

双齿N基配体锇配合物的合成及其在催化氨硼烷释氢中的应用
收稿日期: 2018-05-28
修回日期: 2018-07-10
网络出版日期: 2018-08-14
基金资助
国家自然科学基金(Nos.21501048,21503070,21571052,21771057)及河南省重点科技攻关(No.182102210377)资助项目.
Synthesis of Osmium Complexes with Bidentate Nitrogen-Based Ligands and Their Application in Catalytic Dehydrogenation of Ammonia Borane
Received date: 2018-05-28
Revised date: 2018-07-10
Online published: 2018-08-14
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21501048, 21503070, 21571052, 21771057) and the Key Science and Technology Project of Henan Province (No.182102210377).
赵茜怡 , 梁媛 , 徐霆 , 窦婷 , 张絜 , 陈学年 . 双齿N基配体锇配合物的合成及其在催化氨硼烷释氢中的应用[J]. 有机化学, 2018 , 38(12) : 3286 -3295 . DOI: 10.6023/cjoc201805050
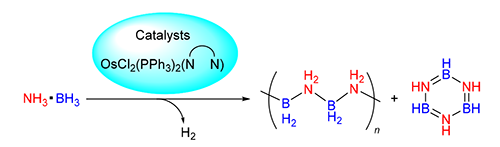
Reactions of OsCl2(PPh3)3 (1) with bidentate nitrogen-based ligands at room temperature led to the formation of complexes OsCl2(PPh3)2(diamine) (2~6) and OsCl2(PPh3)2(Phen*) (7~11). Complexes 2~11 showed high activity in the catalytic dehydrogenation of ammonia borane at 60℃, in THF/DME (V:V=1:1.6) solution with the catalyst loading of 5 mol%. Among all the catalysts, compound 4 demonstrated the highest catalytic activity, which represents the most efficient osmium catalyst in catalytic dehydrogenation of ammonia borane until now.
Key words: osmium; complex; synthesis; catalytic dehydrogenation; ammonia borane
[1] Rand, D. A. J.; Dell, R. M. Hydrogen Energy:Challenges and Prospects, Royal Society of Chemistry, Cambridge, UK, 2008.
[2] (a) Yadav, M.; Xu, Q. Energy Environ. Sci. 2012, 5, 9698.
(b) Dalebrook, A. F.; Gan, W.; Grasemann, M.; Moret, S.; Lau-renczy, G. Chem. Commun. 2013, 49, 8735.
[3] (a) Staubitz, A.; Robertson, A. P. M.; Manners, I. Chem. Rev. 2010, 110, 4079.
(b) Zhang, X.; Kam, L.; Trerise, R.; Williams, T. J. Acc. Chem. Res. 2017, 50, 86.
[4] (a) Tang, Z.; Chen, X.; Chen, H.; Wu, L.; Yu, X. Angew. Chem., Int. Ed. 2013, 52, 5832.
(b) Tang, Z.; Chen, H.; Chen, X.; Wu, L.; Yu, X. J. Am. Chem. Soc. 2012, 134, 5464.
[5] Wang, K.; Zhang, J.-G.; Man, T.-T.; Wu, M.; Chen, C.-C. Chem.-Asian. J. 2013, 8, 1076.
[6] (a) Appelt, C.; Chris Slootweg, J.; Lammertsma, K.; Uhl, W. Angew. Chem., Int. Ed. 2013, 52, 4256.
(b) Kalidindi, S. B.; Joseph, J.; Jagirdar, B. R. Energ. Environ. Sci. 2009, 2, 1274.
[7] (a) Alcaraz, G.; Sabo-Etienne, S. Angew. Chem., Int. Ed. 2010, 49, 7170.
(b) Staubitz, A.; Robertson, A. P. M.; Sloan, M. E.; Manners, I. Chem. Rev. 2010, 110, 4023.
(c) Rossin, A.; Peruzzini, M. Chem. Rev. 2016, 116, 8848.
[8] (a) Esteruelas, M. A.; López, A. M.; Mora, M. ACS Catal. 2015, 5, 187;
(b) Esteruelas, M. A.; Fernández, I.; López, A. M. Organo-metallics 2014, 33, 1104.
[9] (a) Kolb, H. C.; VanNieuwenhze, M. S.; Sharpless, K. B. Chem. Rev. 1994, 94, 2483.
(b) Döbler, C.; Mehltretter, G. M.; Sundermeier, U.; Beller, M. J. Am. Chem. Soc. 2000, 122, 10289.
(c) Döbler, C.; Mehltretter, G. M.; Sundermeier, U.; Beller, M. J. Organomet. Chem. 2001, 621, 70.
(d) Heravi, M. M.; Zadsirjan, V.; Esfandyari, M.; Lashaki, T. B. Tetrahedron:Asymmetry 2017, 28, 987.
[10] (a) Esteruelas, M. A.; Honczek, N.; Oliván, M.; Onate, E.; Valencia, M. Organometallics 2011, 30, 2468.
(b) Bertoli, M.; Choualeb, A.; Lough, A. J.; Moore, B.; Spasyuk, D.; Gusev, D. G. Organometallics 2011, 30, 3479.
(c) Buil, M. L.; Esteruelas, M. A.; Herrero, J.; Izquierdo, S.; Pastor, I. M.; Yus, M. ACS Catal. 2013, 3, 2072.
(d) Chelucci, G.; Baldino, S.; Baratta, W. Acc. Chem. Res. 2015, 48, 363.
(e) Bolaño, T.; Esteruelas, M. A.; Gay, M. P.; Oñate, E.; Pastor, I. M.; Yus, M. Organometallics 2015, 34, 3902.
