

空气条件下温和高效催化N-吡啶吲哚啉的氧化脱氢
收稿日期: 2020-09-27
修回日期: 2020-11-02
网络出版日期: 2020-11-19
基金资助
宁波市自然科学基金(2019A610027)
A Mild and Efficient Catalytic Aerobic Oxidative Dehydrogenation of N-Pyridylindolines
Received date: 2020-09-27
Revised date: 2020-11-02
Online published: 2020-11-19
Supported by
Natural Science Foundation of Ningbo City(2019A610027)
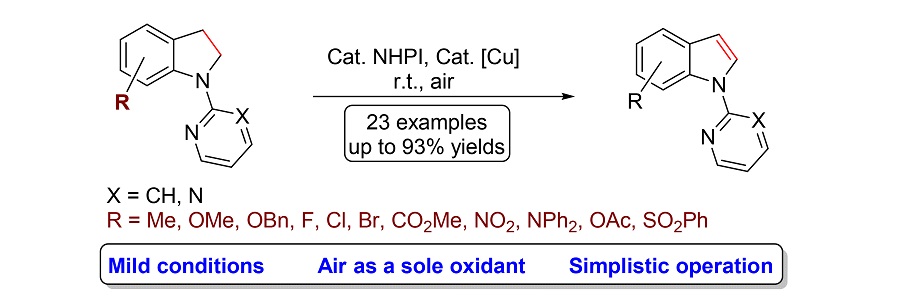
报道了空气条件下温和高效的N-羟基邻苯二甲酰亚胺和铜共同催化N-吡啶吲哚啉的氧化脱氢反应. 已知的N-吡啶吲哚啉的氧化过程只能在过量强氧化剂2,3-二氯-5,6-二氰对苯醌存在下实现, 该反应以空气作为单一的氧化剂, 具有绿色、安全、低成本以及操作简便等优点, 为N-吡啶吲哚啉类化合物氧化生成相应吲哚衍生物提供了一条新的绿色途径.
关键词: N-羟基邻苯二甲酰亚胺; 脱氢; N-吡啶吲哚啉; 空气条件; 氧化反应
唐灏 , 张贝贝 , 陈卫东 , 骆钧飞 . 空气条件下温和高效催化N-吡啶吲哚啉的氧化脱氢[J]. 有机化学, 2021 , 41(3) : 1131 -1137 . DOI: 10.6023/cjoc202009054
An efficient and mild N-hydroxyphthalimide and copper co-catalyzed aerobic oxidative dehydrogenation of N-pyridylindolines is developed. The use of air as sole oxidant allows a green, safe, low-cost and simple operation. This method provides an alternative to the known method for the oxidation of N-pyridylindoline derivatives by the use of excess stoichiometric strong oxidant 2,3-dichloro-5,6-dicyanobenzoquinone.

| [1] | (a) Bariwal, J.; Voskressensky, L. G.; Van der Eycken, E. V. Chem. Soc. Rev. 2018, 47, 3831. |
| [1] | (b) Xie, T.-Z.; Zhao, Y.-L.; Ma, W.-G.; Wang, Y.-F.; Yu, H.-F.; Wang, B.; Wei, X.; Huang, Z.-P.; Zhu, P.-F.; Liu, Y.-P.; Luo, X.-D. Chin. J. Org. Chem. 2020, 40, 679. (in Chinese) |
| [1] | (解天珍, 赵云丽, 马伟光, 王易芬, 于浩飞, 王蓓, 魏鑫, 黄之镨, 朱培凤, 刘亚平, 罗晓东, 有机化学, 2020, 40, 679.) |
| [1] | (c) Huang, J.; Luo, S.; Gong, L. Acta Chim. Sinica 2013, 71, 879. (in Chinese) |
| [1] | (黄建洲, 罗时玮, 龚流柱, 化学学报, 2013, 71, 879.) |
| [1] | (d) America, M.; Asgharia, A.; Amoozadeha, A.; Bakherad, M.; Chin. Chem. Lett. 2017, 28, 1031. |
| [1] | (e) Liang, K. J.; Xia, C. F. Chin. J. Chem. 2017, 35, 255. |
| [2] | (a) Bandini, M.; Eichholzer, A. Angew. Chem.. Int. Ed. 2009, 48, 9608. |
| [2] | (b) Barluenga, J.; Rodriguez, F.; Fananas, F. J. Chem.-Asian J. 2009, 4, 1036. |
| [2] | (c) Shi, Z.; Zhang, C.; Li, S.; Pan, D.; Ding, S.; Cui, Y.; Jiao, N. Angew. Chem.. Int. Ed. 2009, 48, 4572. |
| [2] | (d) Taber, D. F.; Tirunahari, P. K. Tetrahedron 2011, 67, 7195. |
| [2] | (e) Shi, Z.; Nie, K.; Liu, C.; Zhang, M.; Zhang, W. Chin. J. Org. Chem. 2020, 40, 327. (in Chinese) |
| [2] | (施展, 聂克睿, 刘畅, 张明智, 章维华, 有机化学, 2020, 40, 327.) |
| [2] | (f) Yu, K.; Gao, B.; Ding, H. Acta Chim. Sinica 2016, 74, 410. (in Chinese) |
| [2] | (余宽, 高北岭, 丁寒锋, 化学学报, 2016, 74, 410.) a94c3463-b409-4364-8925-1fbcb8fbd4c0 |
