

氰甲基导向的吲哚选择性C—H烯基化
收稿日期: 2022-03-06
修回日期: 2022-03-22
网络出版日期: 2022-03-30
基金资助
国家自然科学基金(22022204); 国家自然科学基金(21633013); 国家自然科学基金(22072167)
Ru(II)-Catalyzed Regioselective C—H Alkenylation of Indoles Using Cyanomethyl Directing Group
Received date: 2022-03-06
Revised date: 2022-03-22
Online published: 2022-03-30
Supported by
the National Natural Science Foundation of China(22022204); the National Natural Science Foundation of China(21633013); the National Natural Science Foundation of China(22072167)
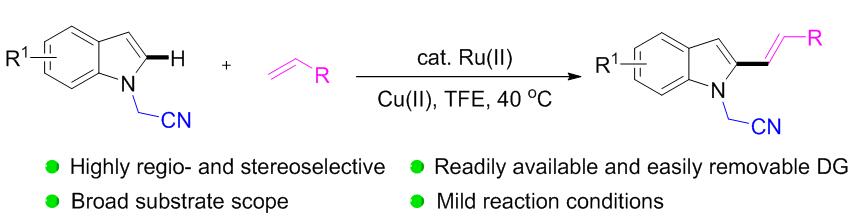
首次利用氰甲基作为导向基团, 在钌(II)催化下实现了吲哚及其衍生物C(2)位C—H的选择性烯基化反应, 制备得到了一系列具有潜在应用价值的吲哚C(2)-烯基化产物. 该反应条件温和, 并表现出较好的底物普适性和官能团兼容性, 酯基、氰基、碘、溴、氟和三氟甲基等均被较好地兼容. 该导向基团可以在相对温和的条件下方便地移除, 生成 N—H吲哚类化合物.
关键词: 钌催化; 选择性C(2)烯基化; 吲哚; 氰甲基导向; C—H官能团化
闫法超 , 李洋 , 李玉东 , Mohamed Makha , 李跃辉 . 氰甲基导向的吲哚选择性C—H烯基化[J]. 有机化学, 2022 , 42(7) : 2192 -2200 . DOI: 10.6023/cjoc202203017
A ruthenium-catalyzed C(2)-alkenylation of indole derivatives by employing cyanomethyl as the directing group was developed for the first time. Site-selective alkenylation was achieved for a broad scope of alkenes as coupling partners with a large number of indoles bearing synthetically useful functional groups. The protocol represents a novel method for C(2)-alkenylation of indoles affording biologically relevant indolic compounds. The reaction conditions were mild and it showed good substrate scope and functional group compatibility. Ester group, cyano group, iodine, bromine, fluorine and trifluoromethyl were all well compatible. The directing group can be easily removed under relatively mild conditions to form N—H indoles.

| [1] | (a) Zhang, M.-Z.; Chen, Q.; Yang, G.-F. Eur. J. Med. Chem. 2015, 89, 421. |
| [1] | (b) Somei, M.; Yamada, F. Nat. Prod. Rep. 2005, 22, 73. |
| [1] | (c) Kochanowska-Karamyan, A. J.; Hamann, M. T. Chem. Rev. 2010, 110, 4489. |
| [1] | (d) Kaushik, N. K.; Kaushik, N.; Attri, P.; Kumar, N.; Kim, C. H.; Verma, A. K.; Choi, E. H. Molecules 2013, 18, 6620. |
| [1] | (e) Liu, X.-Y.; Qin, Y. Green Synth. Catal. 2022, 3, 25. |
| [2] | (a) Xie, W.; Zuo, Z.; Zi, W.; Ma, D. Chin. J. Org. Chem. 2013, 33, 869. (in Chinese) |
| [2] | ( 谢卫青, 左智伟, 资伟伟, 马大为, 有机化学, 2013, 33, 869.) |
| [2] | (b) Zhang, D.; Qin, Y. Acta Chim. Sinica 2013, 71, 147. (in Chinese) |
| [2] | ( 张丹, 秦勇, 化学学报, 2013, 71, 147.) |
| [2] | (c) Wang, W.; Zhang, M.; Yang, W.; Yang, X. Chin. J. Org. Chem. 2022, 42, 75. (in Chinese) |
| [2] | ( 王弯弯, 张明明, 杨文超, 杨小虎, 有机化学, Chin. J. Org. Chem. 2022, 42, 75.) |
