

芳基吡啶酮腙类化合物的设计、合成及杀菌活性研究
收稿日期: 2018-10-16
修回日期: 2018-12-05
网络出版日期: 2018-12-28
基金资助
国家自然科学基金(No.21572265)资助项目.
Design, Synthesis and Activity Evaluation of Arylpyridone Hydrazones
Received date: 2018-10-16
Revised date: 2018-12-05
Online published: 2018-12-28
Supported by
Project supported by the National Natural Science Foundation of China (No.21572265).
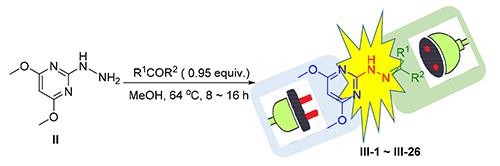
以吡啶二芳酮为分子插件,以嘧菌腙为母体化合物设计并合成了一系列新型芳基吡啶酮腙类化合物.产物结构经1H NMR、13C NMR及HRMS确认,并采用生长速率法对所有化合物进行了离体抑菌活性测试.结果表明:在浓度为70 μmol/L时,大部分化合物对所选8种病菌具有一定的抑制活性,其中2'-甲基乙酰基苯-4,6-二甲氧基嘧啶-2-腙(Ⅲ-3)和2-(2'-((4''-溴-苯基)(3''-氯-吡啶-4''-基)-亚甲基)-肼基)-4,6-二甲氧基嘧啶(Ⅲ-18)对病原菌抑制效果明显高于对照药嘧菌腙.Ⅲ-3对番茄灰霉病菌EC50为22.18 μmol/L (嘧菌腙为31.38 μmol/L),Ⅲ-18对水稻纹枯病菌EC50小于0.35 μmol/L,比对照药嘧菌腙提高了260倍以上.
王家尧 , 陶晗 , 金蜜 , 李丽莎 , 肖玉梅 , 李佳奇 , 覃兆海 . 芳基吡啶酮腙类化合物的设计、合成及杀菌活性研究[J]. 有机化学, 2019 , 39(4) : 1044 -1052 . DOI: 10.6023/cjoc201810019
A series of novel arylpyridone hydrazones were designed and synthesized by using arylpyridinylmethanone as the plug-in molecules and ferimzone as parent compound. The structure of the products were confirmed by 1H NMR, 13C NMR and HRMS. The antifungal activities of all compounds were tested by growth rate method in vitro. The results showed that most of the compounds inhibited eight selected pathogens at the concentration of 70 μmol/L. The inhibitory effect of 2'-methylacetylbenzene-4,6-dimethoxypyrimidine-2-hydrazone (Ⅲ-3) and 2-(2'-((4'-bromo-phenyl)(3'-chloro-pyridine-4'-yl)-methylene-hydrazine)-4,6-dimethoxypyrimidine (Ⅲ-18) on pathogenic fungi was significantly higher than that of ferimzone, in which compound Ⅲ-3 has a great degree of inhibition on Botrytis cinerea with EC50 value of 22.18 μmol/L, and EC50 value of compound Ⅲ-18 is less than 0.35 umol/L on Thanatephorus cucumeris, which is 260 times higher than that of control.

[1] Ameen, D.; Snape, T. J. Med. Chem. Commun. 2013, 4, 893.
[2] Liu, X. L.; Xiao, Y. M.; Li, J. Q.; Fu, B.; Qin, Z. H. Mol. Diversity DOI:10.1007/S11030-018-9895-3.
[3] Song, M. Y.; Cao, C. Y.; He, Q. R.; Dong, Q. M.; Li, D.; Tang, J. J.; Gao, J. M. Bioorg. Med. Chem. 2017, 25, 5290.
[4] Roszkowski, A. P. J. Pharmacol. Exp. Ther. 1965, 149, 288.
[5] Abrahamsson, S.; Nilsson, B. J. Org. Chem. 1966, 31, 3631.
[6] Anon Fain Kemikaru 2010, 39, 39.
[7] Krol, M.; Podsadni, P.; Turlo, J.; Ufnal, M.; Drapala, A.; Szulczyk, B.; Dawidowski, M. Eur. J. Pharm. Sci. 2016, 81, 42.
[8] Reder R. F.; Danilo P. J.; Rosen M. R. Eur. J. Pharmacol. 1980, 61, 321.
