

高效合成含P—O—P键化合物: 焦膦酸酯、四烷基焦膦酸酯和焦膦酰胺的方法
收稿日期: 2022-02-07
修回日期: 2022-03-05
网络出版日期: 2022-03-30
基金资助
国民核生化灾害防护国家重点实验室(SKLNBC2021-09)
Development of an Efficient Method for the Synthesis of Compounds Containing P—O—P Bonds: Pyrophosphonates, Tetraalkyl Pyrophosphates and Pyrophosphonamidates
Received date: 2022-02-07
Revised date: 2022-03-05
Online published: 2022-03-30
Supported by
State Key Laboratory of Nuclear Biological and Chemical Protection for Civilian(SKLNBC2021-09)
张子璇 , 高润利 , 胡慧娟 , 卢晓刚 , 王瑾陈霄 , 王红梅 . 高效合成含P—O—P键化合物: 焦膦酸酯、四烷基焦膦酸酯和焦膦酰胺的方法[J]. 有机化学, 2022 , 42(7) : 2214 -2221 . DOI: 10.6023/cjoc202201050
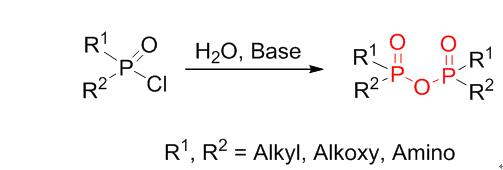
This work describes a method of synthesizing pyrophosphonates, tetraalkyl pyrophosphates and pyrophosphonamidates. Pyrophosphonates can be synthesized by the reaction of alkyl dichlorophosphonate, triethylamine (Et3N), and water, in a molar ratio of 1∶1.2∶0.5 (dichlorophosphonate∶Et3N∶water). When the reaction was allowed to proceed for 15 min in acetone, the maximum yield recorded for the pyrophosphonates was 95%. Et3N was the solvent, with an acid-binding agent. Following this method, the achieved rate of production of tetraalkyl pyrophosphate and pyrophosphonamidate was faster than the rate of production of the compounds reached using traditional methods.
| [1] | (a) Barpanda, P.; Nishimura, S.-I.; Yamada, A. Adv. Energy Mater. 2012, 2, 841. |
| [1] | (b) Ikotun, O. F.; Marino, N.; Kruger, P. E.; Julve, M.; Doyle, R. P. Coord. Chem. Rev. 2010, 254, 890. |
| [1] | (c) Jordan, F. Nat. Prod. Rep. 2003, 20, 184. |
| [1] | (d) Kim, S. K.; Lee, D. H.; Hong, J.-I.; Yoon, J. Acc. Chem. Res. 2009, 42, 23. |
| [1] | (e) Orriss, I. R.; Arnett, T. R.; Russell, R. G. G. Curr. Opin. Pharmacol. 2016, 28, 57. |
| [2] | (a) Lin, H. Org. Biomol. Chem. 2007, 5, 2541. |
| [2] | (b) Pehar, M.; Harlan, B. A.; Killoy, K. M.; Vargas, M. R. Antioxid. Redox Signaling 2018, 28, 1652. |
| [2] | (c) Davila, A.; Liu, L.; Chellappa, K.; Redpath, P.; Nakamaru-Ogiso, E.; Paolella, L. M.; Zhang, Z.; Migaud, M. E.; Rabinowitz, J. D.; Baur, J. A. Elife 2018, 7, 1. |
| [3] | (a) McPhillips, J. J.; Coon, J. M. Toxicol. Appl. Pharmacol. 1966, 8, 66. |
| [3] | (b) Rider, J. A.; Ellinwood, L. E.; Coon, J. M. Proc. Soc. Exp. Biol. Med. 1952, 81, 455. |