| [2] | (g) Huang, E.; Zhang, L., Xiao, C.; Meng, G.; Zhang, B.; Hu, J.; Wan, D. C.-C.; Meng, Q.; Jin, Z.; Hua, C. Chin. Chem. Lett., 2019, 30, 2157. |
| [2] | (h) Cerveri, A.; Bandini, M. Chin. J. Chem. 2020, 38, 287. |
| [2] | (i) Yang, P.; Xu, R.-Q.; Zheng, C.; You, S.-L. Chin. J. Chem. 2020, 38, 235. |
| [3] | (a) Thikekar, T. U.; Selvaraju, M.; Sun, C.-M. Org. Lett. 2016, 18, 316. |
| [3] | (b) Peng, F.; McLaughlin, M.; Liu, Y.; Mangion, I.; Tschaen, D. M.; Xu, Y. J. Org. Chem. 2016, 81, 10009. |
| [3] | (c) Zhang, J.; Chen, S.; Chen, F.; Xu, W.; Deng, G.-J.; Gong, H. Adv. Synth. Catal. 2017, 359, 2358. |
| [3] | (d) Huo, H.-R.; Tang, X.-Y.; Gong, Y.-F. Synthesis 2018, 50, 2727. |
| [3] | (e) Taboonpong, P.; Chavasiri, W. Catal. Commun. 2018, 104, 9. |
| [3] | (f) Zhu, R.; Zhai, M.; Liu, S.; Liu, X.; Wang, Z.; Ju, R.; Yu, X. Chin. J. Org. Chem. 2020, 40, 2045. (in Chinese) |
| [3] | (朱润宇, 翟民, 刘霜, 刘星彤, 王振, 居瑞军, 虞心红, 有机化学, 2020, 40, 2045.) |
| [4] | (a) Pratt, E. F.; McGovern, T. P. J. Org. Chem. 1964, 29, 1540. |
| [4] | (b) Kawase, M.; Miyake, Y.; Kikugawa, Y. J. Chem. Soc.. Perkin Trans. 1 1984,1401. |
| [4] | (c) Giethlen, B.; Schaus, J. M. Tetrahedron Lett. 1997, 38, 8483. |
| [4] | (d) Mukaiyama, T.; Kawana, A.; Fukuda, Y.; Matsuo, J. Chem. Lett. 2001,390. |
| [4] | (e) Tilstam, Kawana, U.; Harre, M.; Heckrodt, T.; Weinmann, H. Tetrahedron Lett. 2001, 42, 5385. |
| [5] | (a) Zhang, X.; Liu, W.; Sun, D.; Huang, J.; Qiu, X.; Li, Z.; Wu, Z. ChemSusChem 2020,4774. |
| [5] | (b) Chen, H.; Liu, L.; Huang, T.; Chen, J.; Chen, T. Adv. Synth. Catal. 2020,3332. |
| [5] | (c) Tang, H.; Luo, J.; Xie, P. Chin. J. Org. Chem. 2019, 39, 2735. (in Chinese) |
| [5] | (唐灏, 骆钧飞, 解攀, 有机化学, 2019, 39, 2735.) |
| [5] | (d) Zhang, Z.; Tian, Q.; Qian, J.; Liu, Q.; Liu, T.; Shi, L.; Zhang, G. J. Org. Chem. 2014, 79, 8182. |
| [5] | (e) Qian, J.; Zhang, Z.; Liu, Q.; Liu, T.; Zhang, G. Adv. Synth. Catal. 2014, 356, 3119. |
| [5] | (f) Zhang, Z.; Zhang, W.; Li, J.; Liu, Q.; Liu, T.; Zhang, G. J. Org. Chem. 2014, 79, 11226. |
| [6] | (a) Wang, Q.; Chai, H.; Yu, Z. Organometallics 2018, 37, 584. |
| [6] | (b) He, K. H.; Tan, F. F.; Zhou, C. Z.; Zhou, G.-J.; Yang, X.-L.; Li, Y. Angew. Chem.. Int. Ed. 2017, 56, 3080. |
| [7] | (a) Lou, C.; Qin, S.; Zhang, S.; Lv, Z.; Senan, A.; Chen, Z.; Yin, G. Catal. Commun. 2017, 90, 5. |
| [7] | (b) Hara, T.; Mori, K.; Mizugaki, T.; Ebitani, K.; Kaneda, K. Tetrahedron Lett. 2003, 44, 6207. |
| [8] | (a) Elumalai, K.; Weng, K. L. Tetrahedron Lett. 2018, 59, 3958. |
| [8] | (b) So, M. H.; Liu, Y.; Ho, C.-M.; Che, C.-M. Chem.-Asian J. 2009, 4, 1551. |
