

SnCl2@MNPs催化Biginelli反应一锅法合成3,4-二氢嘧啶-2-酮衍生物
收稿日期: 2021-01-08
修回日期: 2021-02-04
网络出版日期: 2021-04-16
基金资助
国家自然科学基金(21978154); 山东省自然科学基金(ZR2020QB191); 曲阜师范大学国家级大学生创新创业训练计划(201910446018)
One-Pot Synthesis of 3,4-Dihydropyrimidine-2-one Derivatives via Biginelli Reactions Catalyzed by SnCl2@MNPs
Received date: 2021-01-08
Revised date: 2021-02-04
Online published: 2021-04-16
Supported by
National Natural Science Foundation of China(21978154); Natural Science Foundation of Shandong Province(ZR2020QB191); National Students' Platform for Innovation and Entrepreneurship Training Programs(201910446018)
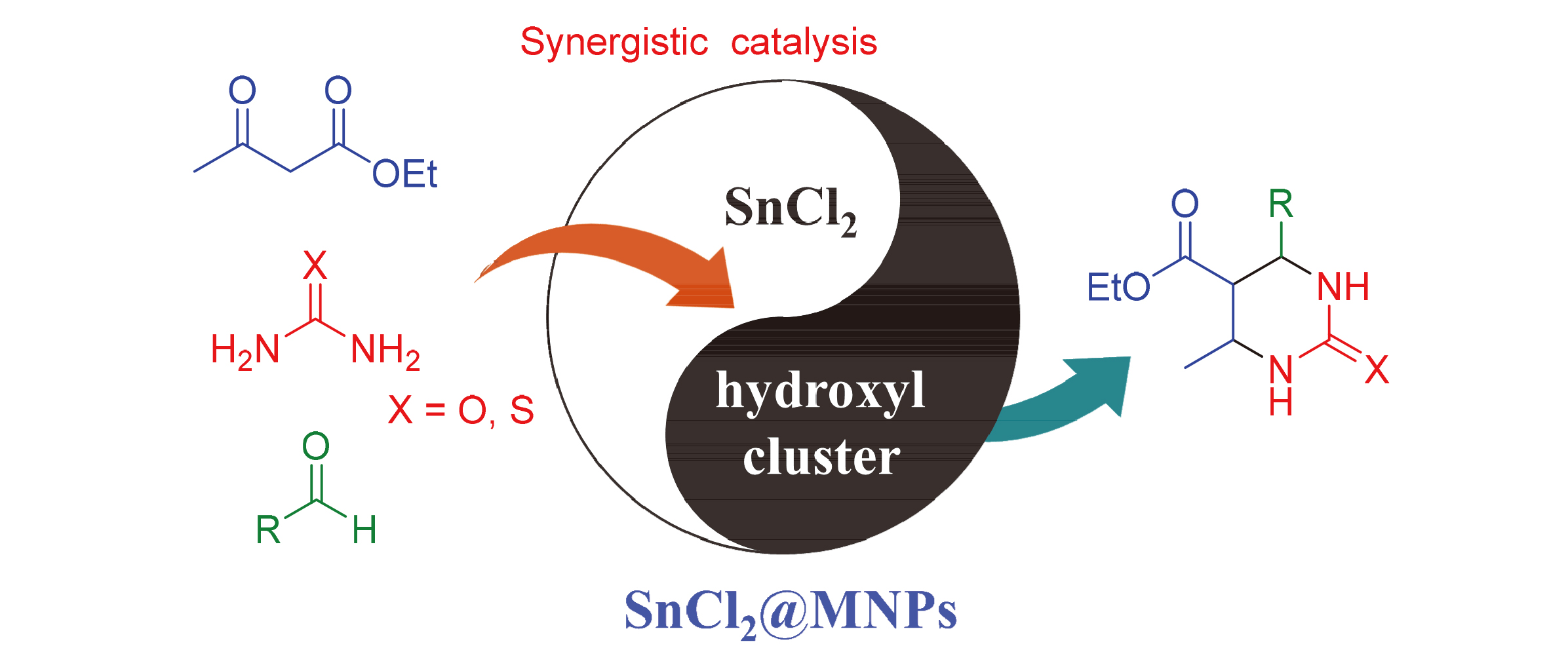
采用化学共沉淀法制备磁性Fe3O4纳米颗粒, 以磁性Fe3O4纳米颗粒为核, 通过碱催化正硅酸四乙酯水解法制备了具有核壳结构的磁性纳米颗粒(MNPs), 然后再利用化学共沉淀法制备了MNPs负载SnCl2催化剂(SnCl2@MNPs). 采用多种现代分析手段表征SnCl2@MNPs的微观结构及其组成, 结果发现, SnCl2均匀分散于MNPs表面, 且与MNPs之间形成较强的作用力. 通过SnCl2@MNPs催化Biginelli反应来探究其催化性能, 结果发现, SnCl2@MNPs能高效催化芳香醛或杂环醛、尿素或硫脲与α,β-二羰基化合物的三组分一锅法合成3,4-二氢嘧啶-2-酮衍生物, 且产率高达78%~96%. 由于SnCl2@MNPs中活性组分SnCl2在载体表面呈微粒形式存在, 从而增大了其与反应物的接触面积, 其高的催化活性归结于高度分散的SnCl2与载体表面丰富的羟基簇的协同作用. 此外, SnCl2@MNPs还具有强磁回收的特点, 该催化剂在磁力回收循环7次后, 仍然保持优异的催化性能.
关键词: 磁纳米颗粒; Biginelli反应; 芳香醛; α,β-二羰基化合物
赵雯辛 , 黄孟君 , 李胜男 , 刘玉静 , 刘中秋 , 应安国 . SnCl2@MNPs催化Biginelli反应一锅法合成3,4-二氢嘧啶-2-酮衍生物[J]. 有机化学, 2021 , 41(7) : 2743 -2749 . DOI: 10.6023/cjoc202101015
