

构象锁定HIV-1融合抑制剂的合成及抗HIV-1活性研究
收稿日期: 2017-10-25
修回日期: 2018-01-08
网络出版日期: 2018-01-18
基金资助
国家自然科学基金(No.21602121)、内蒙古自然科学基金(No.2016BS0201)和包头医学院博士科学基金(No.BSJJ201620)资助项目.
Synthesis and Anti-HIV-1 Activity of Stapled HIV-1 Fusion Inhibitors
Received date: 2017-10-25
Revised date: 2018-01-08
Online published: 2018-01-18
Supported by
Project supported by the National Natural Science Foundation of China (No. 21602121), the Natural Science Foundation of Inner Mongolia (No. 2016BS0201) and the Doctoral Science Foundation of Baotou Medical College (No. BSJJ201620).
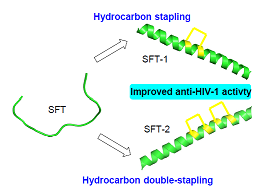
西夫韦肽(SFT)是一种有效抗HIV-1(Human Immunodeficiency Virus-1)融合抑制剂,它比已上市融合抑制药物T20拥有更高的药效及药代稳定性,目前已完成了Ⅱb临床实验.以西夫韦肽为模板,通过构象锁定策略将多肽中原有的盐桥替换为共价键合成了SFT订书肽SFT-1和SFT-2.产物结构经圆二光谱确认形成了α-螺旋.对所有合成多肽进行了抗HIV-1活性检测,结果表明SFT订书肽对7个HIV-1假病毒株抑制活性均高于SFT,其中SFT-1对B'亚型和B'C重组亚型病毒株抑制活性最高,SFT-2对B、CRF01_AE和CRF08_BC亚型病毒株抑制活性最高.
郭叶 , 傅莉莉 , 范晓文 , 史宣玲 . 构象锁定HIV-1融合抑制剂的合成及抗HIV-1活性研究[J]. 有机化学, 2018 , 38(5) : 1267 -1270 . DOI: 10.6023/cjoc201710033
Sifuvirtide (SFT) is a potent anti-HIV-1 (human immunodeficiency virus-1) fusion inhibitor and it shows higher potency and pharmacokinetic stability than the approved fusion inhibitor T20. At present, sifuvirtide has completed the phase Ⅱb clinical trial in China. In this study, SFT-1 and SFT-2 were synthesized via all-hydrocarbon cross-linking system with replacing the original salt bridge in SFT by hydrocarbon covalent bond, using sifuvirtide as template. The anti-HIV-1 activity was evaluated for all synthetic peptides. The results indicated that SFT stapled peptides displayed high inhibitory activity against seven HIV-1 pseudovirus strains, and SFT-1 showed the highest inhibitory activity against B' and B'C subtype virus strains, SFT-2 showed the highest inhibitory activity against B, CRF01_AE and CRF08_BC subtype virus strains.

[1] Dai, S. J.; Dou, G. F.; Qiang, X. H.; Song, H. F.; Tang, Z. M.; Liu, D. S.; Liu, X. W.; Yang, L. M.; Zheng, Y. T.; Liang, Q. Acta Pharmacol. Sin. 2005, 26, 1274.
[2] He, Y.; Xiao, Y.; Song, H.; Liang, Q.; Ju, D.; Chen, X.; Lu, H.; Jing, W.; Jiang, S.; Zhang, L. J. Biol. Chem. 2008, 283, 11126.
[3] Tan, J. J.; Ma, X. T.; Liu, C.; Zhang, X. Y.; Wang, C. X. Curr. Pharm. Des. 2013, 19, 1810.
[4] Tennyson, R. L.; Walker, S. N.; Ikeda, T.; Harris, R. S.; Kennan, A. J.; McNaughton, B. R. ChemBioChem 2016, 17, 1945.
[5] Zhang, D.; Li, W.; Jiang, S. Expert. Opin. Ther. Pat. 2015, 25, 159.
[6] Yu, F.; Lu, L.; Du, L. Y.; Zhu, X. J.; Debnath, A. K.; Jiang, S. B. Viruses 2013, 5, 127.
[7] Yao, X.; Chong, H.; Zhang, C.; Waltersperger, S.; Wang, M.; Cui, S.; He, Y. J. Biol. Chem. 2012, 287, 6788.
[8] Schafmeister, C. E.; Po, J.; Verdine, G. L. J. Am. Chem. Soc. 2000, 122, 5891.
[9] Wachter, F.; Morgan, A. M.; Godes, M.; Mourtada, R.; Bird, G. H.; Walensky, L. D. Oncogene 2017, 36, 2184.
[10] Bird, G. H.; Mazzola, E.; Opoku-Nsiah, K.; Lammert, M. A.; Godes, M.; Neuberg, D. S.; Walensky, L. D. Nat. Chem. Biol. 2016, 12, 845.
[11] Gao, S.; Guo, Y.; Li, H. Y.; Fang, G. M. Prog. Chem. 2014, 26, 100(in Chinese). (高帅, 郭叶, 李海燕, 方葛敏, 化学进展, 2014, 26, 100.)
[12] Hu, X.; He, Y.; Wu, L.; Hao, Y.; Wang, Z.; Zheng, W. Bioorg. Med. Chem. Lett. 2017, 27, 5446.
[13] Cui, H. K.; Qing, J.; Guo, Y.; Wang, Y. J.; Cui, L. J.; He, T. H.; Zhang, L. Q.; Liu, L. Bioorg. Med. Chem. 2013, 21, 3547.
[14] Cui, H. K.; Zhao, B.; Li, Y.; Guo, Y.; Hu, H.; Liu, L.; Chen, Y. G. Cell Res. 2013, 23, 581.
[15] Lau, Y. H.; de Andrade, P.; Wu, Y.; Spring, D. R. Chem. Soc. Rev. 2015, 44, 91.
[16] Keeling, K. L.; Cho, O.; Scanlon, D. B.; Booker, G. W.; Abell, A. D.; Wegener, K. L. Org. Biomol. Chem. 2016, 14, 9731
[17] Wang, D. Y.; Chen, K.; Kulp Ⅲ, J. L.; Arora, P. S. J. Am. Chem. Soc. 2006, 128, 9248.
[18] Fang, G. M.; Wang, J.-X.; Liu, L. Angew. Chem., Int. Ed. 2012, 51, 10347.
[19] Fang, G. M.; Li, Y. M.; Shen, F.; Huang, Y. C.; Li, J. B.; Lin, Y.; Cui, H. K.; Liu, L. Angew. Chem., Int. Ed. 2011, 50, 7645.
[20] Zheng, J. S.; Tang, S.; Qi, Y. K.; Wang, Z. P.; Liu, L. Nat. Protoc. 2013, 8, 2483.
/
| 〈 |
|
〉 |