

含多氟烷基吡唑环的邻甲酰氨基苯甲酰胺类化合物的合成及杀虫活性
收稿日期: 2017-01-09
修回日期: 2017-04-08
网络出版日期: 2017-04-21
基金资助
“十二五”国家科技支撑计划(No.2011BAE06B01-20)资助项目.
Synthesis and Insecticidal Activity of Novel Anthranilic Diamides Containing Polyfluoroalkyl Pyrazole Moiety
Received date: 2017-01-09
Revised date: 2017-04-08
Online published: 2017-04-21
Supported by
Project supported by the National Key Technologies R&D Program of China during the 12th Five-Year Plan Period (No. 2011BAE06B01-20).
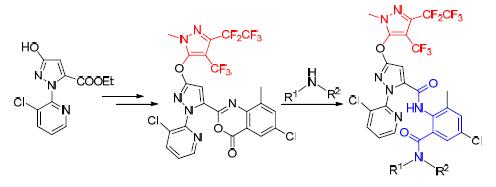
以2,3-二氯吡啶和水合肼为起始原料,经取代、缩合、氧化、醚化、成环和开环等多步反应合成一系列结构新颖的含多氟烷基吡唑环的邻甲酰氨基苯甲酰胺类化合物,结构经1H NMR和HRMS确证,并测试了目标化合物的杀虫活性.杀虫活性结果表明,在浓度0.8 mg/L下,大部分化合物对粘虫(Mythimna separata Walker)具有100%杀虫活性;在浓度2 mg/L下,N-(4-氯-2-(异丙基氨基甲酰)-6-甲基苯基)-1-(3-氯吡啶-2-基)-3-((1-甲基-3-五氟乙基-4-三氟甲基-1H-吡唑-5-基)氧基)-1H-吡唑-5-酰胺(8c)和N-(4-氯-2-(环丙基氨基甲酰)-6-甲基苯基)-1-(3-氯吡啶-2-基)-3-((1-甲基-3-五氟乙基-4-三氟甲基-1H-吡唑-5-基)氧基)-1H-吡唑-5-酰胺(8d)对斜纹夜蛾(Prodenia litura Fabricius)杀虫活性大于60%;在浓度0.08 mg/L下,N-(4-氯-2-(二甲基氨基甲酰)-6-甲基苯基)-1-(3-氯吡啶-2-基)-3-((1-甲基-3-五氟乙基-4-三氟甲基-1H-吡唑-5-基)氧基)-1H-吡唑-5-酰胺(8l)对小菜蛾(Plutella xylostella Linnaeus)杀虫活性达到82.5%.
史建俊 , 任贵华 , 吴宁捷 , 刘幸海 , 许天明 , 谭成侠 . 含多氟烷基吡唑环的邻甲酰氨基苯甲酰胺类化合物的合成及杀虫活性[J]. 有机化学, 2017 , 37(8) : 2131 -2138 . DOI: 10.6023/cjoc201701019
A series of novel anthranilic diamides containing polyfluoroalkyl pyrazole were synthesized using 2,3-dichloropyridine and hydrazine hydrate as starting materials via substitution, condensation, oxidation, etherification, cyclization and ring cleavage reaction. The title products were characterized by 1H NMR and HRMS. The bioassay results indicated that most of title compounds exhibited 100% insecticidal activitiy against Mythimna separata Walker at 0.8 mg/L, N-(4-chloro-2-(isopropylcarbamoyl)-6-methylphenyl)-1-(3-chloropyridin-2-yl)-3-((1-methyl-3-(perfluoroethyl)-4-(trifluoromethyl)-1H-pyrazol-5-yl)oxy)-1H-pyrazole-5-carboxamide (8c) and N-(4-chloro-2-(cyclopropylcarbamoyl)-6-methylphenyl)-1-(3-chloropyridin-2-yl)-3-((1-methyl-3-(perfluoroethyl)-4-(trifluoromethyl)-1H-pyrazol-5-yl)oxy)-1H-pyrazole-5-carboxamide (8d) showed more than 60% larvicidal activity against Prodenia litura Fabricius at 2 mg/L, and the mortality of N-(4-chloro-2-(dimethylcarbamoyl)-6-methylphenyl)-1-(3-chloropyridin-2-yl)-3-((1-methyl-3-(perfluoroethyl)-4-(trifluoromethyl)-1Hpyrazol-5-yl)oxy)-1H-pyrazole-5-carboxamide (8l) against Plutella xylostella Linnaeus reached 82.5% at 0.08 mg/L.