(f) Barbato, C.; Baldino, S.; Ballico, M.; Figliolia, M.; Magnolia, S.; Siega, K.; Herdtweck, E.; Strazzolini, P.; Chelucci, G.; Baratta, W. Organometallics 2018, 37, 65..
[11] Spasyuk, D.; Vicent, C.; Gusev, D. G. J. Am. Chem. Soc. 2015, 137, 3743.
[12] Buil, M. L.; Esteruelas, M. A.; Gay, M. P. Organometallics 2018, 37, 603.
[13] (a) Baratta, W.; Bossi, G.; Putignano, E.; Rigo, P. Chem.-Eur. J. 2011, 17, 3474.
(b) Chelucci, G.; Baldino, S.; Baratta, W. Coord. Chem. Rev. 2015, 300, 29.
[14] Baker, R. T.; Gordon, J. C.; Hamilton, C. W. J. Am. Chem. Soc. 2012, 134, 5598.
[15] When the article was prepared, a similar synthetic method for complex 2 was reported by Baratta. Please see Ref.[10f] for details.
[16] Nascimento, R. D.; Silva, A. K.; Lião, L. M. J. Mol. Struct. 2018, 1151, 277.
[17] Hoffman, P. R.; Caulton, K. G. J. Am. Chem. Soc. 1975, 97, 4221.
[18] (a) Lay, P. A.; Sargeson, A. M.; Skelton, B, W. J. Am. Chem. Soc. 1982, 104, 6161.
(b) Clapham, S. E.; Morris, R. H. Organometallics 2005, 24, 479.
(c) McQueen, J. S.; Nagao, N.; Eberspacher, T. Inorg. Chem. 2003, 42, 3815.
(d) Ettner, N.; Hillen, W.; Ellestad, G. A. J. Am. Chem. Soc. 1993, 115, 2546.
(e) Peacock, A. F. A.; Habtemariam, A.; Moggach, S. A. Inorg. Chem. 2007, 46, 4049.
(f) Gong, L.; Lin, Y.; Wen, T. B. Organometallics 2009, 28, 1101.
(g) Martínez-Peña, F.; Pizarro, A. M. Chem.-Eur. J. 2017, 23, 16231.
[19] Luman, C. R.; Castellano, F. N. In Comprehensive Coordination Chemistry Ⅱ, 2nd ed., Vol. 1, Eds.:Meyer, T. J.; McCleverty, J. A., Elsevier Ltd., Pergamon, 2003, p. 25.
[20] (a) Akerboom, S.; van den Elshout, J. J. M. H.; Mutikainen, I. Eur. J. Inorg. Chem. 2013, 2013, 6137.
(b) Nakagawa, A.; Ito, A.; Sakuda, E. Eur. J. Inorg. Chem. 2017, 3794.
(c) Glazer, E. C.; Magde, D.; Tor, Y. J. Am. Chem. Soc. 2007, 129, 8544.
[21] Sjögren, M. P. T.; Frisell, H. B. Organometallics 1997, 16, 942.
[22] (a) Zheng, A.-X.; Si, J.; Tang, X.-Y.; Miao, L.-L.; Yu, M.; Hou, K.-P.; Wang, F.; Li, H.-X.; Lang, J.-P. Inorg. Chem. 2012, 51, 10262.
(b) Zheng, A.-X.; Wang, H.-F.; Lü, C.-N.; Ren, Z.-G.; Li, H.-X.; Lang, J.-P. Dalton Trans. 2012, 41, 558.
(c) Li, F.-L.; Yang, S.-P.; Zhang, W.-H.; Liu, Q.; Yu, H.; Chen, J.-X.; Lang, J.-P. ChemistrySelect 2016, 1, 2979.
[23] (a) Liu, B.; Zhao, Q.; Wang, H. Chin. J. Chem. 2012, 30, 2158.
(b) Nakamura, A.; Sato, T.; Kuroda, R. Chem. Commun. 2004, 2858.
(c) Carlson, B.; Phelan, G. D.; Kaminsky, W. J. Am. Chem. Soc. 2002, 124, 14162.
(d) Cheng, Y. K.; Cheung, J.; Che, K.-K.; Chi, M. Chem. Commun. 1997, 623.
(e) Carlson, B.; Phelan, G. D.; Benedict, J. B. Inorg. Chim. Acta 2006, 359, 1093.
[24] Bhattacharya, P.; Krause, J. A.; Guan, H. J. Am. Chem. Soc. 2014, 136, 11153.
[25] Duman, S.; Özkar, S. Int. J. Hydrogen Energy 2013, 38, 180.
[26] (a) Rossin, A.; Rossi, A.; Peruzzini, M. ChemPlusChem 2014, 79, 1316.
(b) Metters, O. J.; Chapman, A. M.; Robertson, A. P. M.; Woodall, C. H.; Gates, P. J.; Wass, D. F.; Manners, I. Chem. Commun. 2014, 50, 12146.
(c) Robertson, A. P. M.; Leitao, E. M.; Jurca, T.; Haddow, M. F.; Helten, H.; Lloyd-Jones, G. C.; Manners, I. J. Am. Chem. Soc. 2013, 135, 12670.
(d) Pons, V.; Baker, R. T. Angew. Chem., Int. Ed. 2008, 47, 9600.
(e) Staubitz, A.; Presa Soto, A.; Manners, I. Angew. Chem., Int. Ed. 2008, 47, 6212.
[27] (a) Kalviri, H. A.; Gärtner, F.; Ye, G. Chem. Sci. 2015, 6, 618.
(b) Shaw, W. J.; Linehan, J. C.; Szymczak, N. K. Angew. Chem., Int. Ed. 2008, 47, 7493.
[28] Sayalero, S.; Pericas, M. A. Synlett 2006, 2585.
/
| 〈 |
|
〉 |