| [2] | (d) Hegedus, L. S. Angew. Chem., Int. Ed. Engl. 1988, 27, 1113. |
| [2] | (e) Cacchi, S.; Fabrizi, G. Chem. Rev. 2005, 105, 2873. |
| [2] | (f) Ackermann, L. Synlett 2007, 4, 507. |
| [2] | (g) Ma, W.; Gandeepan, P.; Li, J.; Ackermann, L. Org. Chem. Front. 2017, 4, 1435. |
| [2] | (h) Kumar, P.; Nagtilak, P. J.; Kapur, M. New J. Chem. 2021, 45, 13692. |
| [2] | (i) Rago, A. J.; Dong, G. Green Synth. Catal. 2021, 2, 216. |
| [2] | (j) Cerveria, A.; Bandini, M. Chin. J. Chem. 2020, 38, 287. |
| [2] | (k) Yang, P.; Xu, R.-Q.; Zheng, C.; You, S.-L. Chin. J. Chem. 2020, 38, 235. |
| [2] | (l) Sun, Y.; Wang, Z.; Wu, S.; Zhang, Y.; Shi, F. Green Synth. Catal. 2022, 3, 84. |
| [2] | (m) Li, R.; Jiang, S.; Zheng, H.; Lei, H.; Huang, Z.; Chen, S.; Deng, G.-J. Green Synth. Catal. 2022, 3, 95. |
| [3] | (a) Moritani, I.; Fujiwara, Y. Tetrahedron Lett. 1967, 8, 1119. |
| [3] | (b) Danno, S.; Asano, R.; Teranishi, S. J. Am. Chem. Soc. 1969, 91, 7166. |
| [3] | (c) Jia, C.; Kitamura, T.; Fujiwara, Y. Acc. Chem. Res. 2001, 34, 633. |
| [3] | (d) Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318. |
| [3] | (e) Wang, Z.-T.; Zhang, Y.-S.; Wang, S.-C.; Xia, D.-H. Chin. J. Org. Chem. 2007, 27, 143. (in Chinese) |
| [3] | ( 王宗廷, 张云山, 王书超, 夏道宏, 有机化学, 2007, 27, 143.) |
| [4] | (a) Gai, S.; Zhang, Q.; Hu, X. J. Org. Chem. 2014, 79, 2111. |
| [4] | (b) Basceken, S.; Kaya, S.; Balci, M. J. Org. Chem. 2015, 80, 12552. |
| [4] | (c) Yan, J.; Ni, T.; Yan, F. Tetrahedron Lett. 2015, 56, 1096. |
| [5] | (a) Lyons, T. W.; Sanford, M. S. Chem. Rev. 2010, 110, 1147. |
| [5] | (b) Rousseau, G.; Breit, B. Angew. Chem., Int. Ed. 2011, 50, 2450. |
| [5] | (c) Zhang, M.; Zhang, Y.; Jie, X.; Zhao, H.; Li, G.; Su, W. Org. Chem. Front. 2014, 1, 843. |
| [5] | (d) Huang, Z.; Lim, H. N.; Mo, F.; Young, M. C. Dong, G. Chem. Soc. Rev. 2015, 44, 7764. |
| [6] | (a) Zhang, L.-Q.; Yang, S.; Huang, X.; You, J.; Song, F. Chem. Commun. 2013, 49, 8830. |
| [6] | (b) Li, B.; Ma, J.; Xie, W.; Song, H.; Xu, S.; Wang, B. Eur. J. Chem. 2013, 19, 11863. |
| [6] | (c) Ikemoto, H.; Yoshino, T.; Sakata, K.; Matsunaga, S.; Kanai, M. J. Am. Chem. Soc. 2014, 136, 5424. |
| [6] | (d) Ikemoto, H.; Tanaka, R.; Sakata, K.; Kanai, M.; Yoshino, T.; Matsunaga, S. Angew. Chem., Int. Ed. 2017, 56, 7156. |
| [6] | (e) Li, M.; Yao, T.-Y.; Sun, S.-Z.; Yan, T.-X.; Wen, L.-R.; Zhang, L.-B. Org. Biomol. Chem. 2020, 18, 3158. |
| [6] | (f) Wu, X.; Lu, Y.; Qiao, J.; Dai, W.; Jia, X.; Ni, H.; Zhang, X.; Liu, H.; Zhao, F. Org. Lett. 2020, 22, 9163. |
