

海洋异壁放线菌WH1-2216-6产生的多环含特特拉姆酸大环内酰胺
收稿日期: 2017-03-28
修回日期: 2017-04-27
网络出版日期: 2017-05-10
基金资助
国家自然科学基金(Nos.81561148012,41376148)和国家自然科学基金-广东联合基金(No.U1501221)资助项目.
Polycyclic Tetramate Macrolactams from the Marine-Derived Actinoalloteichus cyanogriseus WH1-2216-6
Received date: 2017-03-28
Revised date: 2017-04-27
Online published: 2017-05-10
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 81561148012, 41376148) and the National Natural Science Foundation of China-Guangdong Fund Joint Project (No. U1501221).
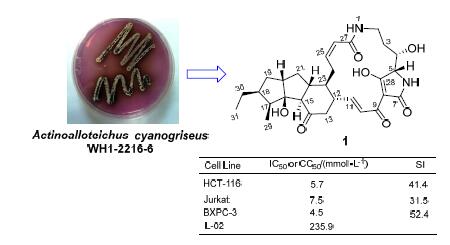
采用紫外跟踪分离和波谱鉴定的方法,从海洋异壁放线菌(Actinoalloteichus cyanogriseus WH1-2216-6)的发酵产物中分离鉴定了6个5,5,6-多环含特特拉姆酸大环内酰胺(PTMs)类天然产物:16-hydroxymaltophilin(1)、dihydromaltophilin(2)、4-deoxydihydromaltophilin(3)、maltophilin(4)、xanthobaccin C(5)和FI-2(6),其中1为新化合物.评价了化合物1~5对人正常肝细胞L-02及人癌细胞A549、MCF-7、Jurkat、BXPC-3、HCT-116、PANC-1和K562的细胞毒活性,结果表明:化合物1~5对上述人癌细胞具有细胞毒活性,其半数抑制浓度(IC50)为0.1~9.7 μmol·L-1;新化合物1对L-02的毒性较低,但对Jurkat、HCT-116和BXPC-3的选择指数(SI)分别高达31.5、41.1和52.4.除化合物2和3对A549和MCF-7的肿瘤细胞毒活性外,其余的肿瘤细胞毒活性是首次报道.还测试了化合物1~6的抗烟曲霉活性,发现化合物2和4的活性较好,其最小抑菌浓度(MIC)分别为3.04和6.12 μmol·L-1,这是首次发现5,5,6-PTMs类化合物具有抗烟曲霉活性.
关键词: 异壁放线菌; 5,5,6-多环含特特拉姆酸大环内酰胺; 抗肿瘤活性; 抗烟曲霉活性
梅显贵 , 王立平 , 王冬阳 , 范杰 , 朱伟明 . 海洋异壁放线菌WH1-2216-6产生的多环含特特拉姆酸大环内酰胺[J]. 有机化学, 2017 , 37(9) : 2352 -2360 . DOI: 10.6023/cjoc201703048
From the fermentation broth of the marine-derived Actinoalloteichus cyanogriseus WH1-2216-6, a new 5,5,6-polycyclic tetramate macrolactam (PTM) named 16-hydroxymaltophilin (1) was isolated and identified along with five known analouges, dihydromaltophilin (2), 4-deoxydihydromaltophilin (3), maltophilin (4), xanthobaccin C (5) and FI-2 (6), by means of UV-guided isolation as well as the spectroscopic identification. The cytotoxicities of compounds 1~5 were tested against the human normal hepatic cell line (L-02) and the seven human cancer cell lines, A549, MCF-7, Jurkat, BXPC-3, HCT-116, PANC-1 and K562. The results showed that compounds 1~5 were active against the above human cancer cell lines with the IC50 values of 0.1~9.7 μmol·L-1, among which the new compound 1 was the lowest toxic to L-02 cell and the most selective to Jurkat, HCT-116 and BXPC-3 cells with the selection index (SI) of 31.5, 41.4 and 52.4, respectively. The antifungal activities of 1~6 against Aspergillus fumigatus AF293 were also tested by two-fold dilution method. Compounds 2 and 4 were active against A. fumigatus AF293 with the minimum inhibitory concentration (MIC) values of 3.04 and 6.12 μmol·L-1, respectively. To the best of our knowledge, this is the first time to report the antifungal activity of 5,5,6-PTMs against A. fumigatus AF293. Apart from the cytotoxicity of compounds 2 and 3 against A549 and MCF-7 tumor cells lines, the other cytotoxicities were reported here for the first time, indicating the potential use of PTMs as the antitumor and antifungal lead compounds against A. fumigatus.

