

银介导的N-芳基丙烯酰胺串联环化反应研究进展
收稿日期: 2022-11-02
修回日期: 2022-11-16
网络出版日期: 2023-01-05
基金资助
国家自然科学基金(21801023); 湖南省自然科学基金(2021JJ40563); 长沙市科技局(kq2004070)
Recent Progress in Silver-Mediated Tandem Cyclization of N-Arylacrylamides
Received date: 2022-11-02
Revised date: 2022-11-16
Online published: 2023-01-05
Supported by
National Natural Science Foundation of China(21801023); Hunan Provincial Natural Science Foundation(2021JJ40563); Changsha Municipal Science and Technology Project(kq2004070)
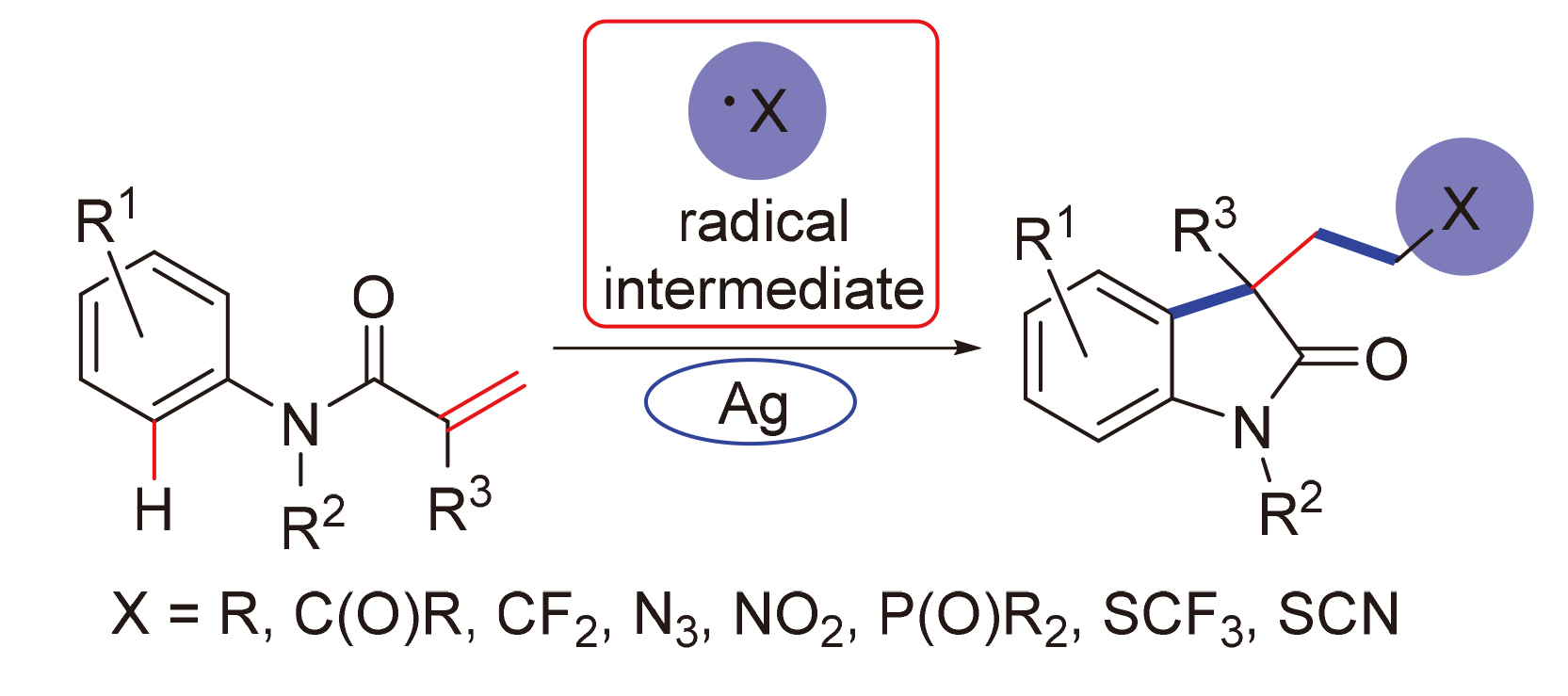
作为明星分子, N-芳基丙烯酰胺可发生自由基加成串联环化反应, 从而高效构建C(3)-二取代吲哚-2-酮骨架. 吲哚-2-酮类化合物是一类重要的杂环化合物, 具有广泛的生物活性, 一直是有机化学和药物化学领域的研究热点. 银催化剂具有独特的催化活性, 毒性低, 在有机合成中的应用愈加广泛. 从不同的自由基前体及成键类型(C—C、C—N、 C—P、C—S)出发, 对银介导的N-芳基丙烯酰胺串联环化反应研究进展进行了综述, 并对代表性底物及反应机理进行了归纳和讨论.
关键词: 银介导反应; N-芳基丙烯酰胺; 串联环化反应; C(3)-二取代吲哚-2-酮
任志军 , 罗维纬 , 周俊 . 银介导的N-芳基丙烯酰胺串联环化反应研究进展[J]. 有机化学, 2023 , 43(6) : 2026 -2039 . DOI: 10.6023/cjoc202211003
As a star molecule, N-arylacrylamides can undergo tandem radical addition/cyclization to efficiently construct C(3)-disubstituted indolin-2-ones. Indolin-2-ones are a class of important heterocyclic compounds with a wide range of biological activities, and the synthesis of which has been a research hotspot in the field of organic chemistry and medicinal chemistry. Silver catalysts are low in toxicity, and have unique catalytic activities, which are widely used in organic synthesis. In this paper, starting from different radical precursors and bond types (C—C, C—N, C—P, C—S), the research progress of silver-mediated synthesis of C(3)-disubstituted indolin-2-ones via tandem radical addition/cyclization of N-arylacrylamides is reviewed, and the representative substrates and reaction mechanisms are also summarized and discussed.

| [1] | Dalpozzo, R. Org. Chem. Front. 2017, 4, 2063. |
| [2] | Kaur, M.; Singh, M.; Chadha, N.; Silakari, O. Eur. J. Med. Chem. 2016, 123, 858. |
| [3] | Millemaggi, A.; Taylor, R.-J.-K. Eur. J. Org. Chem. 2010, 4527. |
| [4] | Ding, Z.; Zhou, M.; Zeng, C. Arch. Pharm. 2020, 353, 1900367. |
| [5] | Luo, W.-M.; Yu, Q.-S.; Kulkarni, S.-S.; Parrish, D.-A.; Holloway, H.-W.; Tweedie, D.; Shafferman, A.; Lahiri, D.-K.; Brossi, A.; Greig, N.-H. J. Med. Chem. 2006, 49, 2174. |
| [6] | Yu, Q.-S.; Zhu, X.-X.; Holloway, H.-W.; Whittaker, N.-F.; Brossi, A.; Greig, N.-H. J. Med. Chem. 2002, 45, 3684. |
| [7] | Sumpter, W.-C. Chem. Rev. 1945, 37, 443. |
| [8] | Yamamoto, K.; Qureshi, Z.; Tsoung, J.; Pisella, G.; Lautens, M. Org. Lett. 2016, 18, 4954. |
| [9] | Jang, Y.-J.; Larin, E.-M.; Lautens, M. Angew. Chem., Int. Ed. 2017, 56, 11927. |
| [10] | (a) Rodríguez, J.-F.; Zhang, A.; Bajohr, J.; Whyte, A.; Mirabi, B.; Lautens, M. Angew. Chem., Int. Ed. 2021, 60, 18478. |
| [10] | (b) Marchese, A.-D.; Wollenburg, M.; Mirabi, B.; Abel-Snape, X.; Whyte, A.; Glorius, F.; Lautens, M. ACS Catal. 2020, 10, 4780. |
| [11] | Sperger, T.; Le, C.-M.; Lautens, M.; Schoenebeck, F. Chem. Sci. 2017, 8, 2914. |
| [12] | Marchese, A.-D.; Larin, E.-M.; Mirabi, B.; Lautens, M. Acc. Chem. Res. 2020, 53, 1605. |
| [13] | (a) Liu, Y.-J.; Lin, L.-Q.; Han, Y.-H.; Zhang, X. Chin. J. Org. Chem. 2019, 39, 2705. (in Chinese) |
| [13] | (刘颖杰, 林立青, 韩莹徽, 张鑫, 有机化学, 2019, 39, 2705.) |
| [13] | (b) Wang, L.-L.; Zhang, Z.-Y.; Han, H.-B.; Liu, X.-L.; Bu, Z.-W.; Wang, Q.-L. Chin. J. Org. Chem. 2021, 41, 12. (in Chinese) |
| [13] | (王乐乐, 张子莹, 韩华彬, 刘雄利, 卜战伟, 王琪琳, 有机化学, 2021, 41, 12.) |
