

烯胺酮α-官能团化反应的研究进展
收稿日期: 2022-07-18
修回日期: 2022-08-26
网络出版日期: 2022-10-14
基金资助
河南省科技攻关(222102310580)
Progress in the Study of α-Functionalization of Enaminone
Received date: 2022-07-18
Revised date: 2022-08-26
Online published: 2022-10-14
Supported by
Scientific and Technological Project of Henan Province(222102310580)
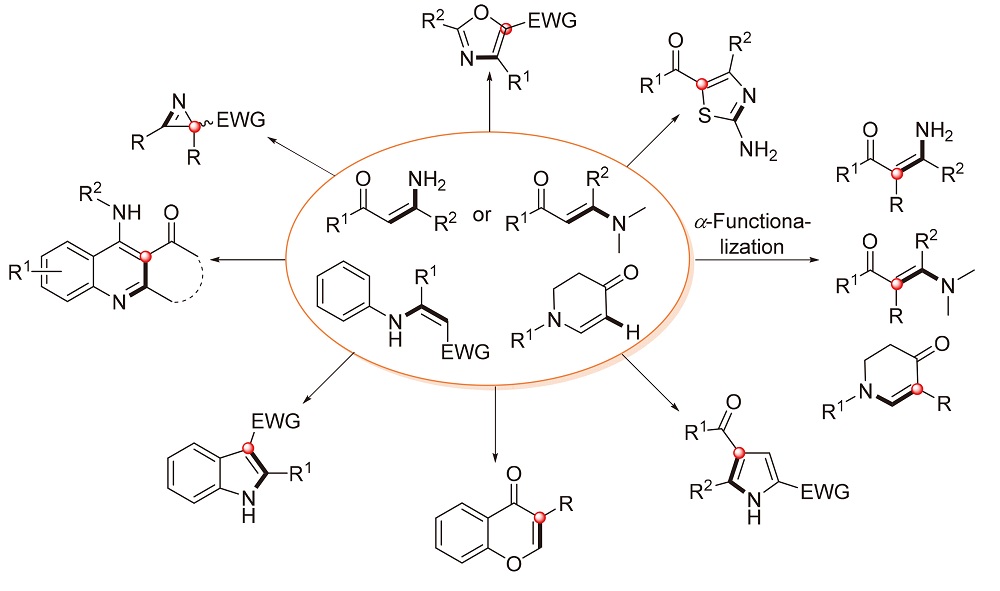
烯胺酮是一类非常重要的有机合成砌块, 具有易获得、储存方便、反应多样性等优点. 更重要的是, 烯胺酮是许多杂环化合物的重要前体. 最近, 通过C-H活化对烯胺酮进行过渡金属催化或无过渡金属的α-官能团化反应已成为构建官能化烯胺酮或杂环化合物的一种更为原子和步骤经济的策略, 并引起了许多有机化学家的关注. 根据成键类型, 该综述分为五个部分: C-C键的形成、C-O键的形成、C-N键的形成和C-X键的形成以及C-S/C-Se键的形成. 主要对烯胺酮α位官能团化反应进行了综述, 从反应机理、反应体系、底物范围等角度系统地综述了烯胺酮α位官能团化反应的进展.
刘宁 , 爨晓丹 , 李慧 , 段希焱 . 烯胺酮α-官能团化反应的研究进展[J]. 有机化学, 2023 , 43(2) : 602 -621 . DOI: 10.6023/cjoc202207027
Enaminones are versatile and extremely useful building blocks in organic synthesis with the advantages of easy availability, convenient storage, and distinct reactivity. In particular, such compounds are important precursors in heterocyclic chemistry. Recently, transition-metal-catalyzed or transition-metal-free α-functionalization of enaminones by C-H activation has emerged as a more atom- and step-economical strategy for the construction of functionalized enaminones or various heterocycles, and it has attracted the attention of many organic chemists. This review is divided into five sections based on the varied chemical bonds: C-C bond formation, C-O bond formation, C-N bond formation, C-X bond formation, and C-S/Se bond formation. The recent progress in functionalization of enaminone based on the reaction mechanism, reaction system, the scope of substrates, etc. is systematically reviewed.

