

海洋来源真菌Eutypella sp. F0219次级代谢产物研究
收稿日期: 2022-06-26
修回日期: 2022-08-16
网络出版日期: 2022-09-15
基金资助
海南省重大科技计划(ZDKJ202008); 海南省自然科学基金(221QN229); 大学生创新创业训练计划(202111658007)
Study on Secondary Metabolites of Marine-Derived Fungus Eutypella sp. F0219
Received date: 2022-06-26
Revised date: 2022-08-16
Online published: 2022-09-15
Supported by
Key Science and Technology Program of Hainan Province(ZDKJ202008); Hainan Provincial Natural Science Foundation of China(221QN229); Innovation and Entrepreneurship Training Program for College Students(202111658007)
弯孢聚壳属真菌能产生结构丰富多样的活性化合物. 对一株来自海洋沉积物的真菌Eutypella sp. F0219代谢产物研究中, 分离得到一个新的异戊烯基取代二氢异香豆素和一个新的色烯酰胺衍生物, 分别命名为eutypellarins A和B (1和2), 以及两个已知的异戊二烯取代苯甲醛衍生物(3和4). 通过高分辨质谱、核磁共振波谱以及量子化学计算对新化合物的结构进行确定. 首次从弯孢聚壳属真菌中发现异戊二烯取代二氢异香豆素和色烯酰胺衍生物. 采用2,2'-联氮-双-3-乙基苯并噻唑啉-6-磺酸(ABTS)自由基清除法和铁离子还原(FRAP)法对所有化合物进行总抗氧化能力的测定. 结果显示化合物1和4具有中等的抗氧化效果, 其Trolox等效抗氧化能力分别为(0.51±0.016)和(0.76±0.007).
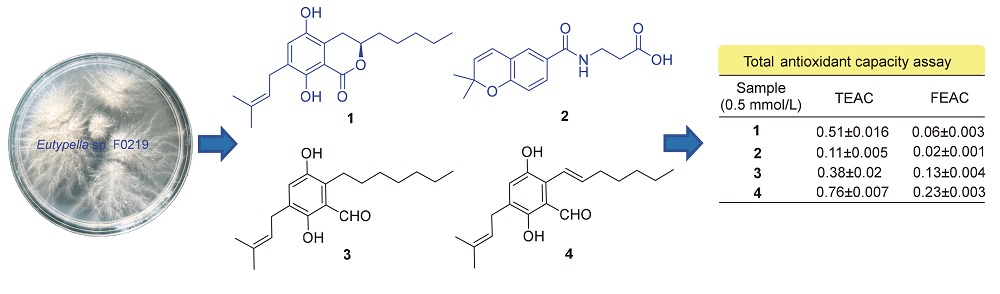
关键词: 海洋真菌; 弯孢聚壳属; 异戊二烯取代二氢异香豆素; 色烯酰胺衍生物; 总抗氧化能力测试
易继凌 , 施康琦 , 吴冰林 , 黎婉珊 , 陈光英 . 海洋来源真菌Eutypella sp. F0219次级代谢产物研究[J]. 有机化学, 2023 , 43(1) : 295 -298 . DOI: 10.6023/cjoc202206046
The fungus Eutypella sp. is well known for producing bioactive compounds with diverse structures. Chemical investigation of the fungi Eutypella sp. F0219 isolated from marine sediment, yielded a new prenylated dihydroisocoumarin and a new chromene amide derivative, named eutypellarins A and B (1 and 2), together with two known prenylated benzaldehyde derivatives (3 and 4). The structures of these compounds were elucidated based on HR-ESIMS, extensive nucler magneti resonance (NMR) experiments, and quantum-chemical electronic circular dichroism (ECD) calculations. Prenylated dihydroisocoumarin and chromene amide derivatives are reported from the genus Eutypella for the first time. The total antioxidant capacities of all compounds were measured through 2,2'-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid (ABTS) and ferric-reducing antioxidant power (FRAP) assay. The results showed that compounds 1 and 4 exhibited moderate antioxidant properities with Trolox equivalent antioxidant capacity (TEAC) values (0.51±0.016) and (0.76±0.007), respectively.

