Tian, H. Sci. China: Chem. 2016, 46, 325 (in Chinese).
(陈薇, 苏建华, 田禾, 中国科学: 化学, 2016, 46, 325.)
[6] Zhou, H.-T.; Sun, L.; Chen, W.; Tian, G.-J.; Chen, Y.; Li, Y.-R.; Su. J.-H. Tetrahedron 2016, 72, 2300.
[7] Metz, A. E.; Podlesny, E. E.; Carroll, P. J.; Klinghoffer, A. N.; Kozlowski, M. C. J. Am. Chem. Soc. 2014, 136, 10601.
[8] Aggarwal, K.; Khurana, J. M. J. Lumin. 2015, 167, 146.
[9] Wei, T.-B.; Wu, G.-Y.; Shi, B.-B.; Lin,-Q.; Yao, H.; Zhang, Y.-M. Chin. J. Chem. 2014, 32, 1238.
[10] Zhang, P.; Zhang, Y.-M.; Lin, Q.; Yao, H.; Wei, T.-B. Chin. J. Org. Chem. 2014, 34, 1300 (in Chinese).
(张鹏, 张有明, 林奇, 姚虹, 魏太保, 有机化学, 2014, 34, 1300.)
[11] Li, C.-W.; Yang, D.; Yin, B.; Guo, Y. Chin. J. Org. Chem. 2016, 36, 787 (in Chinese).
(李长伟, 杨栋, 尹兵, 郭媛, 有机化学, 2016, 36, 787.)
[12] Gao, G.-B.; Gong, D.-J.; Zhang, M.-X.; Sun, T.-L. Acta. Chim. Sinica. 2016, 74, 363 (in Chinese).
(高冠斌, 龚德君, 张明曦, 孙涛垒, 化学学报, 2016, 74, 363.)
[13] Zhang, Y.-P.; You, C.-X.; Yang, Y.-S.; Liu, X.-Y.; Guo, H.-C.; Dong, Y.-Y. Chin. J. Org. Chem. 2016, 36, 1401 (in Chinese).
(张应鹏, 尤彩霞, 杨云裳, 刘小育, 郭慧琛, 董玉莹, 有机化学, 2016, 36, 1401.)
[14] Su, N.; Yang, M.-P.; Meng, W.-P.; Yang, B.-Q. Chin. J. Org. Chem. 2015, 35, 175 (in Chinese).
(苏娜, 杨美盼, 孟文斐, 杨秉勤, 有机化学, 2015, 35, 175.)
[15] Yu, H.-B.; Li, H.-L.; Zhang, X.-F.; Xiao, Y.; Fang, P.-J.; Lü, C.-J.; Hou, W. Acta Chim. Sinica 2015, 73, 450 (in Chinese).
(于海波, 李红玲, 张新富, 肖义, 方沛菊, 吕春娇, 侯伟, 化学学报, 2015, 73, 450.)
[16] Zang, L.-B.; Wei, D.-Y.; Wang, S.-C.; Jiang, S.-M. Tetrahedron 2012, 68, 636.
[17] Gupta, A. S.; Garg, A.; Paul, K.; Luxami, V. J. Lumin. 2016, 173, 165.
[18] Jo, J. Y.; Olasz, A.; Chen, C. H.; Lee, D. J. Am. Chem. Soc. 2013, 135, 3620.
[19] Yan, L.-R.; Yang, M.-P.; Leng, X.; Zhang, M.; Long, Y.; Yang, B.-Q. Tetrahedron 2016, 72, 4361.
[20] Yoo, M.; Park, S.; Kim, H. J. Sen. Actuators, B 2015, 220, 788.
[21] Kim, D.; Na, S.; Y.; Kim, H. J. Sen. Actuators, B 2016, 226, 227.
[22] Wang, S. T.; Chir, J. L.; Jhong, Y.; Wu, A. T. J. Lumin. 2015, 167, 413.