| [9] | (a) Zhou, W.; Chen, D.; Sun, F.; Qian, J.; He, M.; Chen, Q. Tetrahedron Lett. 2018, 59, 949. |
| [9] | (b) Iosub, A. V.; Stahl, S. S. Org. Lett. 2015, 17, 4404. |
| [10] | (a) Jawale, D. V.; Gravel, E.; Shah, N.; Dauvois, V.; Li, H.; Namboothiri, I. N. N.; Doris, E. Chem.-Eur. J. 2015, 21, 7039. |
| [10] | (b) Damodara, D.; Arundhathi, R.; Likhara, P. R. Adv. Synth. Catal. 2014, 356, 189. |
| [10] | (c) Zhou, W.; Taboonpong, P.; Aboo, A. H.; Zhang, L.; Jiang, J.; Xiao, J. Synlett 2016, 27, 1806. |
| [10] | (d) Cui, X.; Li, Y.; Bachmann, S.; Scalone, M.; Surkus, A.; Junge, K.; Topf, C.; Beller, M. J. Am. Chem. Soc. 2015, 137, 10652. |
| [10] | (e) Wendlandt, A. E.; Stahl, S. S. J. Am. Chem. Soc. 2014, 136, 506. |
| [10] | (f) Karki, M.; Araujo, H. C.; Magolan, J. Synlett 2013, 24, 1675. |
| [11] | (a) Kojima, M.; Kanai, M. Angew. Chem., Int. Ed. 2016, 55, 12224. |
| [11] | (b) Maier, A. F. G.; Tussing, S.; Schneider, T.; Florke, U.; Qu, Z.-W.; Grimme, S.; Paradies, J. Angew. Chem.. Int. Ed. 2016, 55, 12219. |
| [12] | Chen, W.; Tang, H.; Wang, W.; Fu, Q.; Luo, J. Adv. Synth. Catal. 2020, 362, 3905. |
| [13] | (a) Burke, J. P.; Bian, Z.; Shaw, S.; Zhao, B.; Goodwin, C. M.; Belmar, J.; Browning, C. F.; Vigil, D.; Friberg, A.; Camper, D. V.; Rossanese, O. W.; Lee, T.; Olejniczak, E. T.; Fesik, S. W. J. Med. Chem. 2015, 58, 3794. |
| [13] | (b) Ozawa, Y.; Kusano, K.; Owa, T.; Yokoi, A.; Asada, M.; Yoshimatsu, K. Cancer Chemother. Pharmacol. 2012, 69, 1353. |
| [13] | (c) Dydio, P.; Zielinski, T.; Jurczak, J. Chem. Commun. 2009,4560. |
| [13] | (d) Ferandin, Y.; Bettayeb, K.; Kritsanida, M.; Lozach, O.; Polychronopoulos, P.; Magiatis, P.; Skaltsounis, A.-L.; Meijer, L. J. Med. Chem. 2006, 49, 4638. |
| [14] | (a) Yang, X.-F.; Hu, X.-H.; Loh, T.-P. Org. Lett. 2015, 17, 1481. |
| [14] | (b) Shin, Y.; Sharma, S.; Mishra, N. K.; Han, S.; Park, J.; Oh, H.; Ha, J.; Yoo, H.; Jung, Y. H.; Kim, I. S. Adv. Synth. Catal. 2015, 357, 594. |
| [14] | (c) Mishra, N. K.; Jeong, T.; Sharma, S.; Shin, Y.; Han, S.; Park, J.; Oh, J. S.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. Adv. Synth. Catal. 2015, 357, 1293. |
| [14] | (d) Xie, W.; Li, B.; Wang, B. J. Org. Chem. 2015, 81, 396. |
| [14] | (e) Wang, X.; Tang, H.; Feng, H.; Li, Y.; Yang, Y.; Zhou, B. J. Org. Chem. 2015, 80, 6238. |
| [14] | (f) Hou, W.; Yang, Y.; Ai, W.; Wu, Y.; Wang, X.; Zhou, B.; Li, Y. Eur. J. Org. Chem. 2015,395. |
| [14] | (g) Park, J.; Mishra, N. K.; Sharma, S.; Han, S.; Shin, Y.; Jeong, T.; Oh, J. S.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. J. Org. Chem. 2015, 80, 1818. |