The magnetic Fe3O4 nanoparticles were prepared by a co-precipitation method. The magnetic nanoparticles (MNPs) with core-shell structure were constructed by alkali-catalyzed hydrolysis of tetraethyl orthosilicate, and SnCl2 grafted on MNPs (SnCl2@MNPs) was successfully synthesized by a co-precipitation method. Then the catalyst of SnCl2@MNPs was further analyzed by a series of modern characterization methods for exploring the microstructure and composition. The results showed that SnCl2 was uniformly dispersed on the surfaces of MNPs and there were strong interaction forces between SnCl2 and MNPs. Subsequently, the Biginelli reaction was performed to investigate the catalyst performance, the results showed that 3,4-dihydropyrimidin-2(1H)-ones were efficiently synthesized by a three component one-pot reactions of aromatic aldehydes or heterocyclic aldehydes, carbamide or thiourea, andα,β-dicarbonyl compounds with high yields of 78%~96%. The active fraction of SnCl2 in SnCl2@MNPs is uniformly distributed on the surface of MNPs and in the form of microcrystalline that increases the effective contact area with substrates. The highly activity of SnCl2@MNPs is ascribed to the synergistic effect of SnCl2 and the abundant hydroxyl cluster in MNPs. In addition, the catalyst can be recovered using an external magnetic field and reused at least 7 times without a considerable decrease in its catalytic activity.

