

一类新型卤化偕二硼试剂的合成
收稿日期: 2022-07-23
修回日期: 2022-08-17
网络出版日期: 2022-09-09
基金资助
国家自然科学基金(22022113)
A Novel Synthesis of Halogenated gem-Diboron Reagents
Received date: 2022-07-23
Revised date: 2022-08-17
Online published: 2022-09-09
Supported by
National Natural Science Foundation of China(22022113)
方通昌 , 徐良轩 , 秦玉承 , 姜南权 , 刘超 . 一类新型卤化偕二硼试剂的合成[J]. 有机化学, 2023 , 43(2) : 777 -780 . DOI: 10.6023/cjoc202207030
A novel synthesis of halogenated gem-diboronates (X-CR(Bpin)2) was developed. The reagents are stable and easy to prepare in a large scale. Tetra-substituted halogenated gem-diboronates can be prepared in high yields when using alkyl- substituted gem-dihalides, providing a new strategy for the synthesis of new gem-diboron compounds.
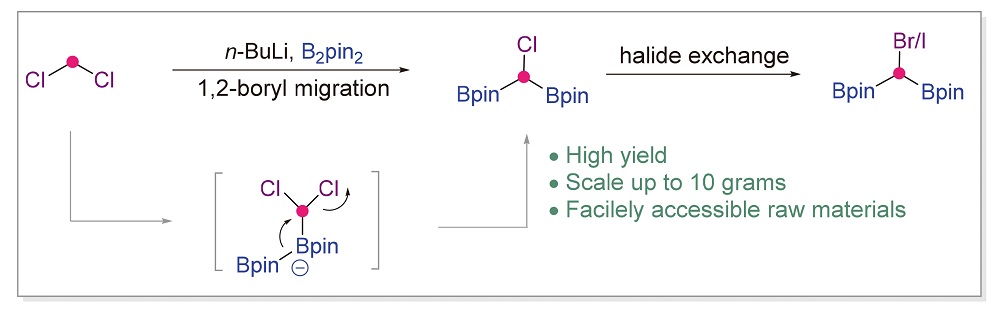
Key words: element-organic chemistry; reagent chemistry; organoboron; gem-diboron
| [1] | (a) Wu, C.; Wang, J. Tetrahedron Lett. 2018, 59, 2128. |
| [1] | (b) Nallagonda, R.; Padala, K.; Masarwa, A. Org. Biomol. Chem. 2018, 16, 1050. |
| [1] | (c) Miralles, N.; Maza, R. J.; Fernández, E. Adv. Synth. Catal. 2018, 360, 1306. |
| [1] | (d) Jo, W.; Lee, J. H.; Cho, S. H. Chem. Commun. 2021, 57, 4346. |
| [1] | (e) Lee, Y.; Han, S.; Cho, S. H. Acc. Chem. Res. 2021, 54, 3917. |
| [1] | (f) Shi, D.; Xia, C.; Liu, C. CCS Chem. 2021, 3, 1718. |
| [1] | (g) Wang, S.; Sun, M.; Zhang, H.; Zhang, J.; He, Y.; Feng, Z. CCS Chem. 2020, 2, 2164. |
| [2] | (a) Endo, K.; Ohkubo, T.; Hirokami, M.; Shibata, T. J. Am. Chem. Soc. 2010, 132, 11033. |
| [2] | (b) Hong, K.; Liu, X.; Morken, J. P. J. Am. Chem. Soc. 2014, 136, 10581. |
| [2] | (c) Potter, B.; Szymaniak, A. A.; Edelstein, E. K.; Morken, J. P. J. Am. Chem. Soc. 2014, 136, 17918. |
| [2] | (d) Sun, C.; Potter, B.; Morken, J. P. J. Am. Chem. Soc. 2014, 136, 6534. |
| [3] | (a) Li, H.; Zhang, Z.; Shangguan, X.; Huang, S.; Chen, J.; Zhang, Y.; Wang, J. Angew. Chem., Int. Ed. 2014, 53, 11921. |
| [3] | (b) Xu, S.; Shangguan, X.; Li, H.; Zhang, Y.; Wang, J. J. Org. Chem. 2015, 80, 7779. |
| [4] | (a) Sun, W.; Wang, L.; Hu, Y.; Wu, X.; Xia, C.; Liu, C. Nat. Commun. 2020, 11, 3113. |
| [4] | (b) Sun, W.; Wang, L.; Xia, C.; Liu, C. Angew. Chem. Int. Ed. 2018, 57, 5501. |
| [4] | (c) Zheng, P.; Zhai, Y.; Zhao, X.; Xu, T. Chem. Commun. 2018, 54, 13375. |
| [4] | (d) Iacono, C. E.; Stephens, T. C.; Rajan, T. S.; Pattison, G. J. Am. Chem. Soc. 2018, 140, 2036. |
| [4] | (e) Lee, B.; Chirik, P. J. J. Am. Chem. Soc. 2020, 142, 2429. |
| [5] | (a) Murray, S. A.; Green, J. C.; Tailor, S. B.; Meek, S. J. Angew. Chem., Int. Ed. 2016, 55, 9065. |
| [5] | (b) Joannou, M. V.; Moyer, B. S.; Meek, S. J. J. Am. Chem. Soc. 2015, 137, 6176. |
