

吡唑化提高喹喔啉酮敏化效率: 氮杂环丁烷的合成及进一步开环反应
收稿日期: 2022-09-21
修回日期: 2022-10-21
网络出版日期: 2022-11-22
基金资助
国家自然科学基金(21871123)
Improving the Efficiency of Sensitized Quinoxlin-2(1H)-ones via Pyrazolylation: Synthesis of Azetidines and Further Ring-Opening Reaction
Received date: 2022-09-21
Revised date: 2022-10-21
Online published: 2022-11-22
Supported by
National Natural Science Foundation of China(21871123)
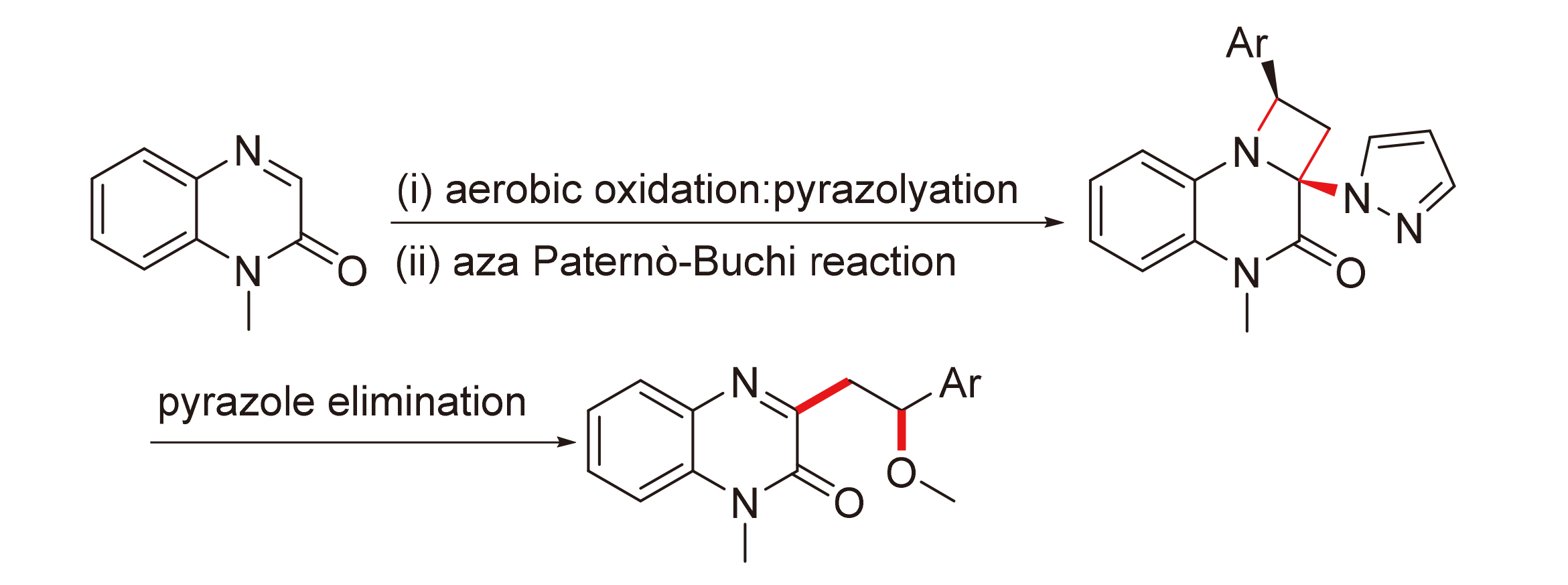
在温和条件下利用吡唑作为合适的辅助基促进喹喔啉酮类化合物被光催化剂敏化, 通过分子间aza Paternò-Büchi反应制备功能化的氮杂环丁烷. 总体而言, 吡唑基具有降低喹喔啉酮类化合物的三线态激发态能量、改变氧化还原电位及作为良好的离去基进行后续转化等关键作用. 研究涉及交叉脱氢偶联(CDC)-胺化、aza Paternò-Büchi反应和消除反应.
关键词: 可见光催化; 需氧氧化; 光环加成; aza Paternò-Büchi反应; 能量转移
赵成军 , 白治琴 , 何建 , 刘强 . 吡唑化提高喹喔啉酮敏化效率: 氮杂环丁烷的合成及进一步开环反应[J]. 有机化学, 2022 , 42(12) : 4300 -4314 . DOI: 10.6023/cjoc202209028
A mild approach to prepare functionalized azetidines by intermolecular aza Paternò-Büchi reaction using suitable auxochrome (pyrazole) to promote 3-pyrazolyl quinoxalin-2(1H)-ones into its excited state has been developed. Overall, pyrazolyl plays a key role in reducing the excited state energy of the triplet state of quinoxalinones, changing the redox potential and subsequent transformation as a good leaving group. The cross-dehydrogenative coupling (CDC)-amination, aza Paternò-Büchi reaction, and elimination reaction were also investigated.

