

银催化的3-三氟乙酰基吲哚与异氰酸酯反应: (Z)-β-三氟甲基取代的脱氢色氨酸衍生物的高效合成
收稿日期: 2020-08-13
修回日期: 2020-09-06
网络出版日期: 2020-09-22
基金资助
国家自然科学基金(21572126); 国家自然科学基金(21901152); 国家自然科学基金(U1904195); 河南省科技攻关(202102310003); 河南省科技攻关(192102210161); 河南省科技创新杰出人才(2018JQ0011)
Ag-Catalyzed Reactions of 3-Trifluoroacetyl Indoles and Isocyano- acetates: An Efficient Process to (Z)-β-Trifluoromethylated Dehydrotryptophan Derivatives
Received date: 2020-08-13
Revised date: 2020-09-06
Online published: 2020-09-22
Supported by
the National Natural Science Foundation of China(21572126); the National Natural Science Foundation of China(21901152); the National Natural Science Foundation of China(U1904195); the Key Scientific and Technological Project of Henan Province(202102310003); the Key Scientific and Technological Project of Henan Province(192102210161); the Science and Technology Innovation Talents of Henan Province(2018JQ0011)
张明亮 , 赵聘 , 袁浩 , 张安安 , 张文宇 , 郑琳琳 , 吴冬青 , 刘澜涛 . 银催化的3-三氟乙酰基吲哚与异氰酸酯反应: (Z)-β-三氟甲基取代的脱氢色氨酸衍生物的高效合成[J]. 有机化学, 2021 , 41(2) : 669 -676 . DOI: 10.6023/cjoc202008023
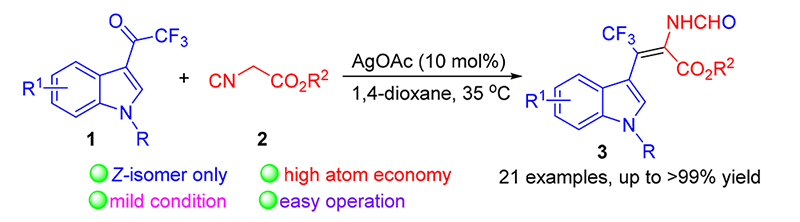
Catalytic cyclization-rearrangement reaction of 3-trifluoroacetyl indole and isocyanoacetate was achieved with AgOAc as catalyst in mild conditions. A serious of β-trifluoromethylated dehydrotryptophan derivatives were obtained with single Z-isomer in excellent yields (up to >99% yields). The large scale experiment proceeded smothly genereting desired products in up to 98% isolated yield, which demostrated the practicality of the method. The plausible mechanism was aslo proposed. This transformation has features of high atom economy, mild reaction condition and easy operation.
Key words: 3-trifluoroacetyl indole; isocyanoacetate; dehydrotryptophan
| [1] | (a) Purser S.; Moore P.R.; Swallow S.; Gouverneur V. Chem. Soc. Rev. 2008, 37, 320. |
| [1] | (b) Wang J.; Sánchez-Roselló M.; Ace?a J.L.; del Pozo C.; Sorochinsky A.E.; Fustero S.; Soloshonok V.A.; Liu H. Chem. Rev. 2014, 114, 2432. |
| [1] | (c) Gillis E.P.; Eastman K.J.; Hill M.D.; Donnelly D.J.; Meanwell N.A. J. Med. Chem. 2015, 58, 8315. |
| [1] | (d) Zhou Y.; Wang J.; Gu Z.; Wang S.; Zhu W.; Acena J.L.; Soloshonok V.A.; Izawa K.; Liu H. Chem. Rev. 2016, 116, 422. |
| [2] | (a) Schlosser M. Angew. Chem., Int. Ed. 2006, 45, 5432. |
| [2] | (b) Hagmann W.K. J. Med. Chem. 2008, 51, 4359. |
| [2] | (c) Kirk K.L. Org. Process Res. Dev. 2008, 12, 305. |
| [2] | (d) Meanwell N.A. J. Med. Chem. 2011, 54, 2529. |
| [3] | For selected reviews, see: a Yang, X.; Wu, T.; Phipps, R. J.; Toste, F. D.Chem. Rev. 2015, 115, 826. |
| [3] | (b) Ma J.-A.; Cahard D. Chem. Rev. 2008, 108, PR1. |
| [3] | (c) Ma J.-A.; Cahard D. Chem. Rev. 2004, 104, 6119. |
