

钌催化烯烃复分解反应中的氢键效应
收稿日期: 2018-05-02
修回日期: 2018-06-12
网络出版日期: 2018-06-29
基金资助
国家自然科学基金(No.21662022)资助项目.
Hydrogen-Bonding Effects in Ruthenium-Catalyzed Olefin Metathesis
Received date: 2018-05-02
Revised date: 2018-06-12
Online published: 2018-06-29
Supported by
Project supported by the National Natural Science Foundation of China (No. 21662022).
颜廷斌 , 刘跃辉 , 沈悦海 . 钌催化烯烃复分解反应中的氢键效应[J]. 有机化学, 2018 , 38(10) : 2491 -2500 . DOI: 10.6023/cjoc201805001
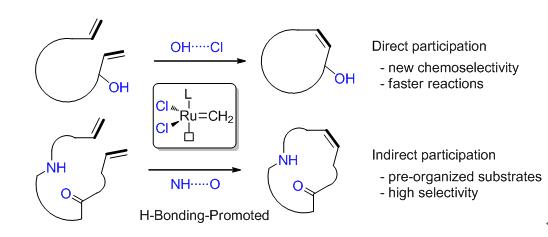
Olefin metathesis is among the most popular synthetic methods over the past two decades. Owing to the good functional group tolerance of ruthenium catalysts, hydrogen bonding has emerged as a powerful controlling element for olefin metathesis recently. Recent studies demonstrated that the hydrogen bonding between ruthenium catalyst and certain types of substrates or additives could significantly improve the rate, chemoselectivity and stereoselectivity of olefin metathesis, while the self-assembly of substrates could be driven by intramolecular or intermolecular hydrogen bonding and result in highly selective reactions. In this review, the development and application of hydrogen bonding-promoted ruthenium-catalyzed olefin metathesis reactions are summarized, and the mechanism insights into these systems are highlighted.
Key words: olefin metathesis; ruthenium-carbene complex; catalysis; hydrogen bonding
[1] Hoveyda, A. H.; Zhugralin, A. R. Nature 2007, 450, 243.
[2] Zielinski, G. K.; Grela, K. Chem.-Eur. J. 2016, 22, 9440.
[3] Kotha, S.; Dipak, M. K. Tetrahedron 2012, 68, 397.
[4] Fürstner, A. Chem. Commun. 2011, 47, 6505.
[5] Han, J. C.; Li, C. C. Synlett 2015, 26, 1289.
[6] Tomasek, J.; Schatz, J. Green Chem. 2013, 15, 2317.
[7] Levin, E.; Ivry, E.; Diesendruck, C. E.; Lemcoff, N. G. Chem. Rev. 2015, 115, 4607.
[8] Lin, Y. A.; Davis, B. G. Beilstein J. Org. Chem. 2010, 6, 1219.
[9] Aullon, G.; Bellamy, D.; Brammer, L.; Bruton, E. A.; Orpen, A. G. Chem. Commun. 1998, 653.
[10] Forman, G. S.; McConnell, A. E.; Tooze, R. P.; van Rensburg, W. J.; Meyer, W. H.; Kirk, M. M.; Dwyer, C. L.; Serfontein, D. W. Organometallics 2005, 24, 4528.
[11] Imahori, T.; Ojima, H.; Yoshimura, Y.; Takahata, H. Chem.-Eur. J. 2008, 14, 10762.
[12] Hoveyda, A. H.; Lombardi, P. J.; O'Brien, R. V.; Zhugralin, A. R. J. Am. Chem. Soc. 2009, 131, 8378.
[13] Feng, L.; Liu, Y. H.; Hou, B.; Yuan, Z. F.; Yu, F. C.; Yan, T. B.; Qin, Q.; Ji, R. G.; Li, Y. M.; Shen, Y. H.; Zuo, Z. L. Org. Biomol. Chem. 2016, 14, 10705.
[14] Hoye, T. R.; Zhao, H. Y. Org. Lett. 1999, 1, 1123.
[15] Schmidt, B.; Nave, S. Adv. Synth. Catal. 2007, 349, 215.
[16] Nicolaou, K. C.; Leung, G. Y. C.; Dethe, D. H.; Guduru, R.; Sun, Y. P.; Lim, C. S.; Chen, D. Y. K. J. Am. Chem. Soc. 2008, 130, 10019.
[17] Matsuya, Y.; Yamakawa, Y.; Tohda, C.; Teshigawara, K.; Yamada, M.; Nemoto, H. Org. Lett. 2009, 11, 3970.
