

基于新型帆船型络合物的超分子组装
收稿日期: 2018-10-01
修回日期: 2018-10-29
网络出版日期: 2018-11-26
基金资助
国家自然科学基金(No.21072066)、广东省省部产学研基金(No.2012B090700003)和华南师范大学青年教师科研培育基金(No.14KJ02)资助项目.
Supramolecular Assembly Based on the Novel Sail-Boat-Shaped Self-Complexes
Received date: 2018-10-01
Revised date: 2018-10-29
Online published: 2018-11-26
Supported by
Project supported by the National Natural Science Foundation of China (No. 21072066), the Key Project on the Integration of Industry, Education and Research of Guangdong Province (No. 2012B090700003), and the Foundation of Young Teachers of South China Normal University (No.14KJ02).
张世龙 , 蒋腊生 . 基于新型帆船型络合物的超分子组装[J]. 有机化学, 2019 , 39(3) : 720 -726 . DOI: 10.6023/cjoc201810001
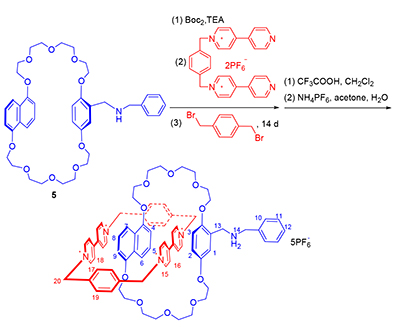
Owing to the unique macrocyclic-cavity structure of crown ether and the capability of complexing with guest molecules, our group successfully designed and synthesized a novel Sailboat-Shaped self-complex by using intramolecular hydrogen bonds, which are able to switch its configuration during the different pH value. Furthermore, a novel catenane which based on the sailboat-shaped self-complex was obtained by using intermolecular charge transfer interaction and through the template-directed method, and its structure was identified by 1H NMR, 13C NMR, HRMS and 1H-1H NOESY. It is hoped that it can be successfully controlled by the pH value conversion.
Key words: self-complexes; charge transfer interaction; catenane
[1] Desiraju, G. R. Acc. Chem. Res. 2002, 35, 565.
[2] Xiao, T. S.; Fan, J. S.; Zhou, H.; Lin, Q. S.; Yang, D. W. Angew. Chem., Int. Ed. 2016, 55, 6869.
[3] He, H. H.; Ye, Z. W.; Zheng, Y.; Xu, X.; Guo, C. L.; Xiao, Y.; Yang, W.; Qian, X. H.; Yang, Y. J. Chem. Commun. 2018, 54, 2842.
[4] Stoddart, J. F. Angew. Chem., Int. Ed., 2017, 56, 11094.
[5] Pressman, B. C.; Harris, H. J.; Jagger, W. S.; Jonson, J. H. Proc. Natl. Acad. Sci. U. S. A. 1967, 58, 32
[6] Yan, Z. Q.; Huang, Q. F.; Liang, W. T.; Yu, X. K.; Zhou, D. Y.; Wu, W. H.; Chruma, J. J.; Yang, C. Org. Lett. 2017, 19, 898.
[7] Chen, Y.; Huang, F. H.; Li, Z. T.; Liu, Y. Sci. China Chem. 2018, 61, 979.
[8] Jiao, Y.; Zhang, X. Acta Chim. Sinica 2018, 76, 659(in Chinese). (焦阳, 张希, 化学学报, 2018, 76, 659.)
[9] Guo, Q. H.; Zhao, L.; Wang, M. X. Angew. Chem., Int. Ed. 2015, 54, 8386.
[10] Chen, H. Q.; Fan, J. Z.; Hu, X. S.; Ma, J. W.; Wang, S. L.; Li, J.; Yu, Y. H.; Jia, X. S.; Li, C. J. Chem. Sci. 2015, 6, 197.
[11] Rudkevich, D. M.; Mercer-Chalmers, J. D.; Verboom, W.; Ungaro, R.; de Jong, F.; Reinhoudt, D. N. J. Am. Chem. Soc. 1995, 117, 6124.
[12] Rose, A. S.; Dennis, M. D.; George, W. G. J. Am. Chem. Soc. 1982, 104, 625.
[13] Zhu, S. S.; Carroll, P. J.; Swager, T. M. J. Am. Chem. Soc. 1996, 118, 8713.
[14] Jiang, F.; Chen, M.; Liang, J.; Gao, Z.; Tang, M.; Xu, Z.; Peng, B.; Zhu, S.; Jiang, L. Eur. J. Org. Chem. 2016, 3310.
[15] Fan, J. Q.; Jiang, L. S.; Zhang, M.; Wei, N.; Feng, Y. F.; Wang, H. Chin. Chem. Lett. 2008. 19, 513.
[16] Steed, J. W.; Atwood, J. L. Supramolecular Chemistry, Trans. by Zhao, Y.-P.; Sun, Z., Chemical Industry Press, Beijing, 2006(in Chinese). (Steed, J. W.; Atwood, J. L. 超分子化学, 赵耀鹏, 孙震译, 化学工业出版社, 北京, 2006.)
[17] Gururaja, T. L.; Ramasubbu, N.; Levine, M. J. Lett. Pept. Sci. 1996, 3, 79.
[18] Guo, Q. H.; Zhao, L.; Wang, M. X. Angew. Chem., Int. Ed. 2015, 54, 8386.
[19] Basu, S.; Coskun, A.; Friedman, D. C.; Olson, M. A.; Bentez, D.; Tkatchouk, E.; Barin, G.; Yang, J.; Fahrenbach, A. C.; Goddard Ⅲ, W. A.; Stoddart, J. F. Chem.-Eur. J. 2011, 17, 2107.
/
| 〈 |
|
〉 |