

铁卟啉催化酮和二甲亚砜以及醋酸铵的环化反应:吡啶的一步合成
收稿日期: 2018-09-07
修回日期: 2018-11-29
网络出版日期: 2018-12-05
基金资助
国家自然科学基金(No.21572049)资助项目.
Iron(Ⅲ) Porphyrin Catalyzed Cyclization of Ketones with Dimethyl Sulfoxide and Ammonium Acetate: One-Pot Synthesis of Pyridines
Received date: 2018-09-07
Revised date: 2018-11-29
Online published: 2018-12-05
Supported by
Project supported by the National Natural Science Foundations of China (No. 21572049).
刘玉峰 , 曹重仲 , 苏淼东 , 李慧 , 符美强 , 刘强 , 罗伟平 , 郭灿城 . 铁卟啉催化酮和二甲亚砜以及醋酸铵的环化反应:吡啶的一步合成[J]. 有机化学, 2019 , 39(1) : 129 -136 . DOI: 10.6023/cjoc201809014
An iron(Ⅲ)-porphyrin-catalyzed cyclization of ketones with dimethyl sulfoxide (DMSO) and ammonium acetate for the synthesis of unsymmetrical and symmetrical pyridines by employing DMSO as C4 or C6 source has been developed. Various aryl ketone derivatives react readily with DMSO, producing the pyridines in yields of 30%~85%. This method uses non-noble metals and proceeds under mild reaction conditions with operational simplicity, which thus allows the expedient assembly of pyridines from readily available ketones. Based on the preliminary experiments, a plausible mechanism of this transformation is disclosed.
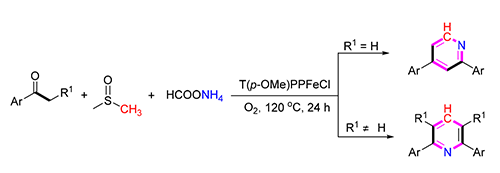
Key words: pyridine; iron (III) porphyrin; dimethyl sulfoxide; ketone
[1] (a) Eicher, T.; Hauptmann, S. The Chemistry of Heterocycles, Wiley-VCH, Weinheim, 2003.
(b) Katritzky, A. R.; Ramsden, C. A.; Scriven, E. F. V.; Taylor, R. J. Comprehensive Heterocyclic Chemistry, Elsevier, Oxford, 2008.
(c) Joule, J. A.; Mills, K. Heterocyclic Chemistry, Wiley, Chichester, 2010.
[2] (a) Forbes, I. T.; Johnson, C. N.; Jones, G. E.; Loudon, J.; Nicholass, J. M.; Thompson, M.; Upton, N. J. Med. Chem. 1990, 33, 2640.
(b) Zhmurenko, L. A.; Molodavkin, G. M.; Voronina, T. A.; Lezina, V. P. Pharm. Chem. J. 2012, 46, 15.
[3] Bahekar, R. H.; Jain, M. R.; Jadav, P. A.; Prajapati, V. M.; Patel, D. N.; Gupta, A. A.; Sharma, A.; Tom, R.; Bandyopadhya, D.; Modi, H.; Patel, P. R. Bioorg. Med. Chem. 2007, 15, 6782.
[4] Rival, Y.; Grassy, G.; Taudou, A.; Ecalle, R. Eur. J. Med. Chem. 1991, 26, 13.
[5] Zhuravel, I. O.; Kovalenko, S. M.; Ivachtchenko, A. V.; Balakin K. V.; Kazmirchuk, V. V. Bioorg. Med. Chem. Lett. 2005, 15, 5483.
[6] (a) Musonda, C. C.; Whitlock, G. A.; Witty, M. J.; Brun, R.; Kaiser, M. Bioorg. Med. Chem. Lett. 2009, 19, 401.
(b) Moreno, D.; Plano, D.; Baquedano, Y.; Jimeez-Ruiz, A.; Palop, J. A.; Sanmartn, C. Parasitol. Res. 2011, 108, 233.
[7] Rupert, K. C.; Henry, J. R.; Dodd, J. H.; Wadsworth, S. A.; Cavender, D. E.; Olini, G. C.; Fahmy, B.; Siekierka, J. J. Bioorg. Med. Chem. Lett. 2003, 13, 347.