Key words: enaminone; functionalization; coupling reaction; hypervalent iodine
| [1] | Greenhill, B. J. V. Chem. Soc. Rev. 1977, 6, 277. |
| [2] | Stanovnik, B.; Svete, J. Chem. Rev. 2004, 104, 2433. |
| [3] | (a) Hou, G.; Li, W.; Ma, M.; Zhang, X.; Zhang, X. J. Am. Chem. Soc. 2010, 132, 12844. |
| [3] | (b) Wang, Q.; Huang, W.; Yuan, H.; Cai, Q.; Chen, L.; Lv, H.; Zhang, X. J. Am. Chem. Soc. 2014, 136, 16120. |
| [4] | Elassara, A.-Z. A.; El-Khair, A. A. Tetrahedron 2003, 59, 8463. |
| [5] | Li, M.; Guo, W. S.; Wen, L. R.; Yang, H. Z. Chin. J. Org. Chem. 2006, 26, 1192. (in Chinese) |
| [5] | (李明, 郭维斯, 文丽荣, 杨华铮, 有机化学, 2006, 26, 1192.) |
| [6] | Stanovnik, B. Eur. J. Org. Chem. 2019, 5120. |
| [7] | (a) Cao, S.; Jing, Y. F.; Liu, Y. Y.; Wan, J. P. Chin. J. Org. Chem. 2014, 34, 876. (in Chinese) |
| [7] | (曹硕, 景艳锋, 刘云云, 万结平, 有机化学, 2014, 34, 876.) |
| [7] | (b) Fu, L.; Wan, J. P. Asian J. Org. Chem. 2019, 8, 767. |
| [7] | (c) Wang, Z.; Zhao, B.; Liu, Y. Y.; Wan, J. P. Adv. Synth. Catal. 2022, 364, 1508. |
| [7] | (d) Chen, X. Y.; Zhang, X. T.; Wan, J. P. Org. Biomol. Chem. 2022, 20, 2356. |
| [8] | Xia, X. F.; Niu, Y. N. Org. Biomol. Chem. 2022, 20, 282. |
| [9] | Gaber, H. M.; Bagley, M. C.; Muhammad, Z. A.; Gomha, S. M. RSC Adv. 2017, 7, 14562. |
| [10] | Yu, Y. Y.; Niphakis, M. J.; Georg, G. I. Org. Lett. 2011, 13, 5932. |
| [11] | Fu, L.; Xu, Z.; Wan, J. P.; Liu, Y. Y. Org. Lett. 2020, 22, 9518. |
| [12] | Fu, L.; Liu, Y. Y.; Wan. J. P. Org. Lett. 2021, 23, 4363. |
| [13] | Wan, J. P.; Pan, J. Y. Chem. Commun. 2009, 2768. |
| [14] | Joussot, J.; Schoenfelder, A.; Larquetoux, L.; Nicolas, M.; Suffert, J.; Blond, G. Synthesis 2016, 48, 3364. |
| [15] | Lin, Y. F.; Fong, C.; Peng, W. L.; Tang, K. C.; Liang, Y. E.; Li. W. T. J. Org. Chem. 2017, 82, 10855. |
| [16] | Akram, M. O.; Patil, N. T. Org. Lett. 2019, 21, 8101. |
| [17] | Akram, M. O.; Bera, S.; Patil, N. T. Chem. Commun. 2016, 52, 12306. |
| [18] | (a) Würtz, S.; Rakshit, S.; Neumann, J. J.; Dr?ge, T.; Glorius, F. Angew. Chem., Int Ed. 2008, 47, 7230. |
| [18] | (b) Neumann, J. J.; Rakshit, S.; Dr?ge, T.; Würtz, S.; Glorius, F. Chem.-Eur. J. 2011, 17, 7298. |
| [18] | (c) Bernini, R.; Fabrizi, G.; Sferrazza, A.; Cacchi, S. Angew. Chem., Int. Ed. 2009, 48, 8078. |
| [18] | (d) Guan, Z.-H.; Yan, Z.-Y.; Ren, Z.-H.; Liu, X.-Y.; Liang, Y.-M. Chem. Commun. 2010, 46, 2823. |
| [18] | (e) Lian, X. L.; Ren, Z. H.; Wang, Y. Y.; Guan, Z. H. Org. Lett. 2014, 16, 3360. |
| [19] | (a) Zoller, J.; Fabry, D. C.; Ronge, M. A.; Rueping, M. Angew. Chem., Int. Ed. 2014, 53, 13264. |
| [19] | (b) Wu, C.; Meng, Q.; Lei, T.; Zhong, J.; Liu, W.; Zhao, L.; Li, Z.; Chen, B.; Tung, C.; Wu, L.-Z. ACS Catal. 2016, 6, 4635. |
| [19] | (c) Liu, W.; Lei, T.; Song, Z.; Yang, X.; Wu, C.; Jiang, X.; Chen, B.; Tung, C.; Wu, L.-Z. Org. Lett. 2017, 19, 3251. |
| [20] | Tang, S.; Gao, X.; Lei, A. Chem. Commun. 2017, 53, 3354. |
| [21] | (a) Yu, W.; Du, Y.; Zhao, K. Org. Lett. 2009, 11, 2417. |
| [21] | (b) Jia, Z.; Nagano, T.; Li, X.; Chan, A. S. C. Eur. J. Org. Chem. 2013, 858. |
| [21] | (c) He, Z.; Liu, W.; Li, Z. Chem. Asian J. 2011, 6, 1340. |
| [21] | (d) Drouhin, P.; Taylor, R. J. K. Eur. J. Org. Chem. 2015, 2333. |
| [21] | (e) Hu, F.; Zhao, S.; Chen, H.; Yu, S.; Xu, X. Yuan, W.; Zhang, X. ChemistrySelect 2017, 2, 1409. |
| [21] | (f) Zhao, L.; Qiu, C.; Zhao, L.; Yin, G.; Li, F.; Wang, C.; Li, Z. Org. Biomol. Chem. 2021, 19, 5377. |
| [22] | Liu, L.; Wang, Q.; Zhang, Z.; Zhang, Q.; Du, Z.; Xue, D.; Wang, T. Mol. Diversity 2014, 18, 777. |
| [23] | Wan, J.; Tu, Z.; Wang, Y. Chem.-Eur. J. 2019, 28, 6907. |
| [24] | Mkrtchyan, S.; Iaroshenko, V. O. Chem. Commun. 2020, 56, 2606. |
| [25] | Ge, H.; Niphakis, M. J.; Georg, G. I. J. Am. Chem. Soc. 2008, 130, 3708. |
| [26] | Yu, Y. Y.; Bi, L.; Georg, G. I. J. Org. Chem. 2013, 78, 6163. |
| [27] | Demina, D. Y.; Myannika, K. A.; Ermolichb, P. A.; Krayushkin, M. M.; Yarovenko, V. N. Mendeleev Commun. 2018, 28, 485. |
| [28] | Gu, Z. Y.; Zhu, T. H.; Cao, J. J.; Xu, X. P.; Wang, S. Y.; Ji, S. J. ACS Catal. 2014, 4, 49. |
| [29] | Hu, W. G.; Zheng, J.; Li, M.; Wu, W. Q.; Liu, H. Y.; Jiang, H. F. Chin. J. Chem. 2018, 36, 712. |
| [30] | Jiang, H.; Huang, W.; Yu, Y.; Yi, S.; Li, J.; Wu, W. Chem. Commun. 2017, 53, 7473. |
| [31] | Xiang, H.; Zhao, Q.; Tang, Z.; Xiao, J.; Xia, P.; Wang, C.; Yang, C.; Chen, X.; Yang, H. Org. Lett. 2017, 19, 146. |
| [32] | Sun, J.; Zhen, X.; Ge, H.; Zhang, G.; An, X.; Du. Y. Beilstein J. Org. Chem. 2018, 14, 1452. |
| [33] | Yu, Q.; Liu, Y. Y.; Wan, J. P. Org. Chem. Front. 2020, 7, 2770. |
| [34] | (a) Liu, X.; Cheng, R.; Zhao, F. F.; Zhang-Negrerie, D.; Du, Y. F.; Zhao, K. Org. Lett. 2012, 14, 5480. |
| [34] | (b) Zhao, F. F.; Liu, X.; Qi, R.; Zhang-Negrerie, D.; Huang, J.; Du, Y. F.; Zhao, K. J. Org. Chem. 2011, 76, 10338. |
| [35] | Wang, F.; Sun, W.; Wang, Y.; Jiang, Y.; Loh, T. P. Org. Lett. 2018, 20, 1256. |
| [36] | Guo, Y.; Xiang, Y.; Wei, L.; Wan, J. P. Org. Lett. 2018, 20, 3971 |
| [37] | Duan, X. Y.; Kong, X.; Zhao, X.; Yang, K.; Zhou, H. Y.; Zhou, D. J.; Zhang, Y.; Liu, J.; Ma, J. Y.; Liu, N.; Wang, Z. Tetrahedron Lett. 2016, 57, 1446. |
| [38] | Sun, X. Q.; Lyu, Y.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. Org. Lett. 2013, 15, 6222. |
| [39] | Yuan, J.; Zhang, Q.; Yu, M.; Huang, P.; Zhang, R.; Dong. D. Org. Lett. 2015, 17, 5012. |
| [40] | Yuan, Y.; Hou, W.; Zhang-Negrerie, D.; Zhao, K.; Du, Y. Org. Lett. 2014, 16, 5410. |
| [41] | Zheng, C.; Wang, Y.; Fan, R. Org. Lett. 2015, 17, 916. |
| [42] | Luo, T.; Wan, J. P.; Liu. Y. Y. Org. Chem. Front. 2020, 7, 1107. |
| [43] | Duan, X. Y.; Li, H. M.; Li, W. N.; Wang, J. Q. ChemistrySelect 2021, 6, 6478. |
| [44] | Wang, Z. W.; Zheng, Y.; Qian, Y. E.; Guan, J. P.; Lu, W. D.; Yuan, C. P.; Xiao, J. A.; Chen, K.; Xiang, H. Y.; Yang, H. J. Org. Chem. 2022, 87, 1477. |
| [45] | (a) Gammill, R. B. Synthesis 1979, 901. |
| [45] | (b) Mutai, P.; Pavadai, E.; Wiid, I.; Ngwane, A.; Baker, B.; Chibale, K. Bloorg. Med. Chem. Lett. 2015, 25, 2510. |
| [45] | (c) Biegasiewicz, K. F.; Gordon IV, J. S.; Rodriguez, D. A.; Priefer, R. Tetrahedron Lett. 2014, 55, 5210. |
| [46] | Miliutina, M.; Janke, J.; Hassan, S.; Zaib, S.; Iqbal, J.; Lecka, J.; Sévigny, J.; Villinger, A.; Friedrich, A.; Lochbrunner, S.; Langer, P. Org. Biomol. Chem. 2018, 16, 717. |
| [47] | Zhang, Y.; Zhao, X.; Zhuang, C.; Wang, S.; Zhang-Negrerie, D.; Du, Y. Adv. Synth. Catal. 2018, 360, 2107. |
