

近年来炔酰胺参与的成环反应研究进展
收稿日期: 2020-09-09
修回日期: 2020-10-21
网络出版日期: 2020-11-19
基金资助
国家自然科学基金(81803406); 国家自然科学基金(U1804283); 河南省教育厅(19A150009)
Recent Advances in the Ring-Forming Reactions of Ynamides
Received date: 2020-09-09
Revised date: 2020-10-21
Online published: 2020-11-19
Supported by
National Natural Science Foundation of China(81803406); National Natural Science Foundation of China(U1804283); Foundation of Henan Educational Committee(19A150009)
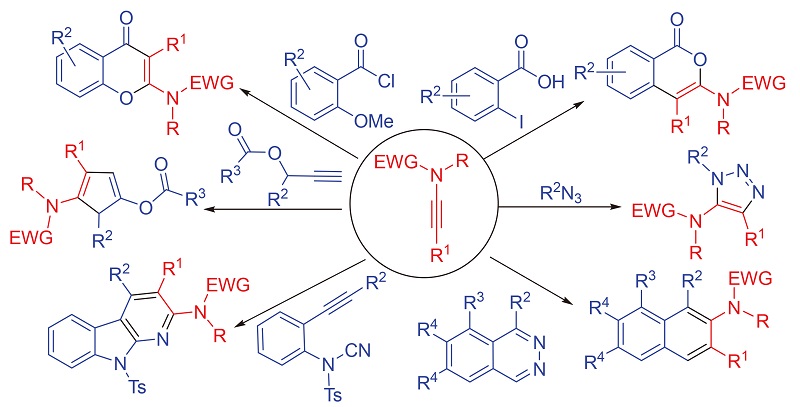
炔酰胺作为一种杂原子取代的炔, 具有独特的结构特征, 其反应活性和稳定性能够达到很好的平衡, 已作为一种多功能型的有机合成子被广泛应用于有机化学中. 尤其是随着炔酰胺高效型和原子经济型制备方法的出现, 炔酰胺参与的反应类型渐渐趋于多样化. 在这多样化的反应类型中有关炔酰胺成环反应的研究占主要部分, 这跟炔酰胺的结构特征密切相关, 炔酰胺炔基上的α碳具有一定的亲电性, β碳具有一定的亲核性, 有利于成环反应的发生. 另一方面, 炔酰胺参与成环反应所生成的含氮环状化合物, 为大量活性天然产物和药物分子的构建提供了重要的结构单元, 因此, 关于炔酰胺参与的成环反应的研究具有重要的意义. 概述了近年来炔酰胺参与成环反应的最新研究进展.
周欣悦 , 梁宗显 , 王晓娜 . 近年来炔酰胺参与的成环反应研究进展[J]. 有机化学, 2021 , 41(4) : 1288 -1318 . DOI: 10.6023/cjoc202009025
As a subgroup of heteroatom-substituted alkynes, ynamide has unique structural characteristics. The alkynyl group of ynamide is activated by the conjugation of nitrogen lone pair, at the same time by simply placing an electron-withdrawing group on the nitrogen atom, the donating ability of the nitrogen lone pair toward the alkynyl motif is greatly diminished through resonance delocalization into the electron-withdrawing group. Consequently, ynamides have set the gold standard for balancing reactivity and stability, and have become highly versatile organic synthons applicable to a diverse array of transformations that can be useful for natural product syntheses. Especially with the emergence of efficient and atom-economical preparation methods, the field of ynamide chemistry has rapidly expanded. Among the various reported organic transformations involving ynamides, research on ring-forming reactions is the most prevalent. On one hand, this is closely related to the structural characteristic of ynamide (this activated alkyne has both electrophilic and nucleophilic properties, which are conducive to the formation of rings). On the other hand, the reactions of ynamides directly afford nitrogen-containing cyclic compounds, which provide access to important structural entities found in natural products and pharmacophores. These properties have contributed to a dramatic increase in the number of publications over the past few years. This review aims to examine the literatures from late 2010 through early 2020 related to the use of ynamides in ring-forming transformations. And it is organized by the reaction types of ring formation including radical cyclizations, ring-closing metathesis, transition metal and non-transition metal mediated cyclizations, cycloaddition reactions, and rearrangements. However, due to the emergence of a large number of ring-forming reaction involving ynamides, not all the beautiful recent works are presented, and representative examples are selected to demonstrate the scope and mechanistic insight of these ring-forming transformations.

