

酒石酸催化四组分反应合成双螺环四氢喹啉双(1,3-二噁烷-4,6-二酮)衍生物
收稿日期: 2020-08-27
修回日期: 2020-12-13
网络出版日期: 2020-12-31
基金资助
江西省研究生创新基金(YC2015-B023); 江西省教育厅科技计划研究项目(GJJ170170); 江西省教育厅科技计划研究项目(GJJ180892); 江西师范大学博士科研启动基金(JXSDBY2020-B012)
A Four-Component Reaction for the Synthesis of Dispirotetrahydro-quinoline-bis(1,3-dioxane-4,6-dione) Derivatives Catalyzed by Tartaric Acid
Received date: 2020-08-27
Revised date: 2020-12-13
Online published: 2020-12-31
Supported by
Graduate Innovation Foundation of Jiangxi Province(YC2015-B023); Science and Technology Research Project of Jiangxi Provincial Education Department(GJJ170170); Science and Technology Research Project of Jiangxi Provincial Education Department(GJJ180892); Jiangxi Normal University Doctoral Research Initiation Fund Project(JXSDBY2020-B012)
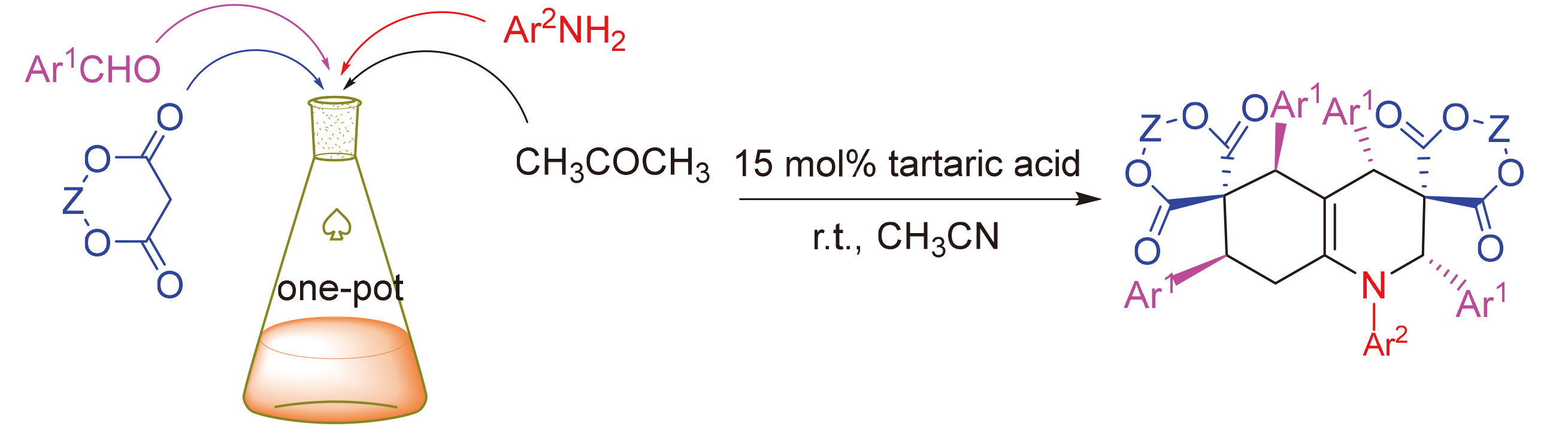
在酒石酸催化作用下, 以1,3-二噁烷-4,6-二酮、芳香醛、芳香胺和丙酮为原料, 通过四组分反应有效地合成了11种双螺环四氢喹啉双(1,3-二噁烷-4,6-二酮)衍生物. 该方法具有产物收率高、适用范围较广、反应温和、操作简单及环境友好等优点, 同时对反应机理进行了推测.
许招会 , 叶华涛 , 张文峰 , 肖强 . 酒石酸催化四组分反应合成双螺环四氢喹啉双(1,3-二噁烷-4,6-二酮)衍生物[J]. 有机化学, 2021 , 41(5) : 2127 -2133 . DOI: 10.6023/cjoc202008051
Eleven dispirotetrahydroquinoline-bis(1,3-dioxane-4,6-dione) derivatives were synthesized by a four-component reaction of 1,3-dioxane-4,6-dione, aromatic aldehydes, arylamines and acetone in the presence of tartaric acid. This method can provide the advantages of high yields, broader substrate scope, mild conditions, simple operation and environmental friendliness. Additionally, the reaction mechanism was also speculated.