| [5] | (c) Joannou, M. V.; Moyer, B. S.; Goldfogel, M. J.; Meek, S. J. Angew. Chem., Int. Ed. 2015, 54, 14141. |
| [5] | (d) Kim, J.; Ko, K.; Cho, S. H. Angew. Chem. Int. Ed. 2017, 56, 11584. |
| [6] | Cuenca, A. B.; Fernández, E. Chem. Soc. Rev. 2021, 50, 72. |
| [7] | (a) Wang, L.; Zhang, T.; Sun, W.; He, Z.; Xia, C.; Lan, Y.; Liu, C. J. Am. Chem. Soc. 2017, 139, 5257. |
| [7] | (b) He, Z.; Zhu, Q.; Hu, X.; Wang, L.; Xia, C.; Liu, C. Org. Chem. Front. 2019, 6, 900. |
| [7] | (c) Li, H.; Shangguan, X.; Zhang, Z.; Huang, S.; Zhang, Y.; Wang, J. Org. Lett. 2014, 16, 448. |
| [7] | (d) Palmer, W. N.; Obligacion, J. V.; Pappas, I.; Chirik, P. J. J. Am. Chem. Soc. 2016, 138, 766. |
| [7] | (e) Li, L.; Gong, T.; Lu, X.; Xiao, B.; Fu, Y. Nat. Commun. 2017, 8, 345. |
| [7] | (f) Zhao, H.; Tong, M.; Wang, H.; Xu, S. Org. Biomol. Chem. 2017, 15, 3418. |
| [7] | (g) Gao, G.; Kuang, Z.; Song, Q. Org. Chem. Front. 2018, 5, 2249. |
| [7] | (h) Teo, W. J.; Ge, S. Angew. Chem. Int. Ed. 2018, 57, 1654. |
| [7] | (i) Lee, H.; Lee, Y.; Cho, S. H. Org. Lett. 2019, 21, 5912. |
| [7] | (j) Docherty, J. H.; Nicholson, K.; Dominey, A. P.; Thomas, S. P. ACS Catal. 2020, 10, 4686. |
| [7] | (k) Wu, F.-P.; Wu, X.-F. Angew. Chem. Int. Ed. 2021, 60, 11730. |
| [7] | (l) Wang, X.; Cui, X.; Li, S.; Wang, Y.; Xia, C.; Jiao, H.; Wu, L. Angew. Chem. Int. Ed. 2020, 59, 13608. |
| [8] | (a) Kim, J.; Cho, S. H. ACS Catal. 2018, 9, 230. |
| [8] | (b) Kim, J.; Lee, E.; Cho, S. H. Asian J. Org. Chem. 2019, 8, 1664. |
| [9] | Lee, Y.; Park, J.; Cho, S. H. Angew. Chem. Int. Ed. 2018, 57, 12930. |
| [10] | (a) Matteson, D. S. Chem. Rev. 2002, 89, 1535. |
| [10] | (b) Zeng, Y. F.; Ji, W. W.; Lv, W. X.; Chen, Y.; Tan, D. H.; Li, Q.; Wang, H. Angew. Chem. Int. Ed. 2017, 56, 14707. |
| [10] | (c) Fang, T.; Qiu, J.; Yang, K.; Song, Q. Org. Chem. Front. 2021, 8, 1991. |
| [10] | (d) Yang, L.; Tan, D. H.; Fan, W. X.; Liu, X. G.; Wu, J. Q.; Huang, Z. S.; Li, Q.; Wang, H. Angew. Chem. Int. Ed. 2021, 60, 3454. |
| [11] | Matteson, D. S.; Davis, R. A.; Hagelee, L. A. J. Organomet. Chem. 1974, 69, 45. |
| [12] | During our progress of developing the synthesis of α-halo gem- diboronates, a synthesis and transformation of α-halo gem-diborylmethane via halogenation of (gem-diborylmethyl)lithium appeared online: Hwang, C.; Lee, Y.; Kim, M.; Seo, Y.; Cho, S. H. Angew. Chem. Int. Ed. 2022, e202209079. |
| [13] | (a) Hu, Y.; Sun, W.; Zhang, T.; Xu, N.; Xu, J.; Lan, Y.; Liu, C. Angew. Chem. Int. Ed. 2019, 58, 15813. |
| [13] | (b) Kuang, Z.; Chen, H.; Yan, J.; Yang, K.; Lan, Y.; Song, Q. Org. Lett. 2018, 20, 5153. |
| [14] | Matteson, D. S.; Majumdar, D. Organometallics 2002, 2, 1529. |
/
| 〈 |
|
〉 |