

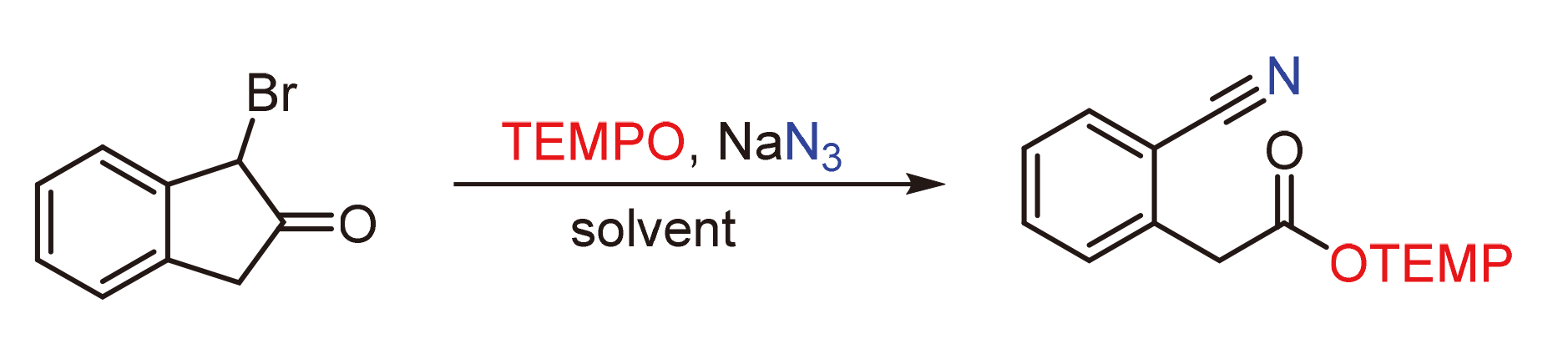
无过渡金属催化的α-溴代茚酮自由基裂解反应合成(2-氰基苯基)乙酸-2,2,6,6-四甲基哌啶酯
收稿日期: 2022-08-05
修回日期: 2022-10-31
网络出版日期: 2022-12-28
基金资助
宁夏自然科学基金(2021AAC03100)
Synthesis of 2,2,6,6-Tetramethylpiperidin-1-yl-2-(2-cyanophenyl)-acetate by Transition Metal-Free Radical Cleavage Reaction from α-Bromoindanone
Received date: 2022-08-05
Revised date: 2022-10-31
Online published: 2022-12-28
Supported by
Ningxia Natural Science Foundation(2021AAC03100)
秦娇 , 陈杰 , 苏艳 . 无过渡金属催化的α-溴代茚酮自由基裂解反应合成(2-氰基苯基)乙酸-2,2,6,6-四甲基哌啶酯[J]. 有机化学, 2023 , 43(6) : 2171 -2177 . DOI: 10.6023/cjoc202208006
In this paper, the synthesis of 2,2,6,6-tetramethylpiperidin-1-yl-2-(2-cyanophenyl)acetate from bromoindanone with sodium azide and 2,2,6,6-tetramethylpiperidine nitrogen oxide was reported. The carbon-carbon bond cleavage of bromoindenone was realized with transition metal-free catalysis and a series of aryl 2-cyano-phenylacetate compounds were obtained. This method was an expansion of cyanophenylacetate’ synthesis, and the reaction mechanism was verified by computational chemistry. The experiment was carried out under mild conditions with transition metal-free catalysis and with broad substrate scope.