[1] Oerke, E. C. J. Agric. Sci. 2006, 144, 31.
[2] Wang, B. L.; Zhu, H. W.; Ma, Yi.; Xiong, L. X.; Li, Y. Q.; Zhao, Y.; Zhang, J. F.; Chen, Y. W.; Zhou, S.; Li, Z. M. J. Agric. Food Chem. 2013, 61, 5483.
[3] Cordova, D.; Benner, E. A.; Sacher, M. D.; Rauh, J. J.; Sopa, J. S.; Lahm, G. P.; Selby, T. P.; Stevenson, T. M.; Flexner, L.; Gutteridge, S.; Rhoades, D. F.; Wu, L.; Smith, R. M.; Tao, Y. Pestic. Biochem. Physiol. 2006, 84, 196.
[4] Clark, D, A.; Lahm, G. P.; Smith, B. K.; Barry, J. D.; Clagg, D. G. Bioorg. Med. Chem. 2008, 16, 3163.
[5] Wu, J.; Song, B. A.; Hu, D. Y.; Yue, M.; Yang, S. Pest Manage. Sci. 2012, 68, 801.
[6] Zhang, J. F.; Xu, J. Y.; Wang, B. L.; Li, Y. X.; Xiong, L. X.; Li, Y. Q.; Ma, Y.; Li, Z, M. J. Agric. Food Chem. 2012, 60, 7565.
[7] Chen, K.; Liu, Q.; Ni, P. J.; Zhu, H. J.; Lia, Y. F.; Wang, Q. Pest Manage. Sci. 2015; 71, 1503.
[8] Lahm, G. P.; Stevenson, T. M.; Selby, T. P.; Freudenberger, J. H.; Cordova, D.; Flexner, L.; Bellin, C. A.; Dubas, C. M.; Smith, B. K.; Hughes, K. A.; Hollingshaus, J. G.; Clark, C. E.; Benner, E. A. Bioorg. Med. Chem. Lett. 2007, 17, 6274.
[9] Hughes, K. A.; Lahm, G. P.; Selby, T. P.; Stevenson, T. M. WO 2004067528, 2004[Chem. Abstr. 2004, 141, 190786].
[10] Joseph, S. V.; Grettenberger, I.; Godfrey, L. Crop Prot. 2016, 87, 68.
[11] Weller, P.; Boner M.; Foerstel, H.; Becker, H.; Peikert, B.; Dreher, W. J. Agric. Food Chem. 2011, 59, 4365.
[12] Motoba, K.; Nishizawa, H.; Suzuki, T.; Hamaguchi, H.; Uchida, M.; Funayama, S. Pestic. Biochem. Physiol. 2000, 67, 73.
[13] Monicka, J. C.; James, C. J. Mol. Struct. 2014, 1075, 335.
[14] Quayle, W. C.; Oliver, D. P.; Zrna. S.; Fattore, A. J. Agric. Food Chem. 2007, 55, 5199.
[15] Chupp, J. P.; Hamper, B. C.; Wettach, R. H. WO 9602515, 1996[Chem. Abstr. 1996, 125, 10808].
[16] Wang, W. Q.; Zhang. P.; Meng, R. J.; Zhao, J. J.; Huang, Q. L.; Han, X. Y.; Ma, Z. Q.; Zhang, X. F. Crop Prot. 2014, 57, 48.
[17] Fulcher, J. M.; Wayment, D. G.; White Jr., P. M.; Webber Ⅲ, C. L.J. Agric. Food Chem. 2014, 62, 2141.
[18] Maue, M.; Decor, A.; Bretschneider, T.; Hahn, J. J.; Hallenbach, W.; Fischer, R.; Schwarz, H. G.; Gorgens, U.; Ilg, K.; Raming, K.; Kobberling, J.; Hubsch, W.; Turberg, A. WO 2015193218, 2015[Chem. Abstr. 2015, 164, 127051].