Key words: ynamide; nitrogen heterocycle; cyclization; cycloaddition; rearrangement
| [1] | Viehe, H.G. Angew. Chem., Int. Ed. 1963, 2,477. |
| [2] | Janousek, Z.; Collard, J.; Viehe, H.G. Angew. Chem., Int. Ed. 1972, 11,917. |
| [3] | (a) Evano, G.; Coste, A.; Jouvin, K. Angew. Chem., Int. Ed. 2010, 49,2840. |
| [3] | (b) DeKorver, K.A.; Li, H.; Lohse, A.G.; Hayashi, R.; Lu, Z.; Zhang, Y.; Hsung, R.P. Chem. Rev. 2010, 110,5064. |
| [3] | (c) Wang, X.-N.; Yeom, H.-S.; Fang, L.-C.; He, S.; Ma, Z.-X.; Kedrowski, B.L.; Hsung, R.P. Acc. Chem. Res. 2014, 47,560. |
| [3] | (d) Evano, G.; Theunissen, C.; Lecomte, M. Aldrichim. Acta 2015, 48,59. |
| [3] | (e) Pan, F.; Shu, C.; Ye, L.-W. Org. Biomol. Chem. 2016, 14,9456. |
| [3] | (f) Liao, Y.; Zhu, L.; Yu, Y.; Chen, G.; Huang, X. Chin. J. Org. Chem. 2017, 37,2785. (in Chinese) |
| [3] | ( 廖云, 朱磊, 俞颖华, 陈贵, 黄学良, 有机化学, 2017, 37,2785.) 2578dc51-76c9-4272-b262-bd95d94f25c8 |
| [3] | (g) Zhou, B.; Tan, T.-D.; Zhu, X.-Q.; Shang, M.; Ye, L.-W. ACS Catal. 2019, 9,6393. |
| [3] | (h) Hong, F.-L.; Ye, L.-W. Acc. Chem. Res. 2020, 53,2003. |
| [4] | Balieu, S.; Toutah, K.; Carro, L.; Chamoreau, L.-M.; Rousselière, H.; Courillon, C. Tetrahedron Lett. 2011, 52,2876. fd0395e4-17bb-4cea-b580-bf27d1e41cf9 |
| [5] | Marion, F.; Courillon, C.; Malacria, M. Org. Lett. 2003, 5,5095. |
| [6] | (a) Chemla, F.; Dulong, F.; Ferreira, F.; Nüllen, M.; Pérez-Luna, A. Synthesis 2011,1347. |
| [6] | (b) Romain, E.; Fopp, C.; Chemla, F.; Ferreira, F.; Jackowski, O.; Oestreich, M.; Perez-Luna, A. Angew. Chem., Int. Ed. 2014, 53,11333. |
| [6] | (c) de la Vega-Hernández, K.; Romain, E.; Coffinet, A.; Bijouard, K.; Gontard, G.; Chemla, F.; Ferreira, F.; Jackowski, O.; Perez-Luna, A. J. Am. Chem. Soc. 2018, 140,17632. |
| [7] | Dutta, S.; Mallick, R.K.; Prasad, R.; Gandon, V.; Sahoo, A.K. Angew. Chem., Int. Ed. 2019, 58,2289. |
| [8] | Casse, M.; Nisole, C.; Dossmann, H.; Gimbert, Y.; Fourquez, J.-M.; Haberkorn, L.; Ollivier, C.; Fensterbank, L. Sci. China: Chem. 2019, 62,1542. |
| [9] | Wakamatsu, H.; Sakagami, M.; Hanata, M.; Takeshita, M.; Mori, M. Macromol. Symp. 2010, 293,5. |
| [10] | Wakamatsu, H.; Sasaki, Y.; Kawahata, M.; Yamaguchi, K.; Yoshimura, Y. Synthesis 2018, 50,3467. |
| [11] | Poloukhtine, A.; Rassadin, V.; Kuzmin, A.; Popik, V.V. J. Org. Chem. 2010, 75,5953. |