| [1] | Hela, S.; Younes, M.; Ridhaben, S. Arab. J. Chem. 2016, 9,S510. |
| [2] | Zhang,Y. H.; Wang, B.; Zhang,X. M.; Huang,J. B.; Liu,C. J. Molecules 2015, 20,3811. |
| [3] | Kuraitheerthakumaran, A.; Pazhamalai, S.; Gopalakrishnan, M. Arab. J. Chem. 2016, 9,S461. |
| [4] | Biginelli, P. Gazz. Chim. Ital. 1893, 23,360. |
| [5] | Abdolkarim, Z.; Zahra, N. J. Mol. Liq. 2016, 216,364. |
| [6] | Lu, J.; Bai,Y. J.; Wang,Z. J.; Yang,B. Q.; Ma,H. R. Tetrahedron Lett. 2000, 41,9075. |
| [7] | Wang,C. F.; Jiang, H.; Gong, H.; Wang,Z. C. Chin. J. Org. Chem. 2004, 24,333 (in Chinese). |
| [7] | ( 王彩凤, 姜恒, 宫红, 王敏, 王之昌, 有机化学, 2004, 24,333.) |
| [8] | Pravinsing,S. G.; Bhikan,J. K.; Deepak,V. N.; Bhata,R. C. Pharma Chem. 2015, 7,241. |
| [9] | Li,S. N.; Zhao,W. X.; Liu,Y. J.; Liu,Z. Q.; Ying,A. G. Chin. J. Org. Chem. 2020, 40,1835 (in Chinese). |
| [9] | ( 李胜男, 赵雯辛, 刘玉静, 刘中秋, 应安国, 有机化学, 2020, 40,1835.) |
| [10] | Ying,A. G.; Li,S. N.; Liu,X. Q.; Wang, J.; Liu,Y. J.; Liu,Z. Q. J. Catal. 2020, 391,312. |
| [11] | Lu, J.; Ma,H. R.; Li,W. H. Chin. J. Org. Chem. 2000, 20,815 (in Chinese). |
| [11] | ( 路军, 马怀让, 李万华, 有机化学, 2000, 20,815.) |
| [12] | Wang,J. C.; Han,H. Q. Polyurethane Ind. 2005, 20,11 (in Chinese). |
| [12] | ( 王景存, 韩怀强 聚氨酯工业, 2005, 20,11.) |
| [13] | Lu, J.; Wang, F.; Bai, Y.; Li,W. H. Chin. J. Org. Chem. 2002, 22,788 (in Chinese). |
| [13] | ( 路军, 王飞利, 白银娟, 李万华, 有机化学, 2002, 22,788.) |
| [14] | Ma, Y.; Qian, C.; Wang, L.; Yang, M. J. Org. Chem. 2000, 65,3864. |
| [15] | Song,X. Y. The Brochure of Polyurethane Elastomer. Chemical Industry Press, Beijing, 2001, pp.652-653 (in Chinese). |
| [15] | ( 宋向雁, 聚氨酯弹性体手册. 化学工业出版社, 北京, 2001, pp.652-653.) |
| [16] | Wang, M.; Song,J. L.; Liu, Y. Chem. Res. Appl. 2013, 25,1717. |
| [17] | Tao, G.; Xu,Y. H.; Zhu, B. Doctoral Expert Forum. 2013, 8,408 (in Chinese). |
| [17] | ( 陶果, 徐友辉, 朱斌, 博士专家论坛., 2013, 8,408.) |
| [18] | Zheng,X. D. J. Baoji Univ. Arts Sci. (Nat. Sci. Ed.)2004, 24,194 (in Chinese). |
| [18] | ( 郑旭东, 宝鸡文理学院学报(自然科学版), 2004, 24,194.) |
| [19] | Wang, X.; Dou,P. P.; Zhao, P.; Yi, D.; Ping, X. ChemSusChem 2009, 2,947. |
| [20] | Hasaninejad, A.; Zare, A.; Jafari, F.; Moosavi-Zare,A. R. Eur.-J. Chem. 2009, 6,459. |
| [21] | Tayebee, R.; Amini,M. M.; Ghadamgahi, M.; Armaghan, M. J. Mol. Catal. A:Chem. 2013, 366,266. |
| [22] | Ranu,B. C.; Hajra, A.; Jana, U. J. Org. Chem. 2000, 65,6270. |
| [23] | Paraskar,A. S.; Dewkar,G. K.; Sudalai, A. Tetrahedron Lett. 2003, 44,3305. |
| [24] | Hu,E. H.; Sidler,D. R.; Dolling,U. H. J. Org. Chem. 1998, 63,3454. |
| [25] | Wang, R.; Liu,Z. Q. J. Org. Chem. 2012, 77,3952. |
/
| 〈 |
|
〉 |