

I型聚酮合酶中酰基转移酶结构域的研究进展
收稿日期: 2018-06-12
修回日期: 2018-06-29
网络出版日期: 2018-07-16
基金资助
国家自然科学基金(Nos.3173002,31520103901,31470212,31571284)资助项目.
Recent Advances in Acyltransferase Domain of Type I Polyktide Synthases
Received date: 2018-06-12
Revised date: 2018-06-29
Online published: 2018-07-16
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 3173002, 31520103901, 31470212, 31571284).
沈洁洁 , 毛旭明 , 陈新爱 , 李永泉 . I型聚酮合酶中酰基转移酶结构域的研究进展[J]. 有机化学, 2018 , 38(9) : 2377 -2385 . DOI: 10.6023/cjoc201806016
Most polyketide natural compounds, such as antibiotics, antineoplastics and immunosuppressants, are produced by type I polyketide synthases (PKSs). Type I PKSs are composed of several catalytic modules, each of which contains iterative domains, such as acyltransferase (AT) domain, for one round of polyketide chain elongation. The recent advances on AT domains of type I PKS modules and analyzes their categories, the diverse acyl subunits they transfer, their catalytic mechanisms and their protein structures are summarized. Moreover, the recent progress in AT engineering (AT domains swaps, AT site-directed mutagenesis and trans-AT complementation) for new polyketide derivatives is summarized, to show that the substrate specificity of AT domains is one of the key factors on determining the diversity of polyketide backbones. These works have laid a theoretical foundation for the further development of novel polyketides with multi-functions and in high-yields by AT domain engineering.
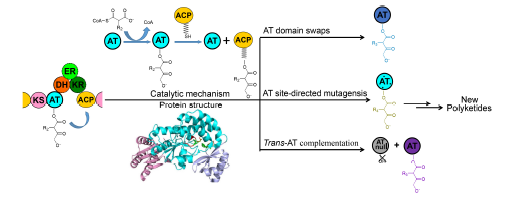
Key words: AT domain; substrate specificity; catalytic mechanism; type I PKS; polyketide
[1] Dunn, B. J.; Khosla, C. J. R. Soc., Interface 2013, 10, 20130297.
[2] Shen, B. Curr. Opin. Chem. Biol. 2003, 7, 285.
[3] Demydchuk, Y.; Sun, Y.; Hong, H.; Staunton, J.; Spencer, J. B.; Leadlay, P. F. ChemBioChem 2008, 9, 1136.
[4] Khosla, C.; Tang, Y.; Chen, A. Y.; Schnarr, N. A.; Cane, D. E. Annu. Rev. Biochem. 2007, 76, 195.
[5] Bailey, C. B.; Pasman, M. E.; Keatinge-Clay, A. T. Chem. Commun. (Camb) 2016, 52, 792.
[6] Li, S.; Lu, C.; Chang, X.; Shen, Y. Appl. Microbiol. Biotechnol. 2016, 100, 2641.
[7] Luhavaya, H.; Williams, S. R.; Hong, H.; Gonzaga de Oliveira, L.; Leadlay, P. F. ChemBioChem 2014, 15, 2081.
[8] Liang, Z. X. Nat. Prod. Rep. 2010, 27, 499.
[9] Lanen, S. G. V.; Shen, B. Curr. Top. Med. Chem. 2008, 8, 448.
[10] Fu, L. F.; Tao, Y.; Jin, M. Y.; Jiang, H. Biotechnol. Lett. 2016, 38, 2015.
[11] Taguchi, C.; Taura, F.; Tamada, T.; Shoyama, Y.; Tanaka, H.; Shoyama, Y.; Kuroki, R.; Morimoto, S. Acta Crystallogr., Sect. F 2008, 64, 217.
[12] Yu, D. Y.; Xu, F. C.; Zeng, J.; Zhan, J. X. IUBMB Life 2012, 64, 285.
[13] Fang, W. J.; Wang, C. J.; He, Y.; Zhou, Y. L.; Peng, X. D.; Liu, S. K. Acta Pharmacol. Sin. 2018, 39, 59.
[14] Fischbach, M. A.; Walsh, C. T. Chem. Rev. 2006, 106, 3468.
[15] Kotowska, M.; Pawlik, K.; Smulczyk-Krawczyszyn, A.; Bar-tosz-Bechowski, H.; Kuczek, K. Appl. Environ. Microbiol. 2009, 75, 887.
[16] Musiol, E. M.; Weber, T. Med. Chem. Commun. 2012, 3, 871.
[17] Ye, Z.; Musiol, E. M.; Weber, T.; Williams, G. J. Chem. Biol. 2014, 21, 636.
[18] Wong, F. T.; Jin, X.; Mathews, I. I.; Cane, D. E.; Khosla, C. Biochemistry 2011, 50, 6539.
[19] Cheng, Y. Q.; Tang, G. L.; Shen, B. Proc. Natl. Acad. Sci. U. S. A. 2003, 100, 3149.
