

稠合四环噻嗪烷-4-酮衍生物的合成及其抗肿瘤活性
收稿日期: 2018-07-24
修回日期: 2018-08-23
网络出版日期: 2018-10-26
基金资助
国家自然科学基金(No.21372060)和河北省自然科学基金重点(No.B2016201031)资助项目.
Synthesis of the Fused Tetracyclic Thiazinan-4-one Derivatives and Their Anti-tumor Activitiy
Received date: 2018-07-24
Revised date: 2018-08-23
Online published: 2018-10-26
Supported by
Project supported by the National Natural Science Foundation of China (No. 21372060) and the Natural Science Fundation of Hebei Province (No. B2016201031).
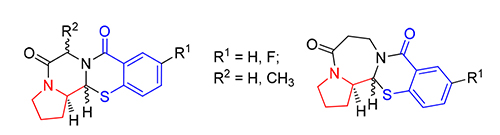
以叔丁氧羰基(Boc)保护的脯氨醛1、氨基酸酯盐酸盐2a~2d和巯基水杨酸3a~3b为原料,三组分一锅法得到苯并噻嗪烷-4-酮中间体.酸性条件下脱除Boc,分子内酰胺缩合制备稠合四环噻嗪烷-4-酮衍生物6~11.新生成手性碳(1-C)的构型通过H-1和H-2的偶合常数及X-ray单晶衍射确定.测试了化合物抗Hela和A549的肿瘤细胞增殖活性.结果表明,部分化合物具有中等的抗Hela细胞活性,其中(13aR,13bR)-1,2,3,13b-四氢苯并[e]吡咯并[2',1':3,4]吡嗪并[2,1-b][1,3]噻嗪-5,8(6H,13aH)-二酮(6b)的IC50值为9.50 μmol/L.所有化合物对A549的细胞没有抑制活性.
牛丽萍 , 邢顺凯 , 李小六 , 陈华 . 稠合四环噻嗪烷-4-酮衍生物的合成及其抗肿瘤活性[J]. 有机化学, 2019 , 39(3) : 771 -777 . DOI: 10.6023/cjoc201807043
The benzothiazin-4-one intermediates were prepared by the one-pot three-components condensation from the N-Boc-L-prolinal 1, amino acid ethyl/methyl ester hydrochlorides 2a~2d, and mercaptobenzoic acids 3a~3b. After removal of Boc, the target novel fused tetracyclic thiazinan-4-one derivatives 6~11 were afforded by the intramolecular cyclo-amidation reaction. The absolute configurations of the newly generated chiral carbon (1-C) were determined by the coupling constants of H-1 and H-2 and the X-ray crystallographic structures. The tetracyclic alkaloids were examined for their anti-proliferative activity against Hela and A549 tumor cells. The results showed that some compounds could moderately inhibit the growth of Hela cells, and among them, (13aR,13bR) -1,2,3,13b-tetrahydrobenzo[e]pyrrolo[2',1':3,4]pyrazino[2,1-b] [1,3]thiazine-5,8(6H,13aH) -dione (6b) was the best one with the IC50 value of 9.50 μmol/L. However, all the compounds showed no anti-tumor activity against A549.

[1] Kim, J.; Movassaghi, M. Acc. Chem. Res. 2015, 48, 1159.
[2] Amit Kumar Chattopadhyay, A. K.; Hanessian, S. Chem. Rev. 2017, 117, 4104.
[3] Chrzanowska, M.; Grajewska, A.; Rozwadowska, M. D. Chem. Rev. 2016, 116, 12369.
[4] Zi, W. W.; Zuo, Z. W.; Ma, D. W. Acc. Chem. Res. 2015, 48, 702.
[5] Tripathi, A. C.; Gupta, S. J.; Fatima, G. N.; Sonar, P. K.; Verma, A.; Saraf, S. K. Eur. J. Med. Chem. 2014, 72, 52.
[6] Zarghi, A.; Zebardast, T.; Daraie, B.; Hedayati, M. Bioorg. Med. Chem. 2009, 17, 5369.
[7] Geng, J. H.; Li, Y. X.; Zhang, W. J.; Su, X.; Guo, C. J. Shenyang Pharm. Univ. 2012, 29, 834(in Chinese). (耿红健, 高宁, 李裕鑫, 张卫军, 苏昕, 郭春, 沈阳药科大学学报, 2012, 29, 834.)
[8] Tiwari, R.; Moraski, G. C.; Krchňak, V.; Miller, P. A.; Colon-Martinez, M.; Herrero, E.; Oliver, A. G.; Miller, M. J. J. Am. Chem. Soc. 2013, 135, 3539.
[9] Cao, X.; Wang, S. B.; Deng, X. Q.; Liu, D. C.; Quan, Z. S. Med. Chem. Res. 2014, 23, 1829.
[10] Feng, J. N.; Li, X. H.; Shao, J.; Zhu, M.; Li, Y.; Chen, H.; Li, X. L. Chin. J. Org. Chem. 2015, 35, 1370(in Chinese). (冯俊娜, 李晓慧, 邵洁, 朱墨, 李妍, 陈华, 李小六, 有机化学, 2015, 35, 1370.)
[11] Stössel, A.; Schlenk, M.; Hinz, S.; Küppers, P.; Heer, J.; Gütschow, M.; Müller, C. E. J. Med. Chem. 2013, 56, 4580.
[12] Bates, R. W. Tetrahedron Lett. 2018, 59, 559.
[13] Xiong, Y. X.; Huang, Z. S.; Tan, J. H. Eur. J. Med. Chem. 2015, 97, 538.
[14] Manjal, S. K.; Kaur, R.; Bhatia, R.; Kumar, K.; Singh, V.; Shankar, R.; Kaur, R.; Rawal, R. K. Bioorg. Chem. 2017, 75, 406.
[15] Shao, J.; Zhu, M.; Gao, L. G.; Chen, H.; Li, X. L. Carbohydr. Res. 2018, 456, 45.
[16] Yin, Z. Q.; Zhu, M.; Wei, S. N.; Shao, J.; Hou, Y. H.; Chen, H.; Li, X. L. Bioorg. Med. Chem. Lett. 2016, 26, 1738.
[17] Chen, H.; Shao, J.; Zhu, M.; Li, X. L. Chin. J. Org. Chem. 2016, 36, 527(in Chinese). (陈华, 邵洁, 朱墨, 李小六, 有机化学, 2016, 36, 527.)
[18] Jain, A. K.; Vaidya, A.; Ravichandran, V.; Kashaw, S. K.; Agrawal, R. K. Bioorg. Med. Chem. 2012, 20, 3378.
[19] Chen, H.; Yin, Q. M.; Li, C. X.; Wang, E. K.; Gao, F.; Zhang, X. B.; Yin, Z.; Wei, S. N.; Li, X. L.; Meng, M.; Zhang, P. Z.; Li, N.; Zhang, J. C. ACS Med. Chem. Lett. 2011, 2, 845.
[20] Chen, H.; Bai, J.; Jiao, L. L.; Guo, Z. H.; Yin, Q. M.; Li, X. L. Bioorg. Med. Chem. 2009, 17, 3980.
[21] CCDC-1857715 for 10a and CCDC-1857714 for 11b contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
[22] Mosmann, T. J. Immunol. Methods 1983, 65, 55.
/
| 〈 |
|
〉 |