| [6] | (g) Zhao, F.; Gong, X.; Lu, Y.; Qiao, J.; Jia, X.; Ni, H.; Wu, X.; Zhang, X. Org. Lett. 2021, 23, 727. |
| [7] | Lanke, V.; Prabhu, K. R. Org. Lett. 2013, 15, 2818. |
| [8] | (a) Ding, Z.; Yoshikai, N. Angew. Chem., Int. Ed. 2012, 51, 4698. |
| [8] | (b) Liang, L.; Fu, S.; Lin, D.; Zhang, X.-Q.; Deng, Y.; Jiang, H.; Zeng, W. J. Org. Chem. 2014, 79, 9472. |
| [8] | (c) Wong, M. Y.; Yamakawa, T.; Yoshikai, N. Org. Lett. 2015, 17, 442. |
| [8] | (d) Tian, P.; Feng, C.; Loh, T.-P. Nat. Commun. 2015, 6, 7472. |
| [8] | (e) Kong, L.; Zhou, X.; Li, X. Org. Lett. 2016, 18, 6320. |
| [8] | (f) Cai, S.-H.; Ye, L.; Wang, D.-X.; Wang, Y.-Q.; Lai, L.-J.; Zhu, C.; Feng, C.; Loh, T.-P. Chem. Commun. 2017, 53, 8731. |
| [8] | (g) Nakanowatari, S.; Mei, R.; Feldt, M.; Ackermann, L. ACS Catal. 2017, 7, 2511. |
| [8] | (h) Zhou, C.-N.; Xie, H.-H.; Zheng, Z.-A.; Xiao, Y.-C.; Li, G.; Shen, Y.-H.; Peng, W.-M.; Wang, L. Chem.-Eur. J. 2018, 24, 5469. |
| [8] | (i) Jagtap, R. A.; Vinod, C. P.; Punji, B. ACS Catal. 2019, 9, 431. |
| [8] | (j) Yang, L.; Zhang, G.; Huang, H. Adv. Synth. Catal. 2014, 356, 1509. |
| [8] | (k) Zhou, X.; Luo, Y.; Kong, L.; Xu, Y.; Zheng, G.; Lan, Y.; Li, X. ACS Catal. 2017, 7, 7296. |
| [8] | (l) Wang, Z.-T.; Zheng, Z.-A.; Li, P.-J.; Zhou, C.-N.; Cai, S.-J.; Xiao, B.; Wang, L. Chin. J. Chem. 2021, 39, 2823. |
| [9] | (a) Garc, A.; Ram, R.; Mez, G.; Array, S.; Carretero, J. C. Angew. Chem., Int. Ed. 2009, 121, 6633. |
| [9] | (b) Yan, Z.-L.; Chen, W.-L.; Gao, Y.-R.; Mao, S.; Zhang, Y.-L.; Wang, Y.-Q. Adv. Synth. Catal. 2014, 356, 1085. |
| [10] | Sharma, S.; Han, S.; Kim, M.; Mishra, N. K.; Park, J.; Shin, Y.; Ha, J.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. Org. Biomol. Chem. 2014, 12, 1703. |
| [11] | (a) Anbarasan, P.; Schareina, T.; Beller, M. Chem. Soc. Rev. 2011, 40, 5049. |
| [11] | (b) Fleming, F. F.; Yao, L.; Ravikumar, P. C.; Funk, L.; Shook, B. C. J. Med. Chem. 2010, 53, 7902. |
| [12] | (a) Ping, Y.; Wang, L.; Ding, Q.; Peng, Y. Adv. Synth. Catal. 2017, 359, 3274. |
| [12] | (b) Sinha, S. K.; Guin, S.; Maiti, S.; Biswas, J. P.; Porey, S.; Maiti, D. Chem. Rev. 2022, 122, 5682. |
| [13] | (a) Gandeepan, P.; Cheng, C.-H. Chem.-Asian J. 2015, 10, 824. |
| [13] | (b) Gandeepan, P.; Cheng, C.-H. J. Am. Chem. Soc. 2012, 134, 5738. |
| [13] | (c) Reddy, M. C.; Jeganmohan, M. Chem. Commun. 2015, 51, 10738. |
| [14] | (a) Leow, D.; Li, G.; Mei, T.-S.; Yu, J.-Q. Nature 2012, 486, 518. |