| [12] | Gati, W.; Rammah, M.M.; Rammah, M.B.; Couty, F.; Evano, G. J. Am. Chem. Soc. 2012, 134,9078. |
| [13] | Meng, T.-J.; Chen, R.-X.; Liu, L.-T.; Wang, T.; Liu, X.-M.; Zhao, W.-X. Chin. J. Org. Chem. 2015, 35,2108. (in Chinese) |
| [13] | ( 孟团结, 陈荣祥, 刘澜涛, 王涛, 刘新明, 赵文献, 有机化学, 2015, 35,2108.) |
| [14] | Kong, Y.; Jiang, K.; Cao, J.; Fu, L.; Yu, L.; Lai, G.; Cui, Y.; Hu, Z.; Wang, G. Org. Lett. 2013, 15,422. |
| [15] | Willumstad, T.P.; Boudreau, P.D.; Danheiser, R.L. J. Org. Chem. 2015, 80,11794. |
| [16] | Liu, H.; Yang, Y.; Wang, S.; Wu, J.; Wang, X.-N.; Chang, J. Org. Lett. 2015, 17,4472. |
| [17] | Lecomte, M.; Evano, G. Angew. Chem., Int. Ed. 2016, 55,4547. |
| [18] | Wang, Y.; Lin, J.; Wang, X; Wang, G.; Zhang, X.; Yao, B.; Zhao, Y.; Yu, P.; Lin, B.; Liu, Y.; Cheng, M. Chem.-Eur. J. 2018, 24,4026. |
| [19] | Brutiu, B.R.; Bubeneck, W.A.; Cvetkovic, O.; Li, J.; Maulide, N. Monatsh. Chem. 2018, 150,3. |
| [20] | Zhang, J.; Li, S.; Qiao, Y.; Peng, C.; Wang, X.-N.; Chang, J. Chem. Commun. 2018, 54,12455. |
| [21] | Yoo, H.J.; Youn, S.W. Org. Lett. 2019, 21,3422. |
| [22] | Li, L.; Zhou, B.; Wang, Y.-H.; Shu, C.; Pan, Y.-F.; Lu, X.; Ye, L.-W. Angew. Chem., Int. Ed. 2015, 54,8245. |
| [23] | Li, H.-H.; Ye, S.-H.; Chen, Y.-B.; Luo, W.-F.; Qian, P.-C.; Ye, L.-W. Chin. J. Chem. 2020, 38,263. |
| [24] | Nishimura, T.; Takiguchi, Y.; Maeda, Y.; Hayashi, T. Adv. Synth. Catal. 2013, 355,1374. |
| [25] | Liao, Y.; Lu, Q.; Chen, G.; Yu, Y.; Li, C.; Huang, X. ACS Catal. 2017, 7,7529. |
| [26] | Okamoto, N.; Yanada, R.; Sueda, T. Eur. J. Org. Chem. 2019,691. |
| [27] | Greenaway, R.L.; Campbell, C.D.; Holton, O.T.; Russell, C.A.; Anderson, E.A. Chem.-Eur. J. 2011, 17,14366. |
| [28] | Greenaway, R.L.; Campbell, C.D.; Chapman, H.A.; Anderson, E.A. Adv. Synth. Catal. 2012, 354,3187. |
| [29] | Cao, J.; Xu, Y.; Kong, Y.; Cui, Y.; Hu, Z.; Wang, G.; Deng, Y.; Lai, G. Org. Lett. 2012, 14,38. |
| [30] | Huang, H.; He, G.; Zhu, G.; Zhu, X.; Qiu, S.; Zhu, H. J. Org. Chem. 2015, 80,3480. |
| [31] | Liu, H.; Yang, Y.; Wu, J.; Wang, X.-N.; Chang, J. Chem. Commun. 2016, 52,6801. |
| [32] | Reddy, A.S.; Kumari, A.L. S.; Swamy, K.C. K. Tetrahedron 2017, 73,2766. |
| [33] | Bhunia, S.; Chang, C.-J.; Liu, R.-S. Org. Lett. 2012, 14,5522. |