[20] Pan, G. H.; Xu, Z. R.; Guo, Z. K.; Hindra; Ma, M.; Yang, D.; Zhou, H.; Gansemans, Y.; Zhu, X. C.; Huang, Y.; Zhao, L. X.; Jiang, Y.; Cheng, J. H.; Nieuwerburgh F. V.; Suh, J. W.; Duan, Y. W.; Shen, B. Proc. Natl. Acad. Sci. U. S. A. 2017, 114, E11131.
[21] Helfrich, E. J. N.; Piel, J. Nat. Prod. Rep. 2016, 33, 231.
[22] Calderone, C. T.; Iwig, D. F.; Dorrestein, P. C.; Kelleher, N. L.; Walsh, C. T. Chem. Biol. 2007, 14, 835.
[23] Butcher, R. A.; Schroeder, F. C.; Fischbach, M. A.; Straight, P. D.; Kolter, R.; Walsh, C. T.; Clardy, J. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 1506.
[24] Yadav, G.; Gokhale, R. S.; Mohanty, D. J. Mol. Biol. 2003, 328, 335.
[25] Jiang, C.; Qi, Z.; Kang, Q.; Liu, J.; Jiang, M.; Bai, L. Angew. Chem., Int. Ed. 2015, 54, 9097.
[26] Luzhetskyy, A.; Mayer, A.; Hoffmann, J.; Pelzer, S.; Holzenkamper, M.; Schmitt, B.; Wohlert, S. E.; Vente, A.; Bechthold, A. ChemBio-Chem 2007, 8, 599.
[27] Blauenburg, B.; Oja, T.; Klika, K. D.; Metsa-Ketela, M. ACS Chem. Biol. 2013, 8, 2377.
[28] Xu, Z.; Schenk, A.; Hertweck, C. J. Am. Chem. Soc. 2007, 129, 6022.
[29] Lowden, P. A.; Wilkinson, B.; Böhm, G. A.; Handa, S.; Floss, H. G.; Leadlay, P. F.; Staunton, J. Angew. Chem., Int. Ed. 2001, 40, 777.
[30] Paiva, N. L.; Roberts, M. F.; Demain, A. L. J. Ind. Microbiol. 1993, 12, 423.
[31] Mo, S.; Kim, D. H.; Lee, J. H.; Park, J. W.; Basnet, D. B.; Ban, Y. H.; Yoo, Y. J.; Chen, S. W.; Park, S. R.; Choi, E. A.; Kim, E.; Jin, Y. Y.; Lee, S. K.; Park, J. Y.; Liu, Y.; Lee, M. O.; Lee, K. S.; Kim, S. J.; Kim, D.; Park, B. C.; Lee, S. G.; Kwon, H. J.; Suh, J. W.; Moore, B. S.; Lim, S. K.; Yoon, Y. J. J. Am. Chem. Soc. 2011, 133, 976.
[32] Kato, Y.; Bai, L. Q.; Xue, Q.; Revill, W. P.; Yu, T. W.; Floss, H. G. J. Am. Chem. Soc. 2002, 124, 5268.
[33] Xia, M. L.; Huang, D.; Li, S. S.; Wen, J. P.; Jia, X. Q.; Chen Y. Biotechnol. Bioeng. 2013, 110, 2717.
[34] Goranovic, D.; Kosec, G.; Mrak, P.; Fujs, S.; Horvat, J.; Kuscer, E.; Kopitar, G.; Petkovic, H. J. Biol. Chem. 2010, 285, 14292.
[35] Dunn, B. J.; Cane, D. E.; Khosla, C. Biochemistry 2013, 52, 1839.
[36] Marsden, A. F.; Caffrey, P.; Aparicio, J. F.; Loughran, M. S.; Staunton, J.; Leadlay, P. F. Science 1994, 263, 378.
[37] Tsai, S. C.; Lu, H.; Cane, D. E.; Khosla, C.; Stroud, R. M. Bio-chemistry 2002, 41, 12598.
[38] Reeves, C. D.; Murli, S.; Ashley, G. W.; Piagentini, M.; Hutchinson, C. R.; McDaniel, R. Biochemistry 2001, 40, 15464.
[39] Wang, Y. Y.; Bai, L. F.; Ran, X. X.; Jiang, X. H.; Wu, H.; Zhang, W.; Jin, M. Y.; Li, Y. Q.; Jiang, H. Protein Pept. Lett. 2015, 22, 2.
[40] Jiang, H.; Wang, Y. Y.; Guo, Y. Y.; Shen, J. J.; Zhang, X. S.; Luo, H. D.; Ren, X. X.; Jiang, X. H.; Li, Y. Q. FEBS J. 2015, 282, 2527.
[41] Liew, C. W.; Nilsson, M.; Chen, M. W.; Sun, H. H.; Cornvik, T.; Liang, Z. X.; Lescar, J. J. Biol. Chem. 2012, 287, 23203.
[42] Tang, Y.; Kim, C. Y.; Mathews, I. I.; Cane, D. E.; Khosla, C. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 11124.
[43] Tang, Y.; Chen, A. Y.; Kim, C. Y.; Cane, D. E.; Khosla, C. Chem Biol. 2007, 14, 931.