[23] Yang, L.; Li, X.; Yang, J.-B.; Qu, Y.; Hua, J.-L. ACS Appl. Mater. Interfaces 2013, 5, 1317.
[24] Yang, L.; Zhang, X.; Qu, W.-S.; Hua, J.-L. Imaging Sci. Photochem. 2014, 32, 1 (in Chinese).
(杨林, 张潇, 瞿卫松, 花建丽, 影像科学与光化学, 2014, 32, 1.)
[25] Wang, F.; Wang, L.; Chen, X. Q.; Yoon, J. Y. Chem. Soc. Rev. 2014, 43, 12.
[26] Dagiliene, M.; Martynaitis, V.; Krisiucniene, V.; Krikstolaityte, S.; Sackus, A. ChemistryOpen 2015, 4, 363.
[27] Wang, K.-N.; Ma, L.; Liu, G.-Q.; Cao, D.-X.; Guan, R.-F.; Liu, Z.-Q. Dyes Pigm. 2016, 126, 104.
[28] Lee, K. S.; Kim, H. J.; Kim, G. H.; Shin, I.; Hong, J. I. Org. Lett. 2008, 10, 49.
[29] El-Shishtawy, R. M.; Al-Zahrani, F. A. M.; Al-amshany, Z. M.; Asiri, A. M. Sen. Actuators, B 2017, 240, 288.
[30] Meng, Q.-T. Ph.D. Dissertation, Dalian University of Technology, Dalian, 2011 (in Chinese).
(孟庆涛, 博士论文, 大连理工大学, 大连, 2011.)
[31] Shi, B.-B.; Zhang, P.; Wei, T.-B.; Yao, H.; Lin, Q.; Liu, J.; Zhang, Y.-M. Tetrahedron 2013, 69, 7981.
[32] Shi, B.-B.; Zhang, Y.-M.; Wei, T.-B.; Lin, Q.; Yao, H.; Zhang, P.; You, X.-M. Sens. Actuators, B 2014, 190, 555.
[33] Lin, Q.; Zhu, X.; Chen, P.; Fu, Y. P.; Zhang, Y. M.; Wei, T. B. Acta Chim. Sinica 2013, 71, 1516 (in Chinese).
(林奇, 朱鑫, 陈佩, 符永鹏, 张有明, 魏太保, 化学学报, 2013, 71, 1516.)
[34] Gao, G.-Y.; Qu, W.-J.; Shi, B.-B.; Lin, Q.; Yao, H.; Yang, W.-L.; Zhang, Y.-M.; Wei, T.-B. Spectrochim. Acta, Part A 2014, 121, 514.
[35] Gao, G.-Y.; Qu, W.-J.; Shi, B.-B.; Lin, Q.; Yao, H.; Zhang, Y.-M.; Chang, J.; Cai, Y.; Wei, T.-B. Sens. Actuators, B 2015, 213, 501.
[36] Hu, J.-Y.; Liu, R.; Zhu, X.-L.; Cai, X.; Zhu, H.-J. Chin. Chem. Lett. 2015, 26, 339.
[37] Wei, T.-B.; Li, W.-T.; Li, Q.; Su, J.-X.; Qu, W.-J. Lin, Q.; Yao, H.; Zhang, Y.-M. Tetrahedron Lett. 2016, 57, 2767.
[38] Wei, T.-B.; Li, W.-T.; Li, Q.; Qu, W.-J.; Li, H.; Yan, G.-T.; Lin, Q.; Yao, H.; Zhang, Y.-M. RSC Adv. 2016, 6, 43832.
[39] Li, W.-T.; Wu, G.-Y.; Qu, W.-J.; Li, Q.; Lou, J.-C.; Lin, Q.; Yao, H.; Zhang, Y.-M.; Wei, T.-B. Sens. Actuators, B 2017, 239, 671.