| [34] | Shen, W.-B.; Xiao, X.-Y.; Sun, Q.; Zhou, B.; Zhu, X.-Q.; Yan, J.-Z.; Lu, X.; Ye, L.-W. Angew. Chem., Int. Ed. 2017, 56,605. |
| [35] | Hashmi, A.S. K.; Schuster, A.M.; Zimmer, M.; Rominger, F. Chem.-Eur. J. 2011, 17,5511. |
| [36] | Gati, W.; Couty, F.; Boubaker, T.; Rammah, M.M.; Rammah, M.B.; Evano, G. Org. Lett. 2013, 15,3122. |
| [37] | Reddy, A.S.; Reddy, M.N.; Swamy, K.C. K. RSC Adv. 2014, 4,28359. |
| [38] | Nickel, J.; Fernández, M.; Klier, L.; Knochel, P. Chem.-Eur. J. 2016, 22,14397. |
| [39] | Baguia, H.; Deldaele, C.; Romero, E.; Michelet, B.; Evano, G. Synthesis 2018, 50,3022. |
| [40] | Hong, F.-L.; Wang, Z.-S.; Wei, D.-D.; Zhai, T.-Y.; Deng, G.-C.; Lu, X.; Liu, R.-S.; Ye, L.-W. J. Am. Chem. Soc. 2019, 141,16961. |
| [41] | Hong, F.-L.; Chen, Y.-B.; Ye, S.-H.; Zhu, G.-Y.; Zhu, X.-Q.; Lu, X.; Liu, R.-S.; Ye, L.-W. J. Am. Chem. Soc. 2020, 142,7618. |
| [42] | Liu, X.; Wang, Z.-S.; Zhai, T.-Y.; Luo, C.; Zhang, Y.-P.; Chen, Y.-B.; Deng, C.; Liu, R.-S.; Ye, L.-W. Angew. Chem., Int. Ed. 2020, 59,17984. |
| [43] | Garcia, P.; Harrak, Y.; Diab, L.; Cordier, P.; Ollivier, C.; Gandon, V.; Malacria, M.; Fensterbank, L.; Aubert, C. Org. Lett. 2011, 13,2952. |
| [44] | Liang, G.; Ji, Y.; Liu, H.; Pang, Y.; Zhou, B.; Cheng, M.; Liu, Y.; Lin, B.; Liu, Y. Adv. Synth. Catal. 2020, 362,192. |
| [45] | Ieawsuwan, W.; Ruchirawat, S. Heterocycles 2019, 99,100. |
| [46] | Blanco Jaimes, M.C.; Weingand, V.; Rominger, F.; Hashmi, A.S. K. Chem.-Eur. J. 2013, 19,12504. |
| [47] | Rettenmeier, E.; Schuster, A.M.; Rudolph, M.; Rominger, F.; Gade, C.A.; Hashmi, A.S. K. Angew. Chem., Int. Ed. 2013, 52,5880. |
| [48] | Tokimizu, Y.; Oishi, S.; Fujii, N.; Ohno, H. Org. Lett. 2014, 16,3138. |
| [49] | Tokimizu, Y.; Wieteck, M.; Rudolph, M.; Oishi, S.; Fujii, N.; Hashmi, A.S. K.; Ohno, H. Org. Lett. 2015, 17,604. |
| [50] | Shu, C.; Wang, Y.-H.; Zhou, B.; Li, X.-L.; Ping, Y.-F.; Lü, X.; Ye, L.-W. J. Am. Chem. Soc. 2015, 137,9567. |
| [51] | Shu, C.; Wang, Y.-H.; Shen, C.-H.; Ruan, P.-P.; Lü, X.; Ye, L.-W. Org. Lett. 2016, 18,3254. |
| [52] | Jin, H.; Tian, B.; Song, X.; Xie, J.; Rudolph, M.; Rominger, F.; Hashmi, A.S. K. Angew. Chem., Int. Ed. 2016, 55,12688. |
| [53] | Shen, W.-B.; Sun, Q.; Li, L.; Liu, X.; Zhou, B.; Yan, J.-Z.; Lü, X.; Ye, L.-W. Nat. Commun. 2017, 8,1748. |
