

铜催化氟代丙烯酸与氧杂吖丙啶的脱羧交叉偶联反应
收稿日期: 2022-08-08
修回日期: 2022-11-01
网络出版日期: 2023-01-12
基金资助
国家自然青年科学基金(22001029); 安徽省青年科学基金(2008085QB92); 安徽省高等学校自然科学重点研究(KJ2020A0708)
Copper-Catalyzed Decarboxylative Cross-Coupling of α‑Fluoroacrylic Acids with N-Tosyl Oxaziridines
Received date: 2022-08-08
Revised date: 2022-11-01
Online published: 2023-01-12
Supported by
National Natural Science Foundation of China(22001029); Natural Youth Science Foundation of Anhui Province(2008085QB92); Natural Science Research Key Project of Anhui Higher Education Institution(KJ2020A0708)
陆晓雨 , 孙晓梅 , 钮亚琴 , 王俊超 , 殷文婧 , 高梦婷 , 刘孜 , 韦正桓 , 陶庭骅 . 铜催化氟代丙烯酸与氧杂吖丙啶的脱羧交叉偶联反应[J]. 有机化学, 2023 , 43(6) : 2110 -2119 . DOI: 10.6023/cjoc202208010
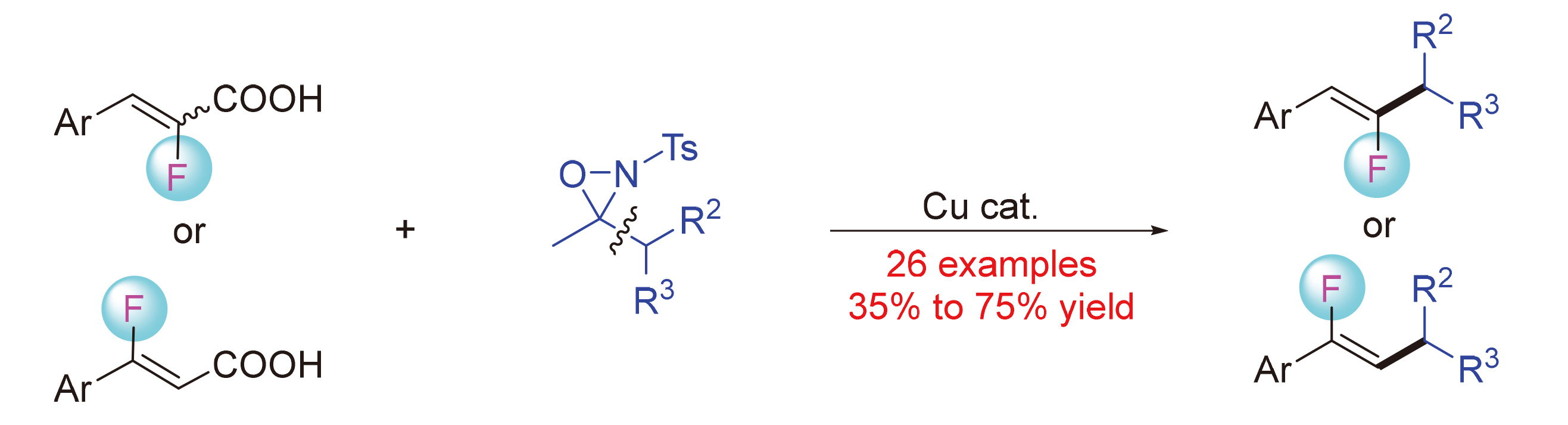
A protocol for the copper-catalyzed decarboxylative cross-coupling of α‑fluoroacrylic acids with N-tosyl oxaziridines was reported. A series of substituted α-fluoroacrylic acids, and primary, secondary and tertiary substituted oxaziridines were suitable reaction substrates. The decarboxylation reaction exhibited good functional group compatibility and excellent Z-stereoselectivity. This method provides a novel and practical strategy for the construction of monofluoroalkenes, which are key functional groups in the pharmaceutical and material sciences. In addition to α-fluoroacrylic acids, β- fluoroacrylic acid could also participate in the reaction smoothly, which provides a protocol to access various substituted monofluoroalkenes. This methodology also provides a platform for the modification of complex biologically active molecules.
| [1] | (a) Hagmann, W. K. J. Med. Chem. 2008, 51, 4359. |
| [1] | (b) Wang, J.; SánchezRoselló, M.; Acen?a, J. L.; del Pozo, C.; Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432. |
| [1] | (c) Liu, Q.; Ni, C.; Hu, J. Natl. Sci. Rev. 2017, 4, 303. |
| [1] | (d) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320. |
| [1] | (d) Liao, F.; Yu, J; Zhou, J. Chin. J. Org. Chem. 2017, 37, 2175. (in Chinese) |
| [1] | (廖富民, 余金生, 周剑, 有机化学, 2017, 37, 2175.) |
| [1] | (e) He, S.; Pi, J.; Li, Y.; Lu, X.; Fu, Y. Acta Chim. Sinica. 2018, 76, 956. (in Chinese) |
| [1] | (何世江, 皮静静, 李炎, 陆熹, 傅尧, 化学学报, 2018, 76, 956.) |
| [2] | (a) O’Hagan, D.; Deng, H. Chem. Rev. 2015, 115, 634. |
| [2] | (b) Shi, Y.; Xiao, T.; Xia, D.; Yang, W. Chin. J. Org. Chem. 2022, 42, 2715. (in Chinese) |
| [2] | (石云, 肖婷, 夏冬, 杨文超, 有机化学, 2022, 42, 2715.) |
| [2] | (c) Chen, D.; Jiang, J.; Wan, J.-P. Chin. J. Chem. 2022, 40, 2582. |
| [3] | (a) Kirsch, P. Modern Fluoroorganic Chemistry: Synthesis Reactivity Applications, Wiley-VCH, Weinheim, Germany, 2007. |
| [3] | (b) Mu?ller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881. |
| [3] | (c) Liang, T.; Neumann, C. N.; Ritter, T. Angew. Chem., Int. Ed. 2013, 52, 8214. |
| [4] | (a) Lin, G.-Q.; You, Q.-D.; Cheng, J.-F. Chiral Drugs: Chemistry and Biological Action, John Wiley & Sