

Daphnezomines A和B的四环核心骨架合成
收稿日期: 2022-08-13
修回日期: 2022-09-02
网络出版日期: 2022-10-10
基金资助
国家自然科学基金(21971104); 广东省教育厅高校重点科研项目(2021ZDZX2035); 深圳湾实验室(SZBL2019062801006); 深圳市诺贝尔奖科学家实验室(C17783101); 广东省本土创新团队项目(2019BT02Y335); 广东省高校创新团队(2020KCXTD016); 广东省催化化学重点实验室(2020B121201002); 深圳市科技创新委员会(ZDSYS20190902093215877)
Synthesis of Tetracyclic Core Structure of Daphnezomines A and B
Received date: 2022-08-13
Revised date: 2022-09-02
Online published: 2022-10-10
Supported by
National Natural Science Foundation of China(21971104); Key Research Projects in Colleges and Universities of Education Department of Guangdong Province(2021ZDZX2035); Shenzhen Bay Laboratory(SZBL2019062801006); Shenzhen Nobel Prize Scientists Laboratory Project(C17783101); Guangdong Innovative Program(2019BT02Y335); Innovative Team of Universities in Guangdong Province(2020KCXTD016); Guangdong Provincial Key Laboratory of Catalysis(2020B121201002); Shenzhen Science and Technology Innovation Committee(ZDSYS20190902093215877)
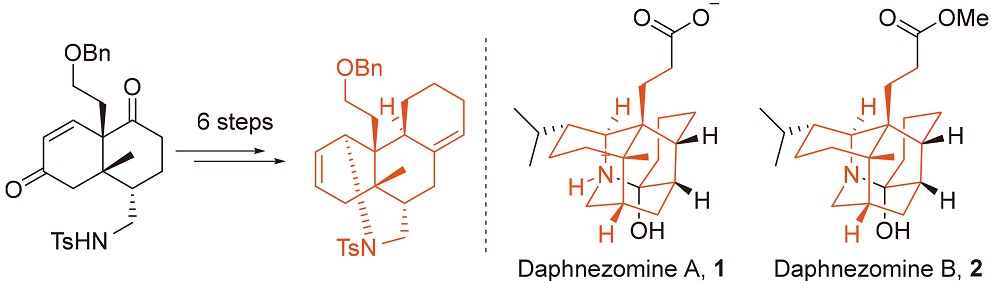
在虎皮楠生物碱家族中, daphnezomine A型生物碱仅包含三位成员: daphnezomine A (1), daphnezomine B (2)以及dapholdhamine B. 这些生物碱含有独特的氮杂金刚烷骨架, 9个连续的手性立体中心, 因此呈现出巨大的合成挑战性. 报道了分子1和2的四环核心骨架的合成. 关键步骤包括一个黄氏酰胺活化增环反应和一个Hutchins-Kabalka还原重排反应.
胡晶平 , 陈文清 , 蒋宇旸 , 徐晶 . Daphnezomines A和B的四环核心骨架合成[J]. 有机化学, 2023 , 43(1) : 171 -177 . DOI: 10.6023/cjoc202208014
Among the tiniest Daphniphyllum alkaloid subfamilies, daphnezomine A-type alkaloids consist of only three known members, namely daphnezomine A (1), daphnezomine B (2) and dapholdhamine B. These alkaloids contain a unique aza-adamantane core along with nine contiguous stereogenic centers, thus presenting remarkable synthetic challenges. The synthesis of tetracyclic core structure of 1 and 2 was reported. The key steps in our approach include a Huang’s amide-activa tion-annulation and a Hutchins-Kabalka reductive rearrangement.

