

可见光促进的含色氨酸寡肽与YlideFluor试剂的直接三氟甲基化反应研究
收稿日期: 2022-09-14
修回日期: 2022-11-14
网络出版日期: 2022-11-22
基金资助
国家重点研发计划(2021YFF0701700)
Visible-Light-Promoted Direct Trifluoromethylation of Tryptophan-Containing Oligapeptides with YlideFluor
Received date: 2022-09-14
Revised date: 2022-11-14
Online published: 2022-11-22
Supported by
National Key Research and Development Program of China(2021YFF0701700)
刘静 , 郝健 , 沈其龙 . 可见光促进的含色氨酸寡肽与YlideFluor试剂的直接三氟甲基化反应研究[J]. 有机化学, 2023 , 43(4) : 1517 -1524 . DOI: 10.6023/cjoc202209018
A photosensitizer-free visible-light-promoted method for site-selective trifluoromethylation of tryptophan- containing oligopeptides using YlideFluor as the trifluoromethyl radical source under mild conditions was developed. Mechanistic studies showed that a donor-acceptor between 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) and YlideFluor is generated. Upon irradiation with blue LEDs, the S—CF3 bond in the adduct undergoes homolytic cleavage to give trifluoromethyl radical, which attacks the electron-rich C-2 position of the indole moiety in the tryptophan-containing oligopeptide.
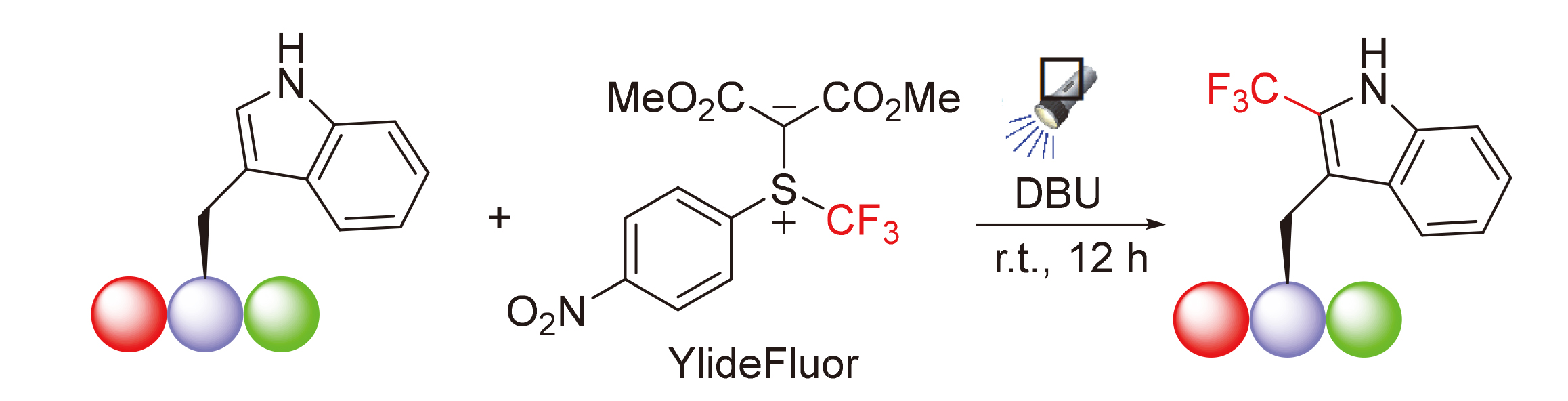
Key words: fluorine; trifluoromethyl; tryptophan; oligapeptide; visible light
| [1] | (a) Erak, M.; Bellmann-Sickert, K.; Els-Heindl, S.; Beck-Sickinger, A. G. Bioorg. Med. Chem. 2018, 26, 2759. |
| [1] | (b) Kaspar, A. A.; Reichert, J. M. Drug Discovery Today 2013, 18, 807. |
| [1] | (c) Malonis, R. J.; Lai, J. R.; Vergnolle, O. Chem. Rev. 2020, 120, 3210. |
| [2] | (a) Reddy, B..Jow T.; Hantash, B. M. Exp. Dermatol. 2012, 21, 563. |
| [2] | (b) Ji, Y.-J.; Qiao, H.-Z.; He, J.-Y.; Li, W.-D.; Chen, R.; Wang, J.-J.; Wu, L.; Hu, R.-F.; Duan, J.-N.; Chen, Z.-P. J. Drug Target. 2017, 25, 597. |
| [3] | deGruyter, J. N.; Malins, L. R.; Baran, P. S. Biochemistry 2017, 56, 3863. |
| [4] | Chalker, J. M.; Bernardes, G. J. L.; Lin, Y. A.; Davis, B. G. Chem. Asian J. 2009, 4, 630. |
| [5] | (a) Ban, H.; Gavrilyuk, J.; Barbas III, C. F. J. Am. Chem. Soc. 2010, 132, 1523. |
| [5] | (b) Ban, H.; Nagano, M.; Gavrilyuk, J.; Hakamata, W.; Inokuma, T.; Barbas III, C. F. Bioconjugate Chem. 2013, 24, 520. |
| [6] | Ohata, J.; Minus, M. B.; Abernathy, M. E.; Ball, Z. T. J. Am. Chem. Soc. 2016, 138, 7472. |
| [7] | (a) Ruiz-Rodriguez, J.; Albericio, F.; Lavilla, R. Chem.-Eur. J. 2010, 16, 1124. |
| [7] | (b) Seki, Y.; Ishiyama, T.; Sasaki, D.; Abe, J.; Sohma, Y.; Oisaki, K.; Kanai, M. J. Am. Chem. Soc. 2016, 138, 10798. |
| [7] | (c) Hansen, M. B.; Hubalek, F.; Skrydstrup, T.; Hoeg-Jensen, T. Chem.-Eur. J. 2016, 22, 1572. |
| [7] | (d) Ruan, Z.; Sauermann, N.; Manoni, E.; Ackermann, L. Angew. Chem., Int. Ed. 2017, 56, 3172. |
| [8] | Guerrero, I.; Correa, A. Asian J. Org. Chem. 2020, 9, 898. |
| [9] | (a) Leo, A.; Hansch, C.; Elkins, D. Chem. Rev. 1971, 71, 525. |
| [9] | (b) Hansch, C.; Leo, A.; Taft, R. W. Chem. Rev. 1991, 91, 165. |
| [9] | (c) Qing, F.-L.; Liu, X.-Y.; Ma, J.-A.; Shen, Q.; Song, Q.-L.; Tang, P.-P. CCS Chem. 2022, 4, 2518. |
| [10] | Wang, Y.-X.; Wang, J.-H.; Li, G.-X..He G. ; Chen, G. Org. Lett. 2017, 19, 1442. |
| [11] | Imiolek, M.; Karunanithy, G.; Ng, W.-L.; Baldwin, A. J.; Gourverneur, V.; Davis, B. G. J. Am. Chem. Soc. 2018, 140, 1568. |
| [12] | Ding, B.; Weng, Y.; Liu, Y.-Q.; Song, C.-L.; Yin, L.; Yuan, J.-F.; Ren, Y.-R.; Lei, A.-W.; Chiang, C.-W. Eur. J. Org. Chem. 2019, 7596. |
| [13] | Guerrero, I.; Correa, A. Org. Lett. 2020, 22, 1754. |
| [14] | Kee, C. W.; Tack, O.; Guibbal, F.; Wilson, T. C.; Iseneggar, P. G.; Imiolek, M.; Verhoog, S.; Tilby, M.; Boscutti, G.; Ashworth, S.; Chupin, J.; Kashani, R.; Poh, A. W. J.; Sosabowski, J. K.; Macholl, S.; Plisson, C.; Cornelissen, B.; Willis, M. C.; Passchier, J.; Davis, B. G.; Gouverneur, V. J. Am. Chem. Soc. 2020, 142, 1180. |
| [15] | Liu, Y.-F.; Shao, X.-X.; Lu, L.; Shen, Q. Org. Lett. 2015, 17, 2752. |
| [16] | (a) Liu, Y.-F.; Ling, Y.-J.; Ge, H.-M.; Lu, L.; Shen, Q. Chin. J. Chem. 2021, 39, 1667. |
| [16] | (b) Yao, R.-C.; Ling, Y.-J.; Chen, W.-B.; Lu, L.; Shen, Q. Org. Process Res. Dev. 2022, 26, 299. |
| [17] | (a) Ge, H.-M.; Shen, Q. Org. Chem. Front. 2019, 10, 2205. |
| [17] | (b) Ge, H.-M.; Wu, B.-T.; Liu, Y.-F.; Wang, H.-Y.; Shen, Q. ACS Catal. 2020, 10, 12414. |
| [17] | (c) Yao, R.-C.; Chen, W.-B.; Shen, Q. Chin. J. Org. Chem. 2021, 41, 2684. (in Chinese) |
| [17] | (姚瑞超, 陈文博, 沈其龙, 有机化学, 2021, 41, 2684.) |
| [18] | Cheng, Y. Z.; Yuan, X. G.; Ma, J.; Yu, S. Y. Chem.-Eur. J. 2015, 21, 8355. |
/
| 〈 |
|
〉 |