| [13] | (c) Abdukader, A.; Zhang, Y.-H.; Zhang, Z.-P.; Liu, C.-J. Chin. J. Org. Chem. 2016, 36, 875. (in Chinese) |
| [13] | (阿布力米提?阿布都卡德尔, 张永红, 张增鹏, 刘晨江 有机化学, 2016, 36, 875.) |
| [13] | (d) Zhao, Y.; Lv, Y.; Xia, W. Chem. Rec. 2019, 19, 424. |
| [13] | (e) Singh, J.; Sharma, A. Adv. Synth. Catal. 2021, 363, 4284. |
| [14] | (a) Weibel, J.-M.; Blanc, A.; Pale, P. Chem. Rev. 2008, 108, 3149. |
| [14] | (b) álvarez-Corral, M.; Mu?oz-Dorado, M.; Rodríguez-Garcí, R. Chem. Rev. 2008, 108, 3174. |
| [14] | (c) Naodovic, M.; Yamamoto, H. Chem. Rev. 2008, 108, 3132. |
| [14] | (d) Fang, G.-C.; Cong, X.-F.; Zanoni, G.; Liu, Q.; Bi, X-H. Adv. Synth. Catal. 2017, 359, 1422. |
| [15] | (a) Mai, W.-P.; Wang, J.-T.; Yang, L.-R.; Yuan, J.-W.; Mao, P.; Xiao, Y.-M.; Qu, L.-B. Chin. J. Org. Chem. 2014, 34, 1958. (in Chinese) |
| [15] | (买文鹏, 王继涛, 杨亮茹, 袁金伟, 毛璞, 肖咏梅, 屈凌波, 有机化学, 2014, 34, 1958.) |
| [15] | (b) Chen, J.-R.; Yu, X.-Y.; Xiao, W.-J. Synthesis 2015, 47, 604. |
| [15] | (c) Song, R.-J.; Liu, Y; Xie, Y.-X.; Li, J.-H. Synthesis 2015, 47, 1195. |
| [15] | (d) Li, C.-C.; Yang, S.-D. Org. Biomol. Chem. 2016, 14, 4365. |
| [15] | (e) Wu, Y.-C.; Xiao, Y.-T.; Yang, Y.-Z.; Song, R.-J.; Li, J.-H.; ChemCatChem 2020, 12, 5312. |
| [16] | For recent reviews, see: (a) Baudoin, O. Angew. Chem. 2007, 119, 1395; Angew. Chem., Int. Ed. 2007, 46, 1373. |
| [16] | (b) Goo?en, L.-J.; Rodríguez, N.; Goo?en, K. Angew. Chem., Int. Ed. 2008, 47, 3100. |
| [16] | (c) Satoh, T.; Miura, M. Synthesis 2010, 3395. |
| [16] | (d) Rodríguez, N.; Goo?en, L.-J. Chem. Soc. Rev. 2011, 40, 5030. |
| [16] | (e) Shang, R.; Liu, L. Sci. China: Chem. 2011, 54,1670. |
| [16] | (f) Dzik, W.-I.; Lange, P.-P.; Goo?en, L.-J. Chem. Sci. 2012, 3, 2671. |
| [16] | (g) Cornella, J.; Larrosa, I. Synthesis 2012, 653. |
| [17] | Wang, H.; Guo, L.-N.; Duan, X.-H. Adv. Synth. Catal. 2013, 355, 2222. |
| [18] | Bai, Q.-F.; Jin, C.-G.; He, J.-Y.; Feng, G.-F. Org. Lett. 2018, 20, 2172. |
| [19] | Gao, Q.-S.; Jin, Q.; Chen, Y.; Sun, J.; Zhou, M.-D. Chin. J. Org. Chem. 2022, 42, 257. (in Chinese) |
| [19] | (高启升, 荆祺, 陈阳, 孙京, 周明东, 有机化学, 2022, 42, 257.) |
| [20] | Beck, E.; Grimster, N.; Hatley, R.; Gaunt, M. J. Am. Chem. Soc. 2006, 128, 2528. |
| [21] | Wang, H.; Guo, L.-N.; Duan, X.-H. Chem. Commun. 2013, 49, 10370. |
| [22] | Li, Y.-H.; Wang, J.-J.; Wei, X.-H.; Yang, S.-D. Chin. J. Org. Chem. 2015, 35, 638. (in Chinese) |
| [22] | (李永红, 王君姣, 魏小红, 杨尚东, 有机化学, 2015, 35, 638.) |
| [23] | Kanyiva, K.-S.; Makino, S.; Shibata, T. Chem. Asian J. 2018, 13, 496. |
| [24] | For selected book chapters and reviews, see: (a) Wang, J.; Sánchez-Rosellò, M.; Ace?a, J.-L.; Delpozo, C.; Sorochinsky, A.-E.; Fustero, S.; Soloshonok, V.-A.; Liu, H. Chem. Rev. 2014, 114, 2432. |