[8] Kouznetsov, V. V.; Mendez, L. Y. V.; Tibaduiza, B.; Ochoa, C.; Pereira, D. M.; Ruiz, J. J. N.; Portillo, C. F.; Serrano, S. M.; Barrio, A. G.; Bahsas, A.; Amaro-Luis, J. Arch. Pharm. Pharm. Med. Chem. 2004, 337, 127.
[9] (a) Gibson, V. C.; Redshaw, C.; Solan, G. A. Chem. Rev. 2007, 107, 1745.
(b) Rycke, N. D.; Couty, F.; David, O. R. P. Chem.-Eur. J. 2011, 17, 12852.
[10] (a) Allais, C.; Grassot, J. M.; Rodriguez, J.; Constantieux, T. Chem. Rev. 2014, 114, 10829.
(b) Zhao, M. N.; Hui, R. R.; Ren, Z. H.; Wang, Y. Y.; Guan, Z. H. Org. Lett. 2014, 16, 3082.
(c) Bai, Y.; Tang, L. C.; Huang, H. W.; Deng, G. J. Org. Biomol. Chem. 2015, 13, 4404.
(d) Yan, Y. Z.; Li, H. Y.; Li, Z.; Niu, B.; Shi, M. M.; Liu, Y. Q. J. Org. Chem. 2017, 82, 8628.
[11] (a) Meunier, B. Biomimetic Oxidations Mediated by Metal Complexes, Imperial College Press, London, 2000.
(b) Meunier, B.; de Visser, S. P.; Shaik, S. Chem. Rev. 2004, 104, 3947.
(c) Zhou, X. T.; Ji, H. B.; Pei, L. X.; She, Y. B.; Xu, J. C.; Wang, L. F. Chin. J. Org. Chem. 2007, 27, 1039(in Chinese). (周贤太, 纪红兵, 裴丽霞, 佘远斌, 徐建昌, 王乐夫, 有机化学, 2007, 27, 1039.)
[12] (a) Collman, J. P.; Wang, Z.; Straumanis, A.; Quelquejeu, M.; Rose, E. J. Am. Chem. Soc. 1999, 121, 460.
(b) Yu, X. Q.; Huang, J. S.; Yu, W. Y.; Che, C. M. J. Am. Chem. Soc. 2000, 122, 5337.
(c) Au, S. M.; Huang, J. S.; Yu, W. Y.; Fung, W. H.; Che, C. M. J. Am. Chem. Soc. 1999, 121, 9120.
[13] (a) Li, Y.; Huang, J. S.; Zhou, Z. Y.; Che, C. M. J. Am. Chem. Soc. 2001, 123, 4843.
(b) Bartoli, J. F.; Mansuy, V. M.; Barch-Ozette, K. L.; Palacio, M.; Battioni, P.; Mansuy, D. Chem. Commun. 2000, 827.
(c) Yang, J.; Weinberg, R.; Breslow, R. Chem. Commun. 2000, 531.
(d) Yu, W. Y.; Huang, J. S.; Zhou, Z. Y.; Che, C. M. Org. Lett. 2000, 2, 2233.
[14] (a) Li, Y. F.; Guo, C, C.; Yan, X. H.; Liu, Q. J. Porphyrins Phthalocyanines 2006, 10, 942.
(b) Jiang, Q.; Sheng, W. B.; Tian, M.; Tang, J. S.; Guo, C. C. Eur. J. Org. Chem. 2013, 10, 1861.
(c) Sheng, W. B.; Jiang, Q.; Luo, W. P.; Guo, C. C. J. Org. Chem. 2013, 78, 5691.
[15] (a) Pan, X. J.; Liu, Q.; Chang, L. M.; Yuan, G. Q. RSC Adv. 2015, 5, 51183.
(b) Wu, X.; Zhang, J. H.; Liu, S.; Gao, Q. H.; Wu, A. X. Adv. Synth. Catal. 2016, 358, 218.
[16] Liu, Y. F.; Ji, P. Y.; Xu, J. W.; Hu, Y. Q.; Liu, Q.; Luo, W. P.; Guo, C. C. J. Org. Chem. 2017, 82, 7159.
/
| 〈 |
|
〉 |