| [1] | Fleming, F. F. Nat. Prod. Rep. 1999, 16, 597. |
| [2] | Wang, Y. X.; Du, Y. F.; Huang, N. Future Med. Chem. 2018, 10, 1756. |
| [3] | (a) Fleming, F.F.; Yao, L.; avikumar, P.C.; Funk, L. B.; Shook, C. J. Med. Chem. 2010, 53, 7902. |
| [3] | (b) Steve, S. Molecules 2020, 25, 4495. |
| [4] | Patai, S.; Rappaport, Z. Preparation and Synthetic Applications of Cyano Compounds, Wiley, New York, 1983, pp. 1064-1272. |
| [5] | Talukdar, R. New J. Chem. 2020, 44, 5303. |
| [6] | Chemat, F. Tetrahedron Lett. 2000, 41, 3855. |
| [7] | (a) Talukdar, R. New J. Chem. 2020, 44, 5303. |
| [7] | (b) Ha, J. H.; Ahn, J. H.; An, D. K. Bull. Korean Chem. Soc. 2006, 27, 121. |
| [8] | Tamura, M.; Tonomura, T; Shimizu, K.; Satsuma, A. Green Chem. 2012, 14, 984. |
| [9] | Srimani, D.; Feller, M.; Ben-David, Y.; Milstein, D. Chem. Commun. 2012, 48, 11853. |
| [10] | Yeung, K.-S.; Farkas, M. E.; Kadow, J. F.; Meanwell, N. A. Tetrahedron Lett. 2005, 46, 3429. |
| [11] | Horneff, T.; Chuprakov, S.; Chernyak, N.; Gevorgyan, V.; Fokin, V. V. J. Am. Chem. Soc. 2008, 130, 14972. |
| [12] | Haddenham, D.; Pasumansky, L.; DeSoto, J.; Eagon, S.; Singaram, B. J. Org. Chem. 2009, 74, 1964. |
| [13] | Burns, A. R.; Kerr, J. H.; Kerr, W. J.; Passmore, J.; Paterson, L. C.; Watson, A. J. B. Org. Biomol. Chem. 2010, 8, 2777. |
| [14] | Yu, X.-Y.; Chen, J.-R.; Xiao, W.-J. Chem. Rev. 2021, 121, 506. |
| [15] | Ding, R.; Liu, Y.; Han, M.; Jiao, W.; Li, J.; Tian, H.; Sun, B. J. Org. Chem. 2018, 83, 12939. |
| [16] | (a) Wang, P. Z.; Yu, X. Y.; Xiao, W. J. Chem. Commun. 2018, 54, 9925. |
| [16] | (b) Aggarwal, V. K.; Olofsson, B. Angew. Chem., Int. Ed. 2005, 44, 5516. |
| [16] | (c) Anand, D.; He, Y.-W.; Zhou, L. Org. Biomol. Chem. 2019, 17, 533. |
| [16] | (d) Yuan, Y.; Dong, W.-H.; Zhang, Z.-G. Chem. Commun. 2018, 54, 9925. |
| [16] | (e) Zhao, B. L; Shi, Z. Z. Angew. Chem., Int. Ed. 2017, 56, 12727. |
| [17] | (a) Bencivenni, G.; Lanza, T.; Leardini, R. Minozzi, M.; Nanni, D; Spagnolo, P; Zanardi, G. J. Org. Chem. 2008, 73, 4721. |
| [17] | (b) Muriel, B.; Waser, J. Angew. Chem. 2021, 133, 4121. |
| [18] | Chiba, S.; Zhang, L.; Ang, G. Y.; Benjamin, W. H. Org. Lett. 2010, 12, 2052. |
| [19] | Muriel, B.; Waser, J. Angew Chem., Int. Ed. 2021, 60, 4075. |
| [20] | Wang, T.; Jiao, N. J. Am. Chem. Soc. 2013, 135, 11692. |
| [21] | Jackman, M. M.; Im, S.; Castle, S. L. Chem. Eur. J. 2018, 24, 594. |
| [22] | Li, M. F.; Shi, S.Q.; Xu, T.; Zhang, Q.; Hao, W. J.; Wang, S. L.; Wang, J. Y.; Tu, S. J.; Jiang, B. Chin. Chem. Lett. 2023, 34, 107751. |
| [23] | The optimized structures were calculated at the B3LYP level using the hybrid DFT functional implemented in the Gaussian 09 software package. The 6-311G(d, p) basis set for the other atoms (H, C, N and Br): Jr., Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Keith, T.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, O.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09, Revision D.01, Wallingford CT, 2013. |
| [24] | Shi, W.; Yang, C.; Guo, L.; Xia, W. Org. Chem. Front. 2022, 9, 6513. |
/
| 〈 |
|
〉 |