| [48] | Zhao, Q. L.; Xia, P. J.; Zheng, L.; Xie, Z. Z.; Hu, Y. Z.; Chen, G. J.; Chen, X. Q.; Xiang, H. Y.; Yang, H. Tetrahedron 2020, 76, 130833. |
| [49] | Sorabad, G. S.; Maddani, M. R. New J. Chem. 2019, 43, 6563. |
| [50] | Lin, Y.; Wan, J. P.; Liu, Y. Y. New J. Chem. 2020, 44, 8120. |
| [51] | Lin, Y.; Jin, J.; Wang, C.; Wan, J. P.; Liu, Y. Y. J. Org. Chem. 2021, 86, 12378. |
| [52] | Wan, J. P.; Zhong, S.; Xie, L.; Cao, X.; Liu, Y. Y.; Wei, L. Org. Lett. 2016, 18, 584. |
| [53] | Sun, J.; Zhang-Negrerie, D.; Du, Y. Adv. Synth. Catal. 2016, 358, 2035. |
| [54] | Shang, Z.; Chen, Q.; Xing, L.; Zhang, Y.; Wait, L.; Du, Y. Adv. Synth. Catal. 2019, 361, 4926. |
| [55] | Jiang, Y.; Liang, G.; Zhang, C.; Loh, T.-P. Eur. J. Org. Chem. 2016, 3326. |
| [56] | (a) Rafique, J.; Saba, S.; Schneider, A. R.; Franco, M. S.; Silva, S. M.; Braga, A. L. ACS Omega 2017, 2, 2280. |
| [56] | (b) Zhong, S. S.; Liu, Y. Y.; Cao, X. J.; Wan, J. P. ChemCatChem 2017, 9, 465. |
| [57] | Guo, Y. H.; Zhong, S. S.; Wei, L.; Wan, J. P. Beilstein J. Org. Chem. 2017, 13, 2017. |
| [58] | Gao, Y.; Wei, L.; Liu, Y. Y.; Wan, J. P. Org. Biomol. Chem. 2017, 15, 4631. |
| [59] | Siddaraju, Y.; Prabhu, K. R. J. Org. Chem. 2017, 82, 3084. |
| [60] | Gao, Y.; Hu, C.; Wen, C.; Wan, J. P. ACS Omega 2017, 2, 7784. |
| [61] | Zhang, T.; Yao, W.; Wan, J. P.; Liu, Y. Y. Adv. Synth. Catal. 2021, 363, 4811. |
| [62] | Wan, J. P.; Zhong, S.; Guo, Y.; Wei, L. Eur. J. Org. Chem. 2017, 4401. |
| [63] | Guo, Y.; Wang, G.; Li, W.; Wan, J. P. J. Org. Chem. 2019, 84, 2984. |
| [64] | Yu, Q.; Liu, Y. Y.; Wan, J. P. Chin. Chem. Lett. 2021, 32, 3514. |
| [65] | Zhang, X. Z.; Ge, D. L.; Chen, S. Y.; Yu, X. Q. RSC Adv. 2016, 6, 66320. |
| [66] | Yang, Z.; Li, L.; Yang, T.; Zhou, C. J. Org. Chem. 2019, 84, 16262. |
| [67] | Gao, Y.; Liu, Y. Y.; Wan, J. P. J. Org. Chem. 2019, 84, 2243. |
| [68] | Duan, X. Y.; Liu, X.; Cuan, X.; Wang, L.; Liu, K.; Zhou, H.; Chen, X.; Li, H.; Wang, J. J. Org. Chem. 2019, 84, 12366. |
| [69] | Chandran, R.; Rahul, D.; Suman, V.; Tiwari, K. N. Org. Lett. 2020, 22, 6557. |
| [70] | Xiang, H.; Yang, C. Org. Lett. 2014, 16, 5686. |
| [71] | Fu, L.; Liu, Y. Y.; Wan, J. P. Chem. Commun. 2022, 58, 1808. |
/
| 〈 |
|
〉 |