[19] Thielert, W.; Maue, M.; Pitta, L.; Weckwert, H. WO 2015132168, 2015[Chem. Abstr. 2015, 163, 408360].
[20] Maue, M.; Harschneck, T.; Fisher, R.; Bretschneider, T.; Haha, J. J.; Decor, A.; Hallenbach, W.; Schwarz, H. G.; Kobberling, J.; Hubbsch, W. WO 2016020441, 2016[Chem. Abstr. 2016, 164, 290366].
[21] Liu, X. H.; Zhao, W.; Shen, Z. H.; Xing, J. H.; Yuan, J.; Yang, G.; Xu, T. M.; Peng, W. L. Bioorg. Med. Chem. Lett. 2016, 26, 3626.
[22] Zhao, W.; Xing, J. H.; Xu, T. M.; Peng, W. L.; Liu, X. H. Front. Chem. Sci. Eng. 2017, 11, 291.
[23] Zhao, W.; Shen, Z. H.; Xu, T. M.; Peng, W. L.; Liu, X. H. Lett. Drug Des. Discovery 2017, 14, 323.
[24] Liu, X. H.; Zhao, W.; Shen, Z. H.; Xing, J. H.; Xu, T. M.; Peng, W. L. Eur. J. Med. Chem. 2017, 125, 881.
[25] Zhao, W.; Shen, Z. H.; Xu, T. M.; Peng, W. L.; Liu, X. H. J. Heterocycl. Chem. 2017, 54, 1751.
[26] Zhai, Z. W.; Wang, Q.; Shen, Z. H.; Tan, C. X.; Weng, J. Q.; Liu, X. H. Chin. J. Org. Chem. 2017, 37, 232(in Chinese). (翟志文, 汪乔, 沈钟华, 谭成侠, 翁建全, 刘幸海, 有机化学, 2017, 37, 232.)
[27] Tan, C. X.; Shi, Y. X.; Liu, X. H.; Li, B. J.; Zhao, W. G. Lett. Drug Des. Discovery 2012, 9, 431.
[28] Gong, Y. Y.; Huang, W.; Liu, X. H.; Weng, J. Q.; Tan, C. X. Chin. J. Org. Chem. 2013, 33, 2396(in Chinese). (贡云芸, 黄伟, 刘幸海, 翁建全, 谭成侠, 有机化学, 2013, 33, 2396.)
[29] Ni, Y.; Xu, T. M.; Zhong, L. K.; Kong, X. Y.; Shi, J. J.; Liu, X. H.; Kong, X. L.; Ji, W. J.; Tan, C. X. Chin. J. Org. Chem. 2015, 35, 2218(in Chinese). (倪芸, 许天明, 钟良坤, 孔晓燕, 史建俊, 刘幸海, 孔小林, 姬文娟, 谭成侠, 有机化学, 2015, 35, 2218.)
[30] Tan, C. X.; Schrader, K. K.; Mizuno, C. S.; Rimando, A. M. J. Agric. Food Chem. 2011, 59, 5977.
[31] Zhao, Y.; Xiong, L. X.; Xu, L. P.; Wang, H. X.; Xu, H.; Li, H. B.; Tong, J.; Li, Z. M. Res. Chem. Intermed. 2013, 39, 3071.
[32] Zhao, Y.; Xu, L. P.; Tong J; Li, Y. Q.; Xiong, L. X.; Li, F.; Peng L. N.; Li, Z. M. Mol. Diversity 2012, 16, 711.
[33] Wu. J.; Xie, D. D.; Shan, W. L.; Zhao, Y. H.; Zhang, W.; Song, B. A.; Yang, S.; Ma. J. Chem. Pap. 2015, 69, 993.
[34] Berger, R., A.; Flexner, J. L. WO 2003024222, 2003[Chem. Abstr. 2003, 138, 267201].
[35] Zhou, S.; Zhang, X. L.; Wei, W.; Liu, J. B.; Xiong, L. X.; Yang N.; Li, Z. M. Chin. J. Org. Chem. 2014, 34, 1424(in Chinese). (周莎, 张秀兰, 魏巍, 刘敬波, 熊丽霞, 杨娜, 李正名, 有机化学, 2014, 34, 1424.)
/
| 〈 |
|
〉 |