| [14] | (b) Yang, Y.-F.; Cheng, G.-J.; Liu, P.; Leow, D.; Sun, T.-Y.; Chen, P.; Zhang, X.; Yu, J.-Q.; Wu, Y.-D.; Houk, K. N. J. Am. Chem. Soc. 2014, 136, 344. |
| [14] | (c) Tang, R.-Y.; Li, G.; Yu, J.-Q. Nature 2014, 507, 215. |
| [14] | (d) Yang, G.; Lindovska, P.; Zhu, D; Kim, J.; Wang, P.; Tang, R.-Y.; Movassaghi, M.; Yu, J.-Q. J. Am. Chem. Soc. 2014, 136, 10807. |
| [14] | (e) Deng, Y.; Yu, J.-Q. Angew. Chem., Int. Ed. 2015, 54, 888. |
| [14] | (f) Xu, H.-J.; Lu, Y.; Farmer, M. E.; Wang, H.-W.; Zhao, D.; Kang, Y.-S.; Sun, W.-Y.; Yu, J.-Q. J. Am. Chem. Soc. 2017, 139, 2200. |
| [14] | (g) Fan, Z.; Bay, K. L.; Chen, X.; Zhuang, Z.; Park, H. S.; Yeung, K.-S.; Houk, K. N.; Yu, J.-Q. Angew. Chem., Int. Ed. 2020, 59, 4770. |
| [14] | (h) Lam, N. Y. S.; Fan, Z.; Wu, K.; Park, H. S.; Shim, S. Y.; Strassfeld, D. A.; Yu, J.-Q. J. Am. Chem. Soc. 2022, 144, 2793. |
| [15] | (a) Bera, M.; Modak, A.; Patra, T.; Maji, A.; Maiti, D. Org. Lett. 2014, 16, 5760. |
| [15] | (b) Bag, S.; Patra, T.; Modak, A.; Deb, A.; Maity, S.; Dutta, U.; Dey, A.; Kancherla, R.; Maji, A.; Hazra, A.; Bera, M.; Maiti, D. J. Am. Chem. Soc. 2015, 137, 11888. |
| [15] | (c) Bera, M.; Maji, A.; Sahoo, S. K.; Maiti, D. Angew. Chem., Int. Ed. 2015, 54, 8515. |
| [15] | (d) Dey, A.; Maity, S.; Maiti, D. Chem. Commun. 2016, 52, 12398. |
| [16] | Lee, S.; Lee, H.; Tan, K. L. J. Am. Chem. Soc. 2013, 135, 18778. |
| [17] | Ramesh, P.; Sreenivasulu, C.; Kishore, D. R.; Srinivas, D.; Gorantla, K. R.; Mallik, B. S.; Satyanarayana, G. J. Org. Chem. 2022, 87, 2204. |
| [18] | (a) Li, S.; Ji, H.; Cai, L.; Li, G. Chem. Sci. 2015, 6, 5595. |
| [18] | (b) Li, S.; Cai, L.; Ji, H.; Yang, L.; Li, G. Nat. Commun. 2016, 7, 10443. |
| [19] | Ping, L.; Chung, D. S.; Bouffard, J.; Lee, S. G. Chem. Soc. Rev. 2017, 46, 4299. |
| [20] | Yan, F.; Huang, Z.; Du, C.-X.; Bai, J.-F.; Li, Y. J. Catal. 2021, 395, 188. |
| [21] | (a) Enders, D.; Shilvock, J. P. Chem. Soc. Rev. 2000, 29, 359. |
| [21] | (b) Otto, N.; Opatz, T. Chem.-Eur. J. 2014, 20, 13064. |
| [22] | Donald, J. R.; Berrell, S. L. Chem. Sci. 2019, 10, 5832. |
| [23] | (a) Wang, Y.-J.; Wang, T.-T.; Yao, L.; Wang, Q.-L.; Zhao, L.-M. J. Org. Chem. 2020, 85, 9514. |
| [23] | (b) Jambu, S.; Tamizmani, M.; Jeganmohan, M. Org. Lett. 2018, 20, 1982. |
| [23] | (c) Chen, W.; Li, H.-J.; Li, Q.-Y.; Wu, Y.-C. Org. Biomol. Chem. 2020, 18, 500. |
| [23] | (d) Leitch, J. A.; Wilson, P. B.; McMullin, C. L.; Mahon, M. F.; Bhonoah, Y.; Williams, I. H.; Frost, C. G. ACS Catal. 2016, 6, 5520. |
/
| 〈 |
|
〉 |