| [54] | Ito, M.; Kawasaki, R.; Kanyiva, K.S.; Shibata, T. Chem.-Eur. J. 2018, 24,3721. |
| [55] | Xu, W.; Wang, G.; Xie, X.; Liu, Y. Org. Lett. 2018, 20,3273. |
| [56] | Hsu, Y.-C.; Hsieh, S.-A.; Liu, R.-S. Chem.-Eur. J. 2019, 25,5288. |
| [57] | Vanjari, R.; Dutta, S.; Gogoi, M.P.; Gandon, V.; Sahoo, A.K. Org. Lett. 2018, 20,8077. |
| [58] | Rode, N.D.; Arcadi, A.; Nicola, A.D.; Marinelli, F.; Michelet, V. Org. Lett. 2018, 20,5103. |
| [59] | Febvay, J.; Sanogo, Y.; Retailleau, P.; Gogoi, M.P.; Sahoo, A.K.; Marinetti, A.; Voituriez, A. Org. Lett. 2019, 21,9281. |
| [60] | Li, H.; Hsung, R.P.; Dekorver, K.A.; Wei, Y. Org. Lett. 2010, 12,3780. |
| [61] | Schotes, C.; Mezzetti, A. Angew. Chem., Int. Ed. 2011, 50,3072. |
| [62] | Smith, D.L.; Chidipudi, S.R.; Goundry, W.R.; Lam, H.W. Org. Lett. 2012, 14,4934. |
| [63] | Yuan, Y.; Bai, L.; Nan, J.; Liu, J.; Luan, X. Org. Lett. 2014, 16,4316. |
| [64] | Wang, X.-N.; Ma, Z.-X.; Deng, J.; Hsung, R.P. Tetrahedron Lett. 2015, 56,3463. |
| [65] | Chen, L.; Cao, J.; Xu, Z.; Zheng, Z.-J.; Cui, Y.-M.; Xu, L.-W. Chem. Commun. 2016, 52,9574. |
| [66] | Yang, Y.; Liu, H.; Peng, C.; Wu, J.; Zhang, J.; Qiao, Y.; Wang, X.-N.; Chang, J. Org. Lett. 2016, 18,5022. |
| [67] | Peng, C.; Zhang, J.; Xue, J.; Li, S.; Wang, X.-N.; Chang, J. J. Org. Chem. 2018, 83,9256. |
| [68] | Davies, P.W.; Cremonesi, A.; Dumitrescu, L. Angew. Chem., Int. Ed. 2011, 50,8931. |
| [69] | Mackay, W.D.; Fistikci, M.; Carris, R.M.; Johnson, J.S. Org. Lett. 2014, 16,1626. |
| [70] | (a) Zhou, A.-H.; He, Q.; Shu, C.; Yu, Y.-F.; Liu, S.; Zhao, T.; Zhang, W.; Lü, X.; Ye, L.-W. Chem. Sci. 2015, 6,1265. |
| [70] | (b) Li, X.-L.; Wang, J.-Q.; Li, L.; Yin, Y.-W.; Ye, L.-W. Acta Chim. Sinica 2016, 74,49. (in Chinese) |
| [70] | ( 李新玲, 王佳琪, 李龙, 尹应武, 叶龙武, 化学学报, 2016, 74,49.) |
| [71] | Yu, Y.; Chen, G.; Zhu, L.; Liao, Y.; Wu, Y.; Huang, X. J. Org. Chem. 2016, 81,8142. |
| [72] | Zhao, Y.; Hu, Y.; Wang, C.; Li, X.; Wan, B. J. Org. Chem. 2017, 82,3935. |
| [73] | Zhao, Y.; Hu, Y.; Li, X.; Wan, B. Org. Biomol. Chem. 2017, 15,3413. |
| [74] | Tian, X.; Song, L.; Han, C.; Zhang, C.; Wu, Y.; Rudolph, M.; Rominger, F.; Hashmi, A.S. K. Org. Lett. 2019, 21,2937. |
| [75] | Lin, P.-P.; Han, X.-L.; Ye, G.-H.; Li, J.-L.; Li, Q.; Wang, H. J. Org. Chem. 2019, 84,12966. |
| [76] | Mak, X.Y.; Crombie, A.L.; Danheiser, R.L. J. Org. Chem. 2011, 76,1852. |