| [3] | (c) Rider, J. A.; Schulman, S.; Richter, R. B.; Moeller, H. C.; DuBois, K. P. J. Am. Med. Assoc. 1951, 145, 967. |
| [4] | (a) Kimmerle, G.; Klimmer, O. R. Arch. Toxicol. 1974, 33, 1. |
| [4] | (b) Wood, S. J.; Osborne, R. H. Pestic. Sci. 1991, 32, 485. |
| [5] | (a) Wolf, R. Ind. Eng. Chem. Prod. Res. Dev. 1981, 20, 413. |
| [5] | (b) Wolf, R. Kunststoffe 1986, 76, 943. |
| [6] | (a) D'Agostino, P. A.; Provost, L. R. J. Chromatogr. 1992, 589, 287. |
| [6] | (b) D'Agostino, P. A.; Hancock, J. R.; Chenier, C. L. Eur. J. Mass Spectrom. 2003, 9, 609. |
| [6] | (c) Terent'ev, A. G.; Morozik, Y. I.; Ivanova, M. V.; Dudkin, A. V. J. Anal. Chem. 2020, 75, 208 |
| [7] | (a) Ahmadibeni, Y.; Parang, K. J. Org. Chem. 2006, 71, 5837. |
| [7] | (b) Dash, C.; Ahmadibeni, Y.; Hanley, M. J.; Pandhare, J.; Gotte, M.; Le Grice, S. F. J.; Parang, K. Bioorg. Med. Chem. Lett. 2011, 21, 3519. |
| [7] | (c) Gaines, J. C.; Ivy, E. E.; Dean, H. A.; Scales, A. L. J. Econ. Entomol. 1950, 43, 614. |
| [7] | (d) Kiran, Y. B.; Reddy, C. D.; Gunasekar, D.; Raju, C. N.; Barbosa, L. C. A.; Marney, D. C. O.; Russell, L. J. J. Fire Sci. 2007, 25, 193. |
| [7] | (e) Davidson, A.; Foley, D. A.; Frericks-Schmidt, H.; Ruggeri, S. G.; Herman, M.; LaCasse, S.; Liu, Y.; McInturff, E. L.; Morris, R.; Mugheirbi, N.; Samas, B.; Sarkar, A.; Singer, R. A.; Witkos, F.; Yu, S. Org. Process Res. Dev. 2021, 25, 621. |
| [8] | (a) Parakin, O. V.; Romakhin, A. S.; Nikitin, E. V.; Ignat'ev, Y. A.; Romanov, G. V.; Mironov, B. S.; Kargin, Y. M.; Pudovik, A. N. Zh. Obshch. Khim. 1985, 55, 2621. |
| [8] | (b) Pavlichenko, V. F.; Presnov, A. E.; Tomilov, A. P. Zh. Obshch. Khim. 1995, 65, 1347. |
| [9] | (a) Kumar, R.; Pardasani, D.; Mazumder, A.; Dubey, D. K.; Gupta, A. K. Aust. J. Chem. 2008, 61, 476. |
| [9] | (b) Kumar, R.; Gupta, A. K.; Kaushik, M. P. Phosphorus, Sulfur Silicon Relat. Elem. 2010, 185, 765. |
| [10] | (a) Toy, A. D. F. J. Am. Chem. Soc. 1948, 70, 3882. |
| [10] | (b) Toy, A. D. F. J. Am. Chem. Soc. 1950, 72, 2065. |
| [11] | Zhou, Y.; Yin, S.; Gao, Y.; Zhao, Y.; Goto, M.; Han, L.-B. Angew. Chem., Int. Ed. 2010, 49, 6852. |
| [12] | Huang, J.; He, W.; Wang, B. Phosphorus, Sulfur Silicon Relat. Elem. 2012, 187, 1125. |
| [13] | Kins, C. F.; Brunklaus, G.; Spiess, H. W. Macromolecules 2013, 46, 2067. |
| [14] | Dunning, T. H. J. Chem. Phys. 1989, 90, 1007. |
/
| 〈 |
|
〉 |