| [1] | Mostafa, E. R; Rainer, E. Nat. Prod. Rep. 2011, 28, 290. |
| [2] | Sun, L.; Li, D.; Tao, M.; Chen, Y.; Dan, F.; Zhang, W. Mar. Drugs 2012, 10, 539. |
| [3] | Zhou, Y.; Zhang, Y.-X.; Zhang, J.-P.; Yu, H.-B.; Liu, X.-Y.; Lu, X.-L.; Jiao, B.-H. Nat. Prod. Res. 2017, 31, 1674. |
| [4] | Wang, Y. Z.; Wang, Y.; Wu, A. A.; Zhang, L.; Hu, Z. Y.; Huang, H. Y.; Xu, Q. Y.; Deng, X. M. J. Antibiot. 2017, 70, 1029. |
| [5] | Isaka, M.; Palasarn, S.; Lapanun, S.; Chanthaket, R.; Boonyuen, N.; Lumyong, S. J. Nat. Prod. 2009, 72, 1720. |
| [6] | Liu, J.-T.; Hu, B.; Gao, Y.; Zhang, J.-P.; Jiao, B.-H.; Lu, X.-L.; Liu, X.-Y. Chem. Biodiversity 2014, 11, 800. |
| [7] | Isaka, M.; Palasarn, S.; Prathumpai, W.; Laksanacharoen, P. Chem. Pharm. Bull. 2011, 59, 1157. |
| [8] | Liu, H. X.; Zhang, L.; Chen, Y. C.; Li, S. N.; Tan, G. H.; Sun, Z. H.; Pan, Q. L.; Ye, W.; Li, H. H.; Zhang, W. M. Nat. Prod. Res. 2017, 31, 401. |
| [9] | Yu, H.-B; Wang, X.-L.; Zhang, Y.-X.; Xu, W.-H.; Zhang, J.-P.; Zhou, X.-Y.; Lu, X.-L.; Liu, X.-Y.; Jiao, B.-H. J. Nat. Prod. 2018, 81, 1553. |
| [10] | Wang, X.; Sun, K.; Wang, B. Chem. Biodiversity 2018, 15, e1700501. |
| [11] | Lu, X.-L.; Liu, J.-T.; Liu, X.-Y.; Gao, Y.; Zhang, J.; Jiao, B.-H.; Zheng, H. J. Antibiot. 2014, 67, 171. |
| [12] | Zhang, L.-Q.; Chen, X.-C.; Chen, Z.-Q.; Wang, G.-M.; Zhu, S.-G.; Yang, Y.-F.; Chen, K.-X.; Liu, X.-Y.; Li, Y.-M. Mar. Drugs 2016, 14, 44. |
| [13] | Yu, H.-B.; Wang, X.-L.; Xu, W.-H.; Zhang, Y.-X.; Qian, Y.-S.; Zhang, J.-P.; Lu, X.-L.; Liu, X.-L. Mar. Drugs 2018, 16, 284. |
| [14] | Zhang, W.; Zhu, Y.; Yu, H.; Liu, X.; Jiao, B.; Lu, X. Molecules 2021, 26, 315. |
| [15] | Niu, S.; Liu, D.; Shao, Z.; Proksch, P.; Lin, W. RSC Adv. 2017, 7, 33580. |
| [16] | Niu, S.; Liu, D.; Shao, Z.; Proksch, P.; Lin, W. Tetrahedron Lett. 2017, 58, 3695. |
| [17] | Liao, H.-X.; Sun, D.-W.; Zheng, C.-J.; Wang, C.-Y. Nat. Prod. Res. 2017, 31, 1640. |
| [18] | Zhang, Y.-X.; Yu, H.-B.; Xu, W.-H.; Hu, B.; Guild, A.; Zhang, J.-P.; Lu, X.-L.; Liu, X.-Y.; Jiao, B.-H. J. Nat. Prod. 2019, 82, 3089. |
| [19] | Chen, M.; Shao, C.-L.; Wang, K.-L.; Xu, Y.; She, Z.-G.; Wang, C.-Y. Tetrahedron 2014, 70, 9123. |
| [20] | Matta, A.; Sharma, A. K.; Tomar, S.; Cao, P.; Kumar, S.; Balwani, S.; Ghosh, B.; Prasad, A. K.; Van der Eycken, E. V.; DePass, A. L.; Wengel, J.; Parmar, V. S.; Len, C.; Singh, B. K. New J. Chem. 2020, 44, 1371. |
| [21] | Fathallah, N.; Raafat, M. M.; Issa, M. Y.; Abdel-Aziz, M. M.; Bishr, M.; Abdelkawy, M. A.; Salama, O. Molecules 2019, 24, 4118. |
/
| 〈 |
|
〉 |