| [24] | (b) Gillis, E.-P.; Eastman, K.-J.; Hill, M.-D.; Donnelly, D.-J.; Meanwell, N.-A. J. Med. Chem. 2015, 58, 8315. |
| [24] | (c) Zhou, Y.; Wang, J.; Gu, Z.; Wang, S.; Zhu, W.; Ace?a, J.-L.; Soloshonok, V.-A.; Izawa, K.; Liu, H. Chem. Rev. 2016, 116, 422. |
| [25] | (a) Blackburn, G.-M.; England, D.-A.; Kolkmann, F. J. Chem. Soc., Chem. Commun. 1981, 930. |
| [25] | (b) Xu, Y.; Aoki, J.; Shimizu, K.; Umezu-Goto, M.; Hama, K.; Takanezawa, Y.; Yu, S.; Mills, G.-B.; Arai, H.; Qian, L.; Prestwich, G.-D. J. Med. Chem. 2005, 48, 3319. |
| [25] | (c) Meanwell, N.-A. J. Med. Chem. 2011, 54, 2529. |
| [26] | Wang, C.; Chen, Q.; Guo, Q.-P.; Liu, H.; Xu, Z.-Q.; Liu, Y.-B.; Wang, M.-R.; Wang, R. J. Org. Chem. 2016, 81, 5782. |
| [27] | Wang, X.-L.; Wan, W.; Chen, Y.-R.; Li, J.-L.; Jiang, H.-Z.; Wang, Y.; Deng, H.-M.; Hao, J. Eur. J. Org. Chem. 2016, 3773. |
| [28] | Wan, W.; Li, J.-L.; Ma, G.-B.; Chen, Y.-R.; Jiang, H.-Z.; Deng, H.-M.; Hao, J. Org. Biomol. Chem. 2017, 15, 5308. |
| [29] | (a) Middleton, W.-J.; Bingham, E.-M. J. Org. Chem. 1980, 45, 2883. |
| [29] | (b) Hagele, G.; Haas, A. J. Fluorine Chem. 1996, 76, 15. |
| [29] | (c) Lal, G.-S.; Pez, G.-P.; Pesaresi, R.-J.; Prozonic, F.-M.; Cheng, H. J. Org. Chem. 1999, 64, 7048. |
| [29] | (d) Singh, R.-P.; Majumder, U.; Shreeve, J.-M. J. Org. Chem. 2001, 66, 6263. |
| [29] | (e) Umemoto, T.; Singh, R.-P.; Xu, Y.; Saito, N. J. Am. Chem. Soc. 2010, 132, 18199. |
| [30] | Li, Y.-L.; Wang, J.-B.; Wang, X.-L.; Cao, Y.; Deng, J. Eur. J. Org. Chem. 2017, 6052. |
| [31] | For some examples, see: (a) Saha, G.; Khanapure, S.-P.; Powell, W.-S.; Rokach, J. Tetrahedron Lett. 2000, 41,6313. |
| [31] | (b) Kim, S.; Adiyaman, Y.; Saha, G.; Powell, W.-S.; Rokach, J. Tetrahedron Lett. 2001, 42,4445. |
| [31] | (c) Pinney, K.-G.; Mejia, M.-P.; Villalobos, V.-M.; Rosenquist, B.-E.; Petit, G.-R.; Verdier-Pinard, P.; Hamel, E. Bioorg. Med. Chem. 2000, 8, 2417. |
| [31] | (d) Dutta, A.-K.; Fei, X.-S.; Vaughan, R.-A.; Gaffaney, J.-D.; Wang, N.-N.; Lever, J.-R.; Reith, M.-E. Life Sci. 2001, 68, 1839. |
| [31] | (e) Hennessy, E.-T.; Betley, T.-A. Science 2013, 340, 591. |
| [31] | For some reviews, see: (f) Gramlich, E.-M.-P.; Wirges, C.-T.; Manetto, A.; Carell, T. Angew. Chem., Int. Ed. 2008, 47, 8350. |
| [31] | (g) Amblard, F.; Cho, J.-H.; Schinazi, R.-F. Chem. Rev. 2009, 109, 4207. |
| [31] | (h) Sletten, E.-M.; Bertozzi, C.-R. Acc. Chem. Res. 2011, 44, 666. |
| [31] | (i) Schilling, C.-I.; Jung, N.; Biskup, M.; Schepers, U.; Br?se, S. Chem. Soc. Rev. 2011, 40, 4840. |
| [31] | (j) Huryn, D.-M.; Okabe, M. Chem. Rev. 1992, 92, 1745. |
