

吡啶盐-氯胺前体的合成及在聚氨酯表面固载的抗菌应用
收稿日期: 2017-08-27
修回日期: 2017-11-28
网络出版日期: 2017-12-08
基金资助
中央高校基本科研业务费(Nos.DUT14RC(3)081,DUT17LK17)和教育部留学回国人员科研启动基金资助项目.
Synthesis and Immobilization of Pyridinium N-Chloramine Precursors on PU Film for Antibacterial Application
Received date: 2017-08-27
Revised date: 2017-11-28
Online published: 2017-12-08
Supported by
Project supported by the Fundamental Research Funds for the Central Universities (Nos. DUT14RC(3)081, DUT17LK17) and the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry.
李令东 , 迟晓芳 , 闫佳威 , 赵梓含 . 吡啶盐-氯胺前体的合成及在聚氨酯表面固载的抗菌应用[J]. 有机化学, 2018 , 38(4) : 955 -962 . DOI: 10.6023/cjoc201708056
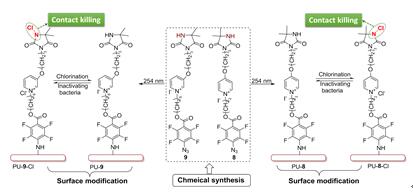
In this work, two novel pyridinium N-chloramine precursors were designed and synthesized, both of which contain perfluorophenyl azide (PFPA) unit as the photo-coupling handle. The synthetic precursors were photo immobilized on commercial PU films upon UV (254 nm) light irradiation. After exposure to diluted household bleach, the resulting PU films were rendered biocidal. Antibacterial tests showed that satisfactory was achieved for both surface modified films and that the film grafted with shorter alkyl linker-contained precursor demonstrated even higher biocidal efficacy. Non-leaching antibacterial PU materials were herein developed based on PFPA-coupling strategy, providing a simple and universal method to confer N-chloramines on inert polymer surface for antibacterial application.
[1] Kang, Z.; Jiao, Y.; Zhang, B.; Liang, J. J. Shanghai Normal Univ. (Nat. Sci. Ed.) 2012, 41, 540(in Chinese). (亢真真, 焦玉超, 张冰, 梁杰, 上海师范大学学报(自然科学版), 2012, 41, 540.)
[2] Hui, F.; Debiemme-Chouvy, C. Biomacromolecules 2013, 14, 585.
[3] Dong, A.; Wang, Y.-J.; Gao, Y.; Gao, T.; Gao G. Chem. Rev. 2017, 117, 4806.
[4] Gottardi, W.; Debabov, D.; Nagl, M. Antimicrob. Agents Chemother. 2013, 57, 1107.
[5] Jie, Z.; Yan, X.; Zhao, L.; Worley, S. D.; Liang. J. RSC Adv. 2014, 4, 6048.
[6] Li, L.; Pu, T.; Zhanel, G.; Zhao, N.; Ens, W.; Liu, S. Adv. Healthcare Mater. 2012, 1, 609.
[7] Ning, C.; Li, L.; Logsetty, S.; Ghanbar, S.; Guo, M.; Ensf, W.; Liu, S. RSC Adv. 2015, 5, 93877.
[8] Li, L.; Zhao, Y.; Zhou, H.; Ning, A.; Zhang, F.; Zhao, Z. Tetrahedron Lett. 2017, 58, 321.
[9] Li, L.; Zhou, H.; Gai, F.; Chi, X.; Zhang, F.; ZhaoZ. RSC Adv. 2017, 7, 13244.
[10] Liang, J.; Chen, Y.; Ren, X. Ind. Eng. Chem. Res. 2007, 46, 6425.
[11] Wu, L.; Liu A.; Li, Z. Fibers Polym. 2015, 16, 550.
[12] Sun, Y. Y.; Sun, G. J. Appl. Polym. Sci. 2001, 81, 1517.
[13] Liu, L.-H.; Yan, M. Acc. Chem. Res. 2010, 43, 1434.
[14] Li, L.; Li, J.; Kulkarni, A.; Liu, S. J. Mater. Chem., 2013, 1, 571.
[15] Williamson, M. R.; Black, R.; Kielty, C. Biomaterials 2006, 27, 3608.
[16] Li, L.; Chi, X.; Gai, F.; Zhou, H.; Zhang, F.; Zhao, Z. J. Appl. Polym. Sci. 2017, 134, 45323.
[17] Dastgir, S.; Coleman, K. S.; Green, M. L. H. Dalton Trans. 2011, 40, 661.
[18] Keana, J. F. W.; Cai, S. X. J. Org. Chem. 1990, 55, 3640.
[19] Xiong, J.; Xia, L.; Shentu, B.; Weng, Z. J. Appl. Polym. Sci. 2014, 131, 39812.
[20] Renaudie, L.; Narvor, C. L.; Lepleux, E.; Roger, P. Biomacromolecules 2007, 8, 679.
[21] Li, L.; Zhao, N.; Liu, S. Polymer 2012, 53, 67.
/
| 〈 |
|
〉 |