[40] Zhang, Y.-M.; Wang, Y.-L.; Lin, Q.; Wang, D.-D.; Wei, T.-B. Chin. J. Org. Chem. 2009, 29, 575 (in Chinese).
(张有明, 王雅琳, 林奇, 王丹丹, 魏太保, 有机化学, 2009, 29, 575.)
[41] Wei, T.-B.; Wang, J.; Luo, R.; Zhang, Y.-M. Chin. J. Org. Chem. 2007, 27, 1381 (in Chinese).
(魏太保, 王军, 罗榕, 张有明, 有机化学, 2007, 27, 1381.)
[42] Zhao, J.; Li, J.; Gao, J.; Kjelleberg, S. L. A.; Loo, S. C. J.; Zhang, Q. J. Heterocycl. Chem. 2015, 52, 1699.
[43] Sahoo, P. K.; Giri, C.; Haldar, T. S.; Puttreddy, R.; Rissanen, K.; Mal, P. Eur. J. Org. Chem. 2016, 1283.
[44] Liu, X, X.; Weinert, Z. J.; Sharafi, M.; Liao, C. Y.; Li, J. N.; Schneebeli, S. T. Angew. Chem., Int. Ed. 2015, 54, 12772.
[45] Miao, S. B.; Bangcuyo, C. G.; Smith, M. D.; Bunz, U. H. F. Angew Chem., Int. Ed. 2006, 118, 677.
[46] Wu, J.-S.; Rui, X.-H.; Long, G.-K.; Chen, W.-Q.; Yan, Q.-Y.; Zhang, Q.-C. Angew. Chem., Int. Ed. 2015, 54, 7354.
[47] Hu, J. W.; Zhang, G. H.; Shih, H. H.; Jiang, X. Q.; Suna, P. P.; Cheng, C. H. J. Organomet. Chem. 2008, 693, 2798.
[48] Lucas, M.-D.; Wang, J.-F.; Nick, A.; Li, H.; Liu, T-B.; Pang, Y. J. Phys. Chem. B 2016, 120, 766.
[49] Yang, J.-R.; Fang, H.-P.; Gao, Y. J. Phys. Chem. Lett. 2016, 7, 1788.
[50] Zhou, H.-T.; Sun, L.; Chen, W.; Tian, G.-J.; Chen, Y.; Li, Y.-R.; Su, J.-H. Tetrahedron 2016, 72, 2300.
[51] Zhou, H.-T.; Mei, J.; Chen, Y.-A.; Chen, C.-L.; Chen, W.; Zhang, Z.-Y.; Su, J.-H.; Chou, P.-T.; Tian, H. Small 2016, 12, 6542.
[52] Yang, Y.-T.; Yin, C.-X.; Huo, F.-J.; Chao, J.-B.; Zhang, Y.-B.; Cheng, F.-Q. Sens. Actuators, B 2014, 193, 220.
[53] Li, Q.; Zhang, J.-H.; Cai, Y.; Qu, W.-J.; Gao, G.-Y.; Lin, Q.; Yao, H.; Zhang, Y.-M.; Wei, T.-B. Tetrahedron 2015, 71, 857.
[54] Shive, M.-S.-C.; Tanuja, B.; Bhaskar, G. Tetrahedron Lett. 2008, 49, 6646.
[55] Celso, R. N.; Leandro, G. N.; Vanderlei, G. M. Anal. Chem. 2015, 87, 362.
[56] Ji, H. K.; Seong, Y. L.; Hye, M. A.; Cheal, K. Sens. Actuators, B 2017, 242, 25.
[57] Duan, Y.-L.; Zheng, Y.-S. Talanta 2013, 107, 332.
[58] Shi, B.-B. M.S. Thesis, Northwest Normal University, Lanzhou, 2014 (in Chinese).
(史兵兵, 硕士论文, 西北师范大学, 兰州, 2014.)
/
| 〈 |
|
〉 |