| [14] | (h) Yang, D.; Mao, S.; Gao, Y.-R.; Guo, D.-D.; Guo, S.-H.; Li, B.; Wang, Y.-Q. RSC Adv. 2015, 5, 23727. |
| [14] | (i) Kim, M.; Mishra, N. K.; Park, J.; Han, S.; Shin, Y.; Sharma, S.; Lee, Y.; Lee, E.-K.; Kwak, J. H.; Kim, I. S. Chem. Commun. 2014, 50, 14249. |
| [14] | (j) Pan, S.; Wakaki, T.; Ryu, N.; Shibata, T. Chem.-Asian J. 2014, 9, 1257. |
| [14] | (k) Urones, B.; Arrayas, R. G.; Carretero, J. C. Org. Lett. 2013, 15, 1120. |
| [15] | (a) Ahmad, A.; Dutta, H. S.; Kumar, M.; Khan, A. A.; Raziullah, Koley, D. Org. Lett. 2020, 22, 5870. |
| [15] | (b) Ahmad, A.; Dutta, H. S.; Khan, B.; Kant, R.; Koley, D. Adv. Synth. Catal. 2018, 360, 1644. |
| [15] | (c) Zhai, W.; Li, B.; Wang, B. ChemistrySelect 2018, 3, 8035. |
| [15] | (d) Ai, W.; Yang, X.; Wu, Y.; Wang, X.; Li, Y.; Yang, Y.; Zhou, B. Chem.-Eur. J. 2014, 20, 17653. |
| [15] | (e) Wu, Y.; Yang, Y.; Zhou, B.; Li, Y. J. Org. Chem. 2015, 80, 1946. |
| [16] | Kumar, M.; Sharma, R.; Raziullah, Khan, A. A.; Ahmad, A.; Dutta, H. S.; Koley, D. Org. Lett. 2020, 22, 2152. |
| [17] | Gao, S.; Xu, X.; Tang, H.; Wu, J.-Q.; Luo, J. ChemistrySelect 2019, 4, 5835. |
| [18] | Zhi, C.; Wang, Q.; Liu, S.; Xue, Y.; Shi, L.; Zhu, X.; Hao, X.-Q.; Song, M.-P. J. Org. Chem. 2020, 85, 1022. |
| [19] | (a) Lee, J. M.; Park, E. J.; Cho, S. H.; Chang, S. J. Am. Chem. Soc. 2008, 130, 7824. |
| [19] | (b) Ishii, Y.; Sakaguchi, S.; Iwahamab, T. Adv. Synth. Catal. 2001, 343, 393. |
| [19] | (c) Bag, R.; Sar, D.; Punniyamurthy, T. Org. Lett. 2015, 17, 2010. |
| [19] | (d) Zhou, W.-J.; Cao, G.-M.; Shen, G.; Zhu, X.-Y.; Gui, Y.-Y.; Ye, J.-H.; Sun, L.; Liao, L.-L.; Li, J.; Yu, D.-G. Angew. Chem.. Int. Ed. 2017, 56, 15683. |
| [19] | (e) Parasram, M.; Chuentragool, P.; Wang, Y.; Shi, Y.; Gevorgyan, V. J. Am. Chem. Soc. 2017, 139, 14857. |
| [20] | Ackermann, L.; Lygin, A. V. Org. Lett. 2011, 13, 3332. |
| [21] | Xu, S.; Huang, X.; Hong, X.; Xu, B. Org. Lett. 2012, 14, 4614. |
| [22] | Liang, L.; Fu, S.; Lin, D.; Zhang, X.-Q.; Deng, Y.; Jiang, H.; Zeng, W. J. Org. Chem. 2014, 79, 9472. |
| [23] | Li, T.; Wang, Z.; Zhang, M.; Zhang, H.-J.; Wen, T.-B. Chem. Commun. 2015, 51, 6777. |
| [24] | Liu, S.-L.; Li, Y.; Guo, J.-R.; Yang, G.-C.; Li, X.-H.; Gong, J.-F.; Song, M.-P. Org. Lett. 2017, 19, 4042. |
| [25] | Manna, M. K.; Bairy, G.; Jana, R. J. Org. Chem. 2018, 83, 8390. |
| [26] | Rull, S. G.; Blandez, J. F.; Fructos, M. R.; Belderrain, T. R.; Nicasio, M. C. Adv. Syn. Catal. 2015, 357, 907. |
| [27] | Gao, S.; Xu, X.; Tang, H.; Wu, J.-Q.; Luo, J. ChemistrySelect 2019, 4, 5835. |
/
| 〈 |
|
〉 |