| [1] | Richardson, A. D.; Becker, M. R.; Schindler, C. S. Chem. Sci. 2020, 11, 7553. |
| [2] | (a) Tsuge, O.; Tashiro, M.; Oe, K. Tetrahedron Lett. 1968, 9, 3971. |
| [2] | (b) Koch, T. H.; Howard, K. H. Tetrahedron Lett. 1972, 13, 4035. |
| [2] | (c) Koch, T. H.; Rodehorst, R. M. Tetrahedron Lett. 1972, 13, 4039. |
| [2] | (d) Howard, K. A.; Koch, T. H. J. Am. Chem. Soc. 1975, 97, 7288. |
| [2] | (e) Rodehorst, R. M.; Koch, T. H. J. Am. Chem. Soc. 1975, 97, 7298. |
| [2] | (f) Sa-kamoto, R.; Inada, T.; Sakurai, S.; Maruoka, K. Org. Lett. 2016, 18, 6252. |
| [3] | (a) Padwa, A. Chem. Rev. 1977, 77, 37. |
| [3] | (b) Pratt, A. C. Chem. Soc. Rev. 1977, 6, 63. |
| [3] | (c) Padwa, A.; Albrecht, F. J. Am. Chem. Soc. 1972, 94, 1000. |
| [3] | (d) Kandappa, S. K.; Valloli, L. K.; Ahuja, S.; Parthiban, J. Chem. Soc. Rev. 2021, 50, 1617. |
| [4] | Becker, M. R.; Wearing, E. R.; Schindler, C. S. Nat. Chem. 2020, 12, 898. |
| [5] | (a) Hyatt, J. A.; Swenton, J. S. J. Chem. Soc., Chem. Commun. 1972, 1144. |
| [5] | (b) Swenton, J. S.; Hyatt, J. A. J. Am. Chem. Soc. 1974, 96, 4879. |
| [5] | (c) Swenton, J. S.; Balchunis, R. J. J. Heterocycl. Chem 1974, 11, 917. |
| [5] | (d) Kumagai, T.; Kawamura, Y.; Mukai, T. Chem. Lett. 1983, 12, 1357. |
| [5] | (e) Kawamura, Y.; Kumagai, T.; Murai, T. Chem. Lett. 1985, 14, 1937. |
| [5] | (f) Becker, M. R.; Wearing, E. R.; Schindler, C. S. Nat. Chem. 2020, 12, 898. |
| [5] | (g) Futamura, S.; Ohta, H.; Kamiya, Y. Chem. Lett. 1980, 9, 655. |
| [5] | (h) Nishio, T. J. Org. Chem. 1984, 49, 827. |
| [5] | (i) Nishio, T.; Omote, Y. J. Org. Chem. 1985, 50, 1370. |
| [5] | (j) Nishio, T.; Omote, Y. J. Chem. Soc., Perkin Trans. 1 1987, 2611. |
| [5] | (k) Nishio, T. J. Chem. Soc., Perkin Trans. 1 1990, 565. |
| [5] | (l) Katagiri, N.; Kasai, K.; Kaneko, C. Chem. Pharm. Bull. 1986, 34, 4429. |
| [5] | (m) Li, X.; Gro?kopf, J.; Jandl, C.; Bach, T. Angew. Chem., Int. Ed. 2021, 60, 2684. |
| [6] | Kumarasamy, E.; Kandappa, S. K.; Raghunathan, R.; Jockusch, S.; Sivaguru, J. Angew. Chem., Int. Ed. 2017, 56, 7056. |
| [7] | Becker, M. R.; Richardson, A. D.; Schindler, C. S. Nat. Commun. 2019, 10, 5095. |
| [8] | Zhu, M.; Zhang, X.; Zheng, C.; You, S.-L. ACS Catal. 2020, 10, 12618. |
| [9] | (a) Wei, W.; Wang, L.; Bao, P.; Shao, Y.; Yue, H.; Yang, D.; Yang, X.; Zhao, X.; Wang, H. Org. Lett. 2018, 20, 7125. |
| [9] | (b) Li, K.-J.; Xu, K.; Liu, Y.-G.; Zeng, C.-C.; Sun, B.-G. Adv. Synth. Catal. 2019, 361, 1033. |
| [10] | Luo, J.; Zhang, J. ACS Catal. 2016, 6, 873. |
| [11] | Sun, M.; Wang, L.; Zhao, L.; Wang, Z.; Li, P. ChemCatChem 2020, 12, 5261. |
| [12] | Niu, K.; Ding, L.; Zhou, P.; Hao, Y.; Liu, Y.; Song, H.; Wang, Q. Green Chem. 2021, 23, 3246. |
/
| 〈 |
|
〉 |