[9] Hart, T.; Macias, A. T.; Benwell, K.; Brooks, T.; D'Alessandro, J.; Dokurno, P.; Francis, G.; Gibbons, B.; Haymes, T.; Kennett, G.; Lightowler, S.; Mansell, H.; Matassova, N.; Misra, A.; Padfield, A.; Parsons, R.; Pratt, R.; Robertson, A.; Walls, S.; Wong, M.; Roughley, S. Bioorg. Med. Chem. Lett. 2009, 19, 4241.
[10] Tanaka, I.; Otani, S.; Nakayashiki, J.; Fujita, S. Yakuzaigaku 1964, 24, 303.
[11] Tanaka, I.; Otani, S.; Seki, T.; Shinkai, H.; Kurashige, T. Yakuzaigaku 1965, 25, 31.
[12] Che, C. L.; Yang, D. Y.; Wan, C.; Wang, J. Y.; Liu, X. L.; Zhao, F. H.; Qin, Z. H. Chin. J. Pestic. Sci. 2017, 19, 533(in Chinese). (车传亮, 杨冬燕, 万川, 王家尧, 刘雪莲, 赵峰海, 覃兆海, 农药学学报, 2017, 19, 533.)
[13] Mu, C. W.; Yuan, H. Z.; Li, N.; Fun, B.; Xiao, Y. M.; Ma, Y. Q.; Qi, S. H.; Qin, Z. H. Chem. J. Chin. Univ. 2007, 28, 1902(in Chinese) (慕长炜, 袁会珠, 李楠, 傅滨, 肖玉梅, 马永强, 齐淑华, 覃兆海, 高等学校化学学报, 2007, 28, 1902.)
[14] Xiao, Y. M.; Esser, L.; Zhou, F.; Li, C.; Zhou, Y. H.; Yu, C.-A.; Qin, Z. H.; Xia, D. PLoS One 2014, 9, e93765.
[15] Yan, X. J.; Qin, W. C.; Sun, L. P.; Qi, S. H.; Yang, D. B.; Qin, Z. H.; Yuan, H. Z. J. Agric. Food Chem. 2010, 58, 2720.
[16] Xiao, Y. M.; Yang, X. L.; Li, B.; Yuan, H. Z.; Wan, S. Q.; Xu, Y. J.; Qin, Z. H. Molecules 2011, 16, 8945.
[17] Li, X.; Han, X.; He, M. M. Xiao, Y. M.; Qin, Z. H. Bioorg. Med. Chem. Lett. 2016, 26, 5936.
[18] Li, X. Y.; Wan, C.; Du, S. J.; Li, H.; Yuan, H. Z.; Jiang, J. Z.; Xiao, Y. M.; Qin, Z. H. Chem. J. Chin. Univ. 2015, 36, 2415(in Chinese). (李秀云, 万川, 杜士杰, 李红, 袁会珠, 蒋家珍, 肖玉梅, 覃兆海, 高等学校化学学报, 2015, 36, 2415.)
[19] Zhang, L. M.S. Thesis, China Agricultural University, Beijing, 2017 (in Chinese). (张磊, 硕士学位论文, 中国农业大学, 北京, 2017.)
[20] Okuno, T.; Furusawa, I.; Matsuura, K.; Shishiyama, J. A. Phytopathol. Soc. Jpn. 1989, 55, 281.
[21] Cechin, S. R.; Schetinger, M. R. C.; Zanatta, N.; Madruga, C. C.; Pacholski, I. L.; Flores, D. C.; Bonacorso, H. G.; Martins, M. A. P.; Morsch, V. M. Chem. Res. Toxicol. 2003, 16, 1433.
[22] Bonnett, S. A.; Ollinger, J.; Chandrasekera, S.; Florio, S.; O'Malley, T.; Files, M.; Jee, J. A.; Ahn, J.; Casey, A.; Ovechkina, Y.; Roberts, D.; Korkegian, A.; Parish, T. ACS Infect. Dis. 2016, 2, 893.
[23] Chen, N. C. Pesticide Bioassay Technology, China Agriculture Press, Beijing, 1991, pp. 208~239(in Chinese). (陈年春, 农药生物测定技术, 中国农业出版社, 北京, 1991, pp. 208~239.)
[24] Konishi, K.; Kuragano, T.; Matsuura, K. Agric. Biol. Chem. 1986, 50, 2427.
/
| 〈 |
|
〉 |