| [3] | (d) Chu L.; Qing F.-L. Acc. Chem. Res. 2014, 47, 1513. |
| [4] | Hassinger H.L.; Soll R.M.; Gribble G.W. Tetrahedron Lett. 1998, 39, 3095. |
| [5] | Li J.; Li B.; Chen X.; Zhang G. Synlett 2003, 1447. |
| [6] | (a) Lanke V.; Bettadapur K.R.; Prabhu K.R. Org. Lett. 2016, 18, 5496. |
| [6] | (b) Lanke V.; Prabhu K.R. Chem. Commun. 2017, 53, 5117. |
| [6] | (c) Sherikar M.S.; Kapanaiah R.; Prabhu K.R. Chem. Commun. 2018, 54, 11200. |
| [6] | (d) Bettadapur K.R.; Kapanaiah R.; Lanke V.; Prabhu K.R. J. Org. Chem. 2018, 83, 1810. |
| [7] | (a) Marco R.D.; Cavina L.; Greco A.; Gentilucci L. Amino Acids 2014, 46, 2823. |
| [7] | Yeh E. Garneau S. Walsh C.T. Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 3960. |
| [7] | (c) Bittner S.; Scherzer R.; Harlev E. Amino Acids 2007, 33, 19. |
| [7] | (d) Jiang J.; Ma Z.; Castle S.L. Tetrahedron 2015, 71, 5431. |
| [7] | (e) Lorena M.-T.; Albornoz-Grados, A.; Bertran, A.; Albericio, F.; Lavilla, R.Chem. Commun. 2017, 53, 2740. |
| [7] | (f) Kelley E.W.; Norman S.G.; Scheerer. J. R.Org. Biomol. Chem. 2017, 15, 8634. |
| [8] | (a) Olsen E.K.; Hansen E.; Moodie L. W. K.; Isaksson J.; Sep?i? K.; Cergolj M.; Svenson J.; Andersen J.H. Org. Biomol. Chem. 2016, 14, 1629. |
| [8] | (b) Lidgren G.; Bohlin L.; Bergman J. Tetrahedron Lett. 1986, 27, 3283. |
| [9] | Du F.-Y.; Li X.-M.; Li C.-S.; Shang Z.; Wang B.-G. Bioorg. Med. Chem. Lett. 2012, 22, 4650. |
| [10] | Block R.J. Chem. Rev. 1946, 38, 501. |
| [11] | Kimura R.; Nagano T.; Kinoshita H. Bull. Chem. Soc. Jpn. 2002, 75, 2517. |
| [12] | Kinoshita H.; Kometani M.; Ihara K.; Kimura R. Bull. Chem. Soc. Jpn. 2009, 82, 364. |
| [13] | (a) RajanBabu T.V.; Lim H.J.; Gallucci J.C. Org. Lett. 2010, 12, 2162. |
| [13] | (b) Feldman K.S.; Eastman K.J.; Lessene G. Org. Lett. 2002, 4, 3525. |
| [14] | Baran P.S.; Hafensteiner B.D.; Ambhaikar N.B.; Guerrero C. A.; Gallagher J.D. J. Am. Chem. Soc. 2006, 128, 8678. |
| [15] | (a) Merlic C.A.; Semmelhack M.F. J. Organomet. Chem. 1990, 391, C23. |
| [15] | (b) Harrington P.J.; Hegedus L. S.; Mcdaniel K.F. J. Am. Chem. Soc. 1987, 109, 4335. |
| [16] | (a) Yokoyama Y.; Takahashi M.; Takashima M.; Kohno Y.; Kobayashi H.; Kataoka K.; Shidori K.; Murakami Y. Chem. Pharm. Bull. 1994, 42, 832. |
| [16] | (b) Yokoyama Y.; Matsumoto T.; Murakami Y. J. Org. Chem. 1995, 60, 1486. |
| [17] | Kaur H.; Heapy A.M.; Brimble M.A. Org. Biomol. Chem. 2011, 9, 5897. |
| [18] | (a) Gulevich A.V.; Zhdanko A.G.; Orru R. V. A.; Nenajdenko V.G. Chem. Rev. 2010, 110, 5235. |
| [18] | (b) Elders N.; Ruijter E.; de Kanter F. J. J.; Groen M.B.; Orru R. V. A.Chem. Eur. J. 2008, 14, 4961. |
| [18] | (c) Enders D.; Chen Z.-X.; Raabe G. Synthesis 2005, 306. |
/
| 〈 |
|
〉 |