[44] Li, Y.; Zhang, W.; Zhang, H.; Tian, W. Y.; Wu, L.; Wang, S. W.; Zheng, M. M.; Zhang, J. R.; Sun, C. H.; Deng, Z. X.; Sun, Y. H.; Qu, X. H.; Zhou, J. H. Angew. Chem., Int. Ed. 2018, 10.1002/anie.201802805.
[45] Caffrey, P.; Lynch, S.; Flood, E.; Finnan, S.; Oliynyk, M. Chem. Biol. 2001, 8, 713.
[46] Feng, J. F.; Zhou, R. C.; Guo, X. T.; Zhang, Y. Mod. Agric. Sci. Technol. 2011, 3, 24(in Chinese). (冯建飞, 周日成, 郭兴庭, 张扬, 现代农业科技, 2011, 3, 24.)
[47] Barajas, J. F.; Blake-Hedges, J. M.; Bailey, C. B.; Curran, S.; Keasling, J. D. Synth. Syst. Biotechnol. 2017, 2, 147.
[48] Oliynyk, M.; Brown, M. J.; Cortes, J.; Staunton, J.; Leadlay, P. F. Chem. Biol. 1996, 3, 833.
[49] Stassi, D. L.; Kakavas, S. J.; Reynolds, K. A.; Gunawardana, G.; Swanson, S.; Zeidner, D.; Jackson, M.; Liu, H.; Buko, A.; Katz, L. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 7305.
[50] Patel, K.; Piagentini, M.; Rascher, A.; Tian, Z. Q.; Buchanan, G. O.; Regentin, R.; Hu, Z.; Hutchinson, C. R.; McDaniel, R. Chem. Biol. 2004, 11, 1625.
[51] Wong, F. T.; Jin, X.; Mathews, I. I.; Cane, D. E.; Khosla, C. Biochemistry 2011, 50, 6539.
[52] Mcdaniel, R.; Thamchaipenet, A.; Gustafsson, C.; Fu, H.; Betlach, M.; Betlach, M.; Ashley, G. Proc. Natl. Acad. Sci. U. S. A. 1999, 96, 1846.
[53] Yuzawa, S.; Deng, K.; Wang, G.; Baidoo, E. E.; Northen, T. R.; Adams, P. D.; Katz, L.; Keasling, J. D. ACS Synth. Biol. 2017, 6, 139.
[54] Hans, M.; Hornung, A.; Dziarnowski, A.; Cane, D. E.; Khosla, C. J. Am. Chem. Soc. 2003, 125, 5366.
[55] Koryakina, I.; Kasey, C.; McArthur, J. B.; Lowell, A. N.; Chemler, J. A.; Hansen, D. A.; Sherman, D. H.; Williams, G. ACS Chem. Biol. 2017, 12, 114.
[56] Petkovic, H.; Sandmann, A.; Challis, I. R.; Hecht, H. J.; Silakowski, B.; Low, L.; Beeston, N.; Kuscer, E.; Gar-cia-Bernardo, J.; Leadlay, P. F.; Kendrew, S. G.; Wilkinson, B.; Müller R. Org. Biomol. Chem. 2008, 6, 500.
[57] Vecchio, F. D.; Petkovic, H.; Kendrew, S. G.; Low, L.; Wilkinson, B.; Lill, R.; Cortés, J.; Rudd, B. A. M.; Staunton, J.; Leadlay, P. F. J. Ind. Microbiol. Biotechnol. 2003, 30, 489.
[58] Ruan, X. A.; Pereda, A.; Stassi, D.; Zeidner, D.; Summers, R. G.; Jackson, M.; Shivakumar, A.; Kakavas, S.; Staver, M. J.; Donadio, S.; Katz, L. J. Bacteriol. 1997, 179, 6416.
[59] Sundermann U.; Bravo-Rodriguez, K.; Klopries, S.; Kushnir, S.; Gomez, H.; Sanchez-Garcia, E.; Schulz, F. ACS. Chem. Biol. 2013, 8, 443.
[60] Klopries, S.; Sundermann U.; Schulz, F. Beilstein J. Org. Chem. 2013, 9, 664.
[61] Koryakina, I.; McArthur, J.; Randall, S.; Draelos, M. M.; Musiol, E. M.; Muddiman, D. C.; Weber, T.; Williams, G. J. ACS Chem. Biol. 2013, 8, 200.
[62] Dunn, B. J.; Watts, K. R.; Robbins. T.; Cane, D. E.; Khosla, C. Biochemistry 2014, 53, 379.
[63] Ad, O.; Thuronyi, B. W.; Chang, M. C. Proc. Natl. Acad. Sci. U. S. A. 2017, 114, 660.
[64] Wesener, S. R.; Potharla, V. Y.; Cheng, Y. Q. Appl. Environ. Microbiol. 2011, 77, 1501.
[65] Lopanik, N. B.; Shields, J. A.; Buchholz, T. J.; Rath, C. M.; Hothersall, J.; Haygood, M. G.; Hakansson, K.; Thomas, C. M.; Sherman, D. H. Chem. Biol. 2008, 15, 1175.
/
| 〈 |
|
〉 |