[18] Sugimoto, K.; Yajima, H.; Hayashi, Y.; Minato, D.; Terasaki, S.; Tohda, C.; Matsuya, Y. Org. Lett. 2015, 17, 5910.
[19] Chowdhury, P. S.; Gupta, P.; Kumar, P. Tetrahedron Lett. 2009, 50, 7188.
[20] Trost, B. M.; Dong, G. B.; Vance, J. A. Chem.-Eur. J. 2010, 16, 6265.
[21] Srihari, P.; Kumaraswamy, B.; Shankar, P.; Ravishashidhar, V.; Yadav, J. S. Tetrahedron Lett. 2010, 51, 6174.
[22] Engelhardt, F. C.; Schmitt, M. J.; Taylor, R. E. Org. Lett. 2001, 3, 209.
[23] Lautens, M.; Maddess, M. L. Org. Lett. 2004, 6, 1883.
[24] Fuwa, H.; Yamaguchi, H.; Sasaki, M. Org. Lett. 2010, 12, 1848.
[25] Fuwa, H.; Saito, A.; Sasaki, M. Angew. Chem., Int. Ed. 2010, 49, 3041.
[26] Huang, Q. Y.; Herdewijn, P. J. Org. Chem. 2011, 76, 3742.
[27] Khan, R. K. M.; Torker, S.; Hoveyda, A. H. J. Am. Chem. Soc. 2013, 135, 10258.
[28] Koh, M. J.; Khan, R. K. M.; Torker, S.; Hoveyda, A. H. Angew. Chem., Int. Ed. 2014, 53, 1968.
[29] Lee, K. S.; Choi, T. L. Org. Lett. 2011, 13, 3908.
[30] Forman, G. S.; Tooze, R. P. J. Organomet. Chem. 2005, 690, 5863.
[31] Schmidt, B.; Staude, L. J. Org. Chem. 2009, 74, 9237.
[32] Schmidt, B.; Kunz, O.; Biernat, A. J. Org. Chem. 2010, 75, 2389.
[33] Schmidt, B.; Hauke, S. Org. Biomol. Chem. 2013, 11, 4194.
[34] Miller, S. J.; Grubbs, R. H. J. Am. Chem. Soc. 1995, 117, 5855.
[35] Saha, B.; Das, D.; Banerji, B.; Iqbal, J. Tetrahedron Lett. 2002, 43, 6467.
[36] Cromm, P. M.; Spiegel, J.; Grossmann, T. N. ACS Chem. Biol. 2015, 10, 1362.
[37] Cromm, P. M.; Schaubach, S.; Spiegel, J.; Fürstner, A.; Grossmann, T. N.; Waldmann, H. Nat. Commun. 2016, 7, 11300.
[38] Clark, T. D.; Ghadiri, M. R. J. Am. Chem. Soc. 1995, 117, 12364.
[39] Yang, X. W.; Gong, B. Angew. Chem., Int. Ed. 2005, 44, 1352.
[40] Zeng, J. S.; Wang, W.; Deng, P. C.; Feng, W.; Zhou, J. J.; Yang, Y. Y.; Yuan, L. H.; Yamamoto, K.; Gong, B. Org. Lett. 2011, 13, 3798.
[41] Kidd, T. J.; Leigh, D. A.; Wilson, A. J. J. Am. Chem. Soc. 1999, 121, 1599.
[42] Hannam, J. S.; Kidd, T. J.; Leigh, D. A.; Wilson, A. J. Org. Lett. 2003, 5, 1907.
[43] Wisner, J. A.; Beer, P. D.; Drew, M. G. B.; Sambrook, M. R. J. Am. Chem. Soc. 2002, 124, 12469.
[44] Kilbinger, A. F. M.; Cantrill, S. J.; Waltman, A. W.; Day, M. W.; Grubbs, R. H. Angew. Chem., Int. Ed. 2003, 42, 3281.
[45] Scherman, O. A.; Ligthart, G. B. W. L.; Ohkawa, H.; Sijbesma, R. P.; Meijer, E. W. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 11850.
[46] Liu, X. G.; Xu, J. F.; Wang, Z. Q.; Zhang, X. Polym. Chem. 2016, 7, 2333.
[47] Neal, J. A.; Mozhdehi, D.; Guan, Z. J. Am. Chem. Soc. 2015, 137, 4846.
[48] Trita, A. S.; Roisnel, T.; Mongin, F.; Chevallier, F. Org. Lett. 2013, 15, 3798.
/
| 〈 |
|
〉 |