| [32] | For some general reviews, see: (a) Chiba, S. Synlett 2012, 21 |
| [32] | (b) Br?se, S.; Gil, C.; Knepper, K.; Zimmermann, V. Angew. Chem. 2005, 117, 5320; Angew. Chem., Int. Ed. 2005, 44, 5188. |
| [32] | (c) Driver, T.-G. Org. Biomol. Chem. 2010, 8, 3831. |
| [32] | (d) Minozzi, M.; Nanni, D.; Spagnolo, P. Chem. Eur. J. 2009, 15, 7830. |
| [33] | Yuan, Y.-Z.; Shen, T.; Wang, K.; Jiao, N. Chem. Asian J. 2013, 8, 2932. |
| [34] | Wei, X.-H.; Li, Y.-M.; Zhou, A.-X.; Yang, T.-T.; Yang, S.-D. Org. Lett. 2013, 15, 4158. |
| [35] | Barrett, A.-G.-M.; Graboski, G.-G. Chem. Rev. 1986, 86, 751. |
| [36] | Li, Y.-M.; Shen, Y.-H.; Chang, K.-J.; Yang, S.-D. Tetrahedron Lett. 2014, 55, 2119. |
| [37] | Wei, X.-H.; Wu, Q.-X.; Yang, S.-D. Synlett 2015, 26, 1417. |
| [38] | (a) Jeught, S.-V.; Stevens, C.-V. Chem. Rev. 2009, 109, 2672. |
| [38] | (b) George, A.; Veis, A. Chem. Rev. 2008, 108, 4670. |
| [38] | (c) Bialy, L.; Waldmann, H. Angew. Chem., Int. Ed. 2005, 44, 3814. |
| [38] | (d) Alexandre, F.; Amador, A.; Bot, S.; Caillet, C.; Convard, T.; Jakubik, J.; Musiu, C.; Poddesu, B.; Vargiu, L.; Liuzzi, M.; Roland, A.; Seifer, M.; Standring, D.; Storer, R.; Dousson, C.-B. J. Med. Chem. 2011, 54, 392. |
| [39] | (a) Glueck, D.-S. Top. Organomet. Chem. 2010, 31, 65. |
| [39] | (b) Baillie, C.; Xiao, J.-L. Curr. Org. Chem. 2003, 7, 477. |
| [39] | (c) Coudray, L.; Mxontchamp, J.-L. Eur. J. Org. Chem. 2008, 3601. |
| [39] | (d) Leroux, F.; Jeschke, P.; Schlosser, M. Chem. Rev. 2005, 105, 827. |
| [39] | (e) Manteau, B.; Pazenok, S.; Vors, J.-P.; Leroux, F.-R. J. Fluorine Chem. 2010, 131, 140. |
| [40] | Li, Y.-M.; Sun, M.; Wang, H.-L.; Tian, Q.-P.; Yang, S.-D. Angew. Chem., Int. Ed. 2013, 52, 3972. |
| [41] | Kong, W.-Q.; Merino, E.; Nevado, C. Angew. Chem., Int. Ed. 2014, 53, 5078. |
| [42] | (a) Leo, A.; Hansch, C.; Elkins, D. Chem. Rev. 1971, 71, 525. |
| [42] | (b) Yagupol’skii, L.-M.; Il'chenko, A.-Y.; Kondratenko, N.-V. Russ. Chem. Rev. 1974, 43, 32. |
| [42] | (c) Hansch, C.; Leo, A.; Taft, R.-W. Chem. Rev. 1991, 91, 165. |
| [42] | (d) Leroux, F.; Jeschke, P.; Schlosser, M. Chem. Rev. 2005, 105, 827. |
| [42] | (e) Manteau, B.; Pazenok, S.; Vors, J.-P.; Leroux, F.-R. J. Fluorine Chem. 2010, 131, 140. |
| [43] | Yin, F.; Wang, X.-S. Org. Lett. 2014, 16, 1128. |
| [44] | Kong, D.-L.; Du, J.-X.; Chu, W.-M.; Ma, C.-Y.; Tao, J.-Y.; Feng, W.-H. Molecules 2018, 23, 2727. |
| [45] | Zeng, Y.-F.; Tan, D.-H.; Chen, Y.; Lv, W.-X.; Liu, X.-G.; Li, Q.; Wang, H. Org. Chem. Front. 2015, 2, 1511. |
| [46] | Bruehlman, R.-J.; Verhoek, F.-H. J. Am. Chem. Soc. 1948, 70, 1401. |
| [47] | Vosburgh, W.-C.; Cogswell, S.-A. J. Am. Chem. Soc. 1943, 65, 2412. |
/
| 〈 |
|
〉 |