[1] Zhao, C.; Zhu, T.; Zhu, W. Chin. J. Org. Chem. 2013, 33, 1195(in Chinese). (赵成英, 朱统汉, 朱伟明, 有机化学, 2013, 33, 1195.)
[2] Blodgett, J. A.; Oh, D. C.; Cao, S.; Currie, C. R.; Kolter, R.; Clardy, J. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 11692.
[3] Zhang, G.; Zhang, W.; Saha, S.; Zhang, C. Curr. Top Med. Chem. 2016, 16, 1727.
[4] Jomon, K.; Kuroda, Y.; Ajisaka, M.; Sakai, H. J. Antibiot. 1972, 25, 271.
[5] Gunasekera, S. P.; Gunasekera, M.; McCarthy, P. J. Org. Chem. 1991, 56, 4830.
[6] Kanaxawa, S.; Fusetani, N.; Matsunaga, S. Tetrahedron Lett. 1993, 34, 1065.
[7] Jakobi, M.; Winkelmann, G.; Kaiser, D.; Kempter, C.; Jung, G.; Berg, G.; Bahl, H. J. Antibiot. 1996, 49, 1101.
[8] Graupner, P. R.; Hornburgh, S. T.; Mathiesonc, J. T.; Chapin, E. L.; Kemmitt, G. M.; Brown, J. M.; Snipes, C. E. J. Antibiot. 1997, 50, 1014.
[9] Yu, F.; Zaleta-Rivera, K.; Zhu, X.; Huffman, J.; Millet, J. C.; Harris, S. D.; Yuen, G.; Li, X. C.; Du, L. Antimicrob. Agents Chemother. 2007, 51, 64.
[10] Nakayama, T.; Homma, Y.; Hashidoko, Y.; Mizutani, J.; Tahara, S. Appl. Environ. Microbiol. 1999, 65, 4334.
[11] Li, Y.; Huffman, J.; Li, Y.; Du, L.; Shen, Y. Med. Chem. Commun. 2012, 3, 982.
[12] Xu, L.; Wu, P.; Wright, S. J.; Du, L.; Wei, X. Y. J. Nat. Prod. 2015, 78, 1841.
[13] Saha, S.; Zhang, W.; Zhang, G.; Zhu, Y. G.; Chen, Y. C.; Liu, W.;Yuan, C. S.; Zhang, Q. B.; Zhang, H. B.; Zhang, L. P.; Zhnag, W. M.; Zhang, C. S. Chem. Sci. 2017, 8, 1607.
[14] Li, S.; Du, L.; Yuen, G.; Harris, S. D. Mol. Biol. Cell 2006, 17, 1218.
[15] Li, S.; Calvo, A. M.; Yuen, G. Y.; Du, L.; Harris, S. D. J. Eukaryot. Microbiol. 2009, 56, 182.
[16] Li, S.; Jochum, C. C.; Yu, F.; Zaleta-Rivera, K.; Du, L.; Harris, S. D.; Yuen, G. Y. Phytopathology 2008, 98, 695.
[17] Qian, G.; Hu, B.; Jiang, Y.; Liu, F. Agric. Sci. China 2009, 8, 68.
[18] Lou, L.; Chen, H.; Cerny, R. L.; Li, Y.; Shen, Y.; Du, L. Biochemistry 2012, 51, 4.
[19] Lou, L.; Qian, G.; Xie, Y.; Hang, J.; Chen, H.; Zaleta-Rivera, K.; Li, Y.; Shen, Y.; Dussault, P. H.; Liu, F.; Du, L. J. Am. Chem. Soc. 2011, 133, 643.
[20] Fu, P.; Wang, S.; Hong, K.; Li, X.; Liu, P.; Wang, Y.; Zhu, W. J. Nat. Prod. 2011, 74, 1751.
[21] Jia, H.-J.; Wang, C.; Wang, Y.; Liu, P.-P.; Zhu, W.-M. Chin. J. Mar. Drug. 2014, 33, 9(in Chinese). (贾海健, 王聪, 王乂, 刘培培, 朱伟明, 中国海洋药物, 2014, 33, 9.)
[22] Lacret, R.; Oves-Costales, D.; Gómez, C.; Díaz, C.; de la Cruz M; Pérez-Victoria, I.; Vicente, F.; Genilloud, O.; Reyes, F. Mar. Drugs 2014, 13, 128.
/
| 〈 |
|
〉 |