| [1] | (a) Li, Z.-Y.; Guo, Y.-W. Chin. J. Org. Chem. 2007, 27, 565. (in Chinese) |
| [1] | (李震宇, 郭跃伟, 有机化学, 2007, 27, 565.) |
| [1] | (b) Liang, X.; Yang, X.-Z.; Chen, L.; Jiang, S.; Chen, Y.-D.; Deng, Q.-Y.; Chen, X.-G.; Yuan, J.-Q. Med. Chem. Res. 2020, 30, 1. |
| [1] | (c) Wu, H.; Zhang, X.; Ding, L.; Chen, S.; Yang, J.; Xu, X. Planta Med. 2013, 79, 1589. |
| [1] | (d) Xu, J. B.; Zhang, H.; Gan, L. S.; Han, Y. S.; Wainberg, M. A.; Yue, J. M. J. Am. Chem. Soc. 2014, 136, 7631. |
| [2] | (a) Kobayashi, J.; Kubota, T. Nat. Prod. Rep. 2009, 26, 936. |
| [2] | (b) Chattopadhyay, A. K.; Hanessian, S. Chem. Rev. 2017, 117, 4104. |
| [2] | (c) Kang, B.; Jakubec, P.; Dixon, D. J. Nat. Prod. Rep. 2014, 31, 550. |
| [2] | (d) Guo, L.-D.; Chen, Y.; Xu, J. Acc. Chem. Res. 2020, 53, 2726. |
| [2] | (e) Zhong, J.; Wang, H.; Zhang, Q.; Gao, S. Alkaloids: Chem. Biol. 2021, 85, 113. |
| [3] | (a) Piettre, S.; Heathcock, C. H. Science 1990, 248, 1532. |
| [3] | (b) Heathcock, C. H.; Davidsen, S. K.; Mills, S.; Sanner, M. A. J. Am. Chem. Soc. 1986, 108, 5650. |
| [3] | (c) Ruggeri, R. B.; Heathcock, C. H. J. Org. Chem. 1990, 55, 3714. |
| [3] | (d) Heathcock, C. H.; Kath, J. C.; Ruggeri, R. B. J. Org. Chem. 1995, 60, 1120. |
| [3] | (e) Ruggeri, R. B.; Hansen, M. M.; Heathcock, C. H. J. Am. Chem. Soc. 1988, 110, 8734. |
| [3] | (f) Stafford, J. A.; Heathcock, C. H. J. Org. Chem. 1990, 55, 5433. |
| [3] | (g) Heathcock, C. H.; Stafford, J. A.; Clark, D. L. J. Org. Chem. 1992, 57, 2575. |
| [3] | (h) Ruggeri, R. B.; McClure, K. F.; Heathcock, C. H. J. Am. Chem. Soc. 1989, 111, 1530. |
| [4] | Weiss, M. E.; Carreira, E. M. Angew. Chem., Int. Ed. 2011, 50, 11501. |
| [5] | (a) Lu, Z. Y.; Li, Y.; Deng, J.; Li, A. Nat. Chem. 2013, 5, 679. |
| [5] | (b) Li, J.; Zhang, W. H.; Zhang, F.; Chen, Y.; Li, A. J. Am. Chem. Soc. 2017, 139, 14893. |
| [5] | (c) Chen, Y.; Zhang, W. H.; Ren, L.; Li, J.; Li, A. Angew. Chem., Int. Ed. 2018, 57, 952. |
| [5] | (d) Zhang, W. H.; Ding, M.; Li, J.; Guo, Z. C.; Lu, M.; Chen, Y.; Liu, L. C.; Shen, Y. H.; Li, A. J. Am. Chem. Soc. 2018, 140, 4227. |
| [6] | (a) Shvartsbart, A.; Smith, A. B., III J. Am. Chem. Soc. 2014, 136, 870. |
| [6] | (b) Shvartsbart, A.; Smith, A. B., III J. Am. Chem. Soc. 2015, 137, 3510. |
| [7] | Yamada, R.; Adachi, Y.; Yokoshima, S.; Fukuyama, T. Angew. Chem.,Int. Ed. 2016, 55, 6067. |
| [8] | Shi, H.; Michaelides, I. N.; Darses, B.; Jakubec, P.; Nguyen, Q. N. N.; Paton, R. S.; Dixon, D. J. J. Am. Chem. Soc. 2017, 139, 17755. |
| [9] | Chen, X.; Zhang, H.-J.; Yang, X.; Lv, H.; Shao, X.; Tao, C.; Wang, H.; Cheng, B.; Li, Y.; Guo, J.; Zhang, J.; Zhai, H. Angew. Chem.,Int. Ed. 2018, 57, 947. |
| [10] | (a) Xu, B.; Wang, B.; Xun, W.; Qiu, F. G. Angew. Chem.,Int. Ed. 2019, 58, 5754. |
| [10] | (b) Wang, B.; Xu, B.; Xun, W.; Guo, Y.; Zhang, J.; Qiu, F. G. Angew. Chem., Int. Ed. 2021, 60, 9439. |
| [11] | Zhong, J.; Chen, K.; Qiu, Y.; He, H.; Gao, S. Org. Lett. 2019, 21, 3741. |
| [12] | (a) Hugelshofer, C. L.; Palani, V.; Sarpong, R. J. Am. Chem. Soc. 2019, 141, 8431. |