| [77] | Pawar, S.K.; Vasu, D.; Liu, R.-S. Adv. Synth. Catal. 2014, 356,2411. |
| [78] | Duret, G.; Quinlan, R.; Martin, R.E.; Bisseret, P.; Neuburger, M.; Gandon, V.; Blanchard, N. Org. Lett. 2016, 18,1610. |
| [79] | Xue, J.; Gao, E.; Wang, X.-N.; Chang, J. Org. Lett. 2018, 20,6055. |
| [80] | Zhao, X.; Song, X.; Jin, H.; Zeng, Z.; Wang, Q.; Rudolph, M.; Rominger, F.; Hashmi, A.S. K. Adv. Synth. Catal. 2018, 360,2720. |
| [81] | Wu, H.; Liu, Y.; He, M.-X.; Wen, H.; Cao, W.; Chen, P.; Tang, Y. Org. Biomol. Chem. 2019, 17,8408. |
| [82] | Nissen, F.; Richard, V.; Alayrac, C.; Witulski, B. Chem. Commun. 2011, 47,6656. |
| [83] | Garcia, P.; Evanno, Y.; George, P.; Sevrin, M.; Ricci, G.; Malacria, M.; Aubert, C.; Gandon, V. Chem.-Eur. J. 2012, 18,4337. |
| [84] | Karad, S.N.; Liu, R.-S. Angew. Chem., Int. Ed. 2014, 53,9072. |
| [85] | Liang, H.; Bi, S.; Liu, Y.; Tang, Y.-N.; Liu, C. Org. Biomol. Chem. 2016, 14,2637. |
| [86] | Liu, D.; Nie, Q.; Cai, M. Tetrahedron 2018, 74,3020. |
| [87] | Chen, P.; Song, C.-X.; Wang, W.-S.; Yu, X.-L.; Tang, Y. RSC Adv. 2016, 6,80055. |
| [88] | Zhang, J.; Zhang, Q.; Xia, B.; Wu, J.; Wang, X.-N.; Chang, J. Org. Lett. 2016, 18,3390. |
| [89] | Wen, H.; Cao, W.; Liu, Y.; Wang, L.; Chen, P.; Tang, Y. J. Org. Chem. 2018, 83,13308. |
| [90] | Zhang, J.; Guo, M.; Chen, Y.; Zhang, S.; Wang, X.-N.; Chang, J. Org. Lett. 2019, 21,1331. |
| [91] | Dateer, R.B.; Pati, K.; Liu, R.-S. Chem. Commun. 2012, 48,7200. |
| [92] | Pawar, S.K.; Sahani, R.L.; Liu, R.-S. Chem.-Eur. J. 2015, 21,10843. |
| [93] | Jadhav, P.D.; Lu, X.; Liu, R.-S. ACS Catal. 2018, 8,9697. |
| [94] | Han, X.-L.; Liu, X.-G.; Lin, E.; Chen, Y.; Chen, Z.; Wang, H.; Li, Q. Chem. Commun. 2018, 54,11562. |
| [95] | Jin, H.; Rudolph, M.; Rominger, F.; Hashmi, A.S. K. ACS Catal. 2019, 9,11663. |
| [96] | Dekorver, K.A.; Hsung, R.P.; Lohse, A.G.; Zhang, Y. Org. Lett. 2010, 12,1840. |
| [97] | Brioche, J.; Meyer, C.; Cossy, J. Org. Lett. 2013, 15,1626. |
| [98] | Zhou, B.; Li, L.; Zhu, X.-Q.; Yan, J.-Z.; Guo, Y.-L.; Ye, L.-W. Angew. Chem., Int. Ed. 2017, 56,4015. |
| [99] | Baker, T.; Davies, P.W. Eur. J. Org. Chem. 2019,5201. |
| [100] | Wang, Z.-S.; Chen, Y.-B.; Zhang, H.-W.; Sun, Z.; Zhu, C.; Ye, L.-W. J. Am. Chem. Soc. 2020, 142,3636. |
/
| 〈 |
|
〉 |