| [1] | Michael, J. P. Nat. Prod. Rep. 1999, 16, 697. |
| [2] | Fokialakis, N.; Magiatis, P.; Chinou, I.; Mitaku, S.; Tillequin, F. Chem. Pharm. Bull. 2002, 50, 413. |
| [3] | Schiff Jr, J. P. Am. J. Pharm. Educ. 2006, 70, 98. |
| [4] | Hamon, M.; Mallat, M.; Herbet, A.; Nelson, D. L.; Audinot, M.; Pichat, L.; Glowinski, J. J. Neurochem. 1981, 36, 613. |
| [5] | Miller, K. J.; King, A.; Demchyshyn, L.; Niznik, H.; Teitler, M. Eur. J. Pharmacol. 1992, 227, 99. |
| [6] | Hirasawa, Y.; Kobayashi, J. I.; Morita, H. Org. Lett. 2006, 8, 123. |
| [7] | Bredberg, U.; Eyjolfsdottir, G. S.; Paalzow, L.; Tfelt-Hansen, P.; Tfelt-Hansen, V. Eur. J. Clin. Pharmacol. 1986, 30, 75. |
| [8] | Nishiyama, T. T.; Hashiguchi, Y. Y.; Sakata, S. T.; Sakaguchi, T. T. Polym. Degrad. Stab. 2003, 79, 225. |
| [9] | Chande, S. M.; Khanwelkar, R. R. Tetrahedron Lett. 2005, 46, 7787. |
| [10] | Ivanov, A. S. Chem. Soc. Rev. 2008, 37, 789. |
| [11] | Pizzirani, D.; Roberti, M.; Recanatini, M. Tetrahedron Lett. 2007, 48, 7120. |
| [12] | Pizzirani, D.; Roberti, M.; Grimaudo, S.; Di Cristina, A.; Pipitone, R. M.; Tolomeo, M.; Recanatini, M. J. Med. Chem. 2009, 52, 6936. |
| [13] | Lashkari, M.; Maghsoodlou, M. T.; Hazeri, N.; Habibi-Khorassani, S. M.; Akbarzadeh-Torbati, N.; García-Granda, S.; Torre-Fernán- dez, L. J. Heterocycl. Chem. 2015, 52, 873. |
| [14] | Salahi, S.; Hazeri, N.; Maghsoodlou, M. T.; García-Granda, S.; Torre-Fernández, L. J. Chem. Res. 2014, 38, 383. |
| [15] | Hazeri, N.; Lashkari, M.; García-Granda, S.; Torre-Fernández, L. Aust. J. Chem. 2014, 67, 1656. |
| [16] | Salahi, S.; Maghsoodlou, M. T.; Hazeri, N.; Lashkari, M.; Garcia-Granda, S.; Torre-Fernandez, L. Chin. J. Catal. 2015, 36, 1023. |
| [17] | Salahi, S.; Hazeri, N.; Maghsoodlou, M. T.; Lashkari, M.; Torbati, N. A.; Garcia-Granda, S.; Torre-Fernandez, L. J. Chil. Chem. Soc. 2018, 63, 4159. |
| [18] | Fatahpour, M.; Lashkari, M.; Hazeri, N.; Sadeh, F. N.; Maghsoodlou, M. T. Org. Prep. Proced. Int. 2019, 51, 576. |
| [19] | Salahi, S.; Maghsoodlou, M. T.; Hazeri, N.; Lashkari, M.; Torbati, N. A.; Kazemian, M.A. García-Granda, S.; Torre-Fernández, L. J. Saudi Chem. Soc. 2016, 20, 349. |
| [20] | Zhou, Q.; Wang, Q. A.; Jia, C. H. Food Res. Dev. 2010, 31, 26. (in Chinese). |
| [20] | (周强, 汪秋安, 贾超华, 食品研究与开发, 2010, 31, 26.) |
| [21] | Fan, N. L.; Chen, F. B.; Xu, Z. H.; Xiao, Q. Chin. J. Synth. Chem. 2018, 26, 479. (in Chinese). |
| [21] | (范乃立, 陈飞彪, 许招会, 肖强, 合成化学, 2018, 26, 479.) |
| [22] | Xu, Z. H.; Li, Y. J.; Liu, D. Y.; Xiao, Q. Chin. J. Org. Chem. 2018, 38, 3118. (in Chinese). |
| [22] | (许招会, 李瑜钰, 刘德永, 肖强, 有机化学, 2018, 38, 3118.) |
| [23] | Kodama, T.; Moquist, P. N.; Schaus, S. E. Org. Lett. 2011, 13, 6316. |
| [24] | Lu, N.; Chen, D. Z.; Zhang, G. Q. Int. J. Quantum. Chem. 2011, 111, 2031. |
| [25] | Khandebharad, A. U.; Sarda, S. R.; Gill, C. H. Res. J. Chem. Sci. 2015, 5, 27. |
| [26] | Aboonajmi, J.; Maghsoodou, M. T.; Hazeri, N. Res. Chem. Intermed. 2015, 41, 8057. |
| [27] | Nidhi, G.; Virendrak, K. T. Synth. Commun. 2011, 41, 2800. |
| [28] | Niralwad, K. S.; Shingate, B. B.; Shingare, M. S. J. Korean Chem. Soc. 2011, 55, 486. |
| [29] | Yan, N.; Xiong, B.; Liao, W. L.; Xu, Z. H. Chin. J. Org. Chem. 2010, 30, 1391. (in Chinese). |
| [29] | (严楠, 熊斌, 廖维林, 许招会, 有机化学, 2010, 30, 1391.) |
| [30] | Zeng, W. L.; Cai, X.; Guo, H. M. Chin. J. Struct. Chem. 2013, 32, 1603. |
| [31] | Fan, N. L.; Chen, F. B.; Xu, Z. H.; Xiao, Q. Chin. J. Synth. Chem. 2017, 26, 479. (in Chinese). |
| [31] | (范乃立, 陈飞彪, 许招会, 肖强, 合成化学, 2017, 26, 479.) |
/
| 〈 |
|
〉 |