| [12] | (b) Hugelshofer, C. L.; Palani, V.; Sarpong, R. J. Org. Chem. 2019, 84, 14069. |
| [13] | Xu, G.; Wu, J.; Li, L.; Lu, Y.; Li, C. J. Am. Chem. Soc. 2020, 142, 15240. |
| [14] | Cao, M.-Y.; Ma, B.-J.; Gu, Q.-X.; Fu, B.; Lu, H.-H. J. Am. Chem. Soc. 2022, 144, 5750. |
| [15] | Chattopadhyay, A. K.; Ly, V. L.; Jakkepally, S.; Berger, G.; Hanessian, S. Angew. Chem.,Int. Ed. 2016, 55, 2577. |
| [16] | (a) Chen, Y.; Hu, J.; Guo, L.-D.; Zhong, W.; Ning, C.; Xu, J. Angew. Chem., Int. Ed. 2019, 58, 7390. |
| [16] | (b) Guo, L.-D.; Hou, J.; Tu, W.; Zhang, Y.; Zhang, Y.; Chen, L.; Xu, J. J. Am. Chem. Soc. 2019, 141, 11713. |
| [16] | (c) Guo, L.-D.; Hu, J.; Zhang, Y.; Tu, W.; Zhang, Y.; Pu, F.; Xu, J. J. Am. Chem. Soc. 2019, 141, 13043. |
| [16] | (d) Guo, L.-D.; Zhang, Y.; Hu, J.; Ning, C.; Fu, H.; Chen, Y.; Xu, J. Nat. Commun. 2020, 11, 3538. |
| [16] | For our very recent total syntheses of other Daphniphyllum alkaloids, see: |
| [16] | (e) Zhang, Y.; Chen, Y.; Song, M.; Tan, B.; Jiang, Y.; Yan, C.; Jiang, Y.; Hu, X.; Zhang, C.; Chen, W.; Xu, J. J. Am. Chem. Soc. 2022, 144, 16042. |
| [16] | (f) Hu, J.; Guo, L.-D.; Chen, W.; Jiang, Y.; Pu, F.; Ning, C.; Xu, J. Org. Lett. 2022, 24, 7416. |
| [17] | Morita, H.; Yoshida, N.; Kobayashi, J. J. Org. Chem. 1999, 64, 7208. |
| [18] | Zhang, Y.; Di, Y.-T.; Mu, S.-Z.; Li, C.-S.; Zhang, Q.; Tan, C.-J.; Zhang, Z.; Fang, X.; Hao, X.-J. J. Nat. Prod. 2009, 72, 1325. |
| [19] | (a) Wu, D.-P.; He, Q.; Chen, D.-H.; Ye, J.-L.; Huang, P.-Q. Chin. J. Chem. 2019, 37, 315. |
| [19] | (b) Huang, P.-Q.; Huang, Y.-H. Chin. J. Chem. 2017, 35, 613. |
| [19] | (c) Huang, P. -, Q.; Huang, Y.-H.; Wang, S.-R. Org. Chem. Front. 2017, 4, 431. |
| [19] | (d) Huang, P.-Q.; Huang, Y.-H.; Geng, H.; Ye, J.-L. Sci. Rep. 2016, 6, 28801. |
| [20] | (a) Hutchins, R. O.; Kacher, M.; Rua, L. J. Org. Chem. 1975, 40, 923. |
| [20] | (b) Kabalka, G. W.; Yang, D. T. C.; Baker, J. D. J. Org. Chem. 1976, 41, 574. |
| [21] | Crystallographic data for compound C9-epi-4 (CCDC 2170411) has been deposited at the Cambridge Crystallographic Data Centre. |
| [22] | Yang, J.; Xiong, Z.; Chen, Y.; Li, Y. Synth. Commun. 1997, 27, 2985. |
| [23] | Wang, Z. Meinwald Rearrangement. In Comprehensive Organic Name Reactions and Reagents, Wiley Online Library, Hoboken, NJ, USA, 2010, pp. 1880-1882. |
| [24] | Crystallographic data for compound 14 (CCDC 2170422) has been deposited at the Cambridge Crystallographic Data Centre. |
| [25] | For our recent synthesis and studies of other caged natural products, see: |
| [25] | (a) Xie, S. Ning, C.; Yu, Q.; Hou, J.; Xu, J. Chin. J. Chem. 2021, 39, 137. |
| [25] | (b) Xie, S.; Chen, G.; Yan, H.; Hou, J.; He, Y.; Zhao, T.; Xu, J. J. Am. Chem. Soc. 2019, 141, 3435. |
| [25] | (c) Zhang, Y.; Guo, L.-D.; Xu, J. Chin. J. Org. Chem. 2019, 39, 1079. (in Chinese) |
| [25] | (张焱, 郭联东, 徐晶, 有机化学, 2019, 39, 079.) |
| [25] | (d) Zhao, N.; Yin, S.; Xie, S.; Yan, H.; Ren, P.; Chen, G.; Chen, F.; Xu, J. Angew. Chem. Int. Ed. 2018, 57, 3386. |
/
| 〈 |
|
〉 |