

新型杂环查尔酮衍生物的合成及其杀虫活性研究
收稿日期: 2017-12-19
修回日期: 2018-01-31
网络出版日期: 2018-03-16
Synthesis and Study on Insecticidal Activity of New Heterocyclic Chalcone Derivatives
Received date: 2017-12-19
Revised date: 2018-01-31
Online published: 2018-03-16
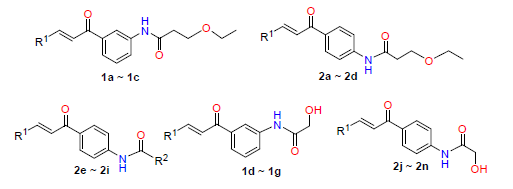
以含杂环的醛和氨基苯乙酮以及酰氯为原料,通过Claisen-Schmidt缩合反应和酰氯的氨解反应合成了21个新的杂环查尔酮衍生物.以蚕豆蚜(Aphis craccivora)和菜青虫(Pieris rapae)为靶标,对合成化合物的杀虫活性进行了实验室生测.结果显示,大部分化合物都对这两种害虫有一定的毒杀活性.其中,化合物2l对蚕豆蚜的半致死浓度(LC50)为1.6 μg/mL,毒杀效果优于吡虫啉(LC50为1.8 μg/mL),1b、1f、2j、2n对蚕豆蚜的毒杀效果也和吡虫啉接近;化合物1f和2m对菜青虫的LC50分别为6.6 μg/mL和8.5 μg/mL,毒杀效果比高效氯氰菊酯(LC50为9.2 μg/mL)更好.
严映坤 , 徐侨 , 高扬 , 刘辉 , 唐孝荣 . 新型杂环查尔酮衍生物的合成及其杀虫活性研究[J]. 有机化学, 2018 , 38(7) : 1763 -1771 . DOI: 10.6023/cjoc201712027
Twenty one new heterocyclic chalcone derivatives were synthesized by the Claisen-Schmidt condensation and ammonolysis of acyl chloride using heterocyclic aldehyde and amino substituted acetophenone as well as acyl chloride as raw materials. The laboratorial bioassay of the insecticidal activity of the synthesized compounds was performed using Aphis craccivora and Pieris rapae as targets. The results showed that most of them had significant insecticidal activity against the two pests. Among them, the median lethal concentration (LC50) of compound 2l was 1.6 μg/mL against Aphis craccivora, whose insecticidal efficacy was superior to imidacloprid with LC50 of 1.8 μg/mL. The insecticidal efficacy of 1b, 1f, 2j and 2n was also close to imidacloprid against Aphis craccivora. The LC50 values of compounds 1f and 2m against Aphis craccivora and Pieris rapae were 6.6 μg/mL and 8.5 μg/mL, respectively and their insecticidal activity exceeded β-cypermethrin, whose LC50 was 9.2 μg/mL.

[1] Ruan, L. L.; Fan, R. J.; Liu, X. H.; Chen, J.; Weng, J. Q. Chin. J. Org. Chem. 2015, 35, 1166 (in Chinese).
(阮铃莉, 范人杰, 刘幸海, 陈杰, 翁建全, 有机化学, 2015, 35, 1166.)
[2] Anuradha, V.; Pullela, V.; Srinivas, R.; Ranga, R. K.; Manjulatha, M. G.; Purohit, J.; Adhusudana, R. Bioorg. Med. Chem. 2006, 14, 6820.
[3] Formagio, A. S. N.; Tonin, L. T D.; Foglio, M. A.; Madjarof, C.; Carvalho. J. E. D.; Costa, W. F. D.; Cardoso, F. P.; Sarragiotto, M. H. Bioorg. Med. Chem. 2008, 16, 9660.
[4] Xe, F.; Liu, T. T.; Yang, G.; Yuan, J.; Kong, X. L.; Xu, T. M.; Tan, C. X. Chin. J. Org. Chem. 2013, 33, 2596 (in Chinese).
(谢峰, 刘婷婷, 杨果, 袁静, 孔小林, 许天明, 谭成侠, 有机化学, 2013, 33, 2596.)
[5] Babasaheb, P.; Bandgar, S. S.; Gawande, J. Bioorg. Med. Chem. 2010, 18, 2060.
[6] Rupak, K. K.; Sarma, N. R.; Jadab, C. S. J. Steroids 2013, 78, 321.
[7] Yin, B. T.; Yan, C. Y.; Peng, X. M.; Zhang, S. L.; Rasheed, S.; Geng, R. X.; Zhou, C. H. Eur. J. Med. Chem. 2014, 71, 148.
[8] Tavares, C.; Susana, J.; Alves, T. A.; Juliana C. G.; Elaine, S. F.; Cisalpino, P. S.; Adailton, J.; Giovanni, F.; Caramori, R. P.; Hugo, T. S.; Mara, E. F.; Braibante, M. G. J. Eur. J. Med. Chem. 2011, 46, 4448.
[9] Hsieh, C. T.; Hsieh, T. J.; Mohamed, E. S.; Chuang, D. W.; Tsai, Y. H.; Yen, C. T.; Wu, S. F.; Wu, Y. C.; Chang, F. R. Bioorg. Med. Chem. Lett. 2012, 22, 3912.
[10] Dai, H.; Liu, J. B.; Miao, W. K.; Wu, S. S.; Zhang, X.; Wang, T. T.; Fang, J. X. Chin. J. Org. Chem. 2011, 31, 1943 (in Chinese).
(戴红, 刘建兵, 苗文科, 吴珊珊, 张欣, 王婷婷, 方建新, 有机化学, 2011, 31, 1943.)
[11] Shang, E. C.; Liu, C. L.; Du, Y. J. Chem. Ind. Eng. Prog. 1996, 5, 11 (in Chinese).
(尚尔才, 刘长令, 杜英娟, 化工进展, 1996, 5, 11.)
[12] Yang, J. C.; Dai, R. H.; Liu, Y. P.; Wu, Q.; Liu, C. L. Chin. J. Pestic. 2011, 50, 625 (in Chinese).
(杨吉春, 戴荣华, 刘允萍, 吴娇, 刘长令, 农药, 2011, 50, 625.)
[13] Wang, Z. J.; Liu, B.; Li, Y. H.; Zhao, W. G. Chin. J. Org. Chem. 2011, 31, 317 (in Chinese).
(王振军, 刘斌, 李永红, 赵卫光, 有机化学, 2011, 31, 317.)
[14] Wang, H. S.; Zhao, W. X. Chin. J. Org. Chem. 2013, 33, 1822 (in Chinese).
(王宏社, 赵卫星, 有机化学, 2013, 33, 1822.)
[15] Sheng, G Z.; Zhang, W. Chin. J. Org. Chem. 2013, 33, 2271 (in Chinese).
(盛国柱, 张炜, 有机化学, 2013, 33, 2271.)
[16] Leroux P. J. J. Pestic. Sci. 1996, 47, 191.
[17] Smiley, R. W.; Wilkins, D. E. Klepper, E. L. Plant Dis. 1990, 74, 782.
[18] Kataria, H. R.; Verma, P. R.; Racow, G. J. Ann. Appl. Biol. 1993, 123:247.
[19] Nakagawa, Y.; Henrich, V. Bioorg. Med. Chem. Lett. 2009, 276, 6128.
[20] Xu, Z. H.; Li, J. K. Jiangsu Agric. Sci. 2015, 45, 5 (in Chinese).
(徐志红, 李俊凯, 江苏农业科学, 2015, 45, 5.)
[21] Wang, J. B.; Tan, F. R.; Wang, L. G. Acta Agric. Shanghai 2009, 25, 131(in Chinese).
(王金斌, 谭芙蓉, 王利刚, 上海农业学报. 2009, 25, 131.)
[22] Catto, A.; Motta, G.; Tajana, A.; Cazzulani, P.; Nardi, D.; Leonardi, A. J. Med. Chem. 1987, 30, 16.
[23] Karaman, I.; Gezegen, H.; Guerdere, M. B.; Dingil, A.; Ceylan, M. Chem. Biodiversity 2010, 2, 400.
[24] Zhang, Z. B. Toxicity Determination of Insecticides, Science Press, Beijing, 1988.
(张宗炳, 杀虫药剂的毒力测定, 科学出版社, 北京, 1988 (in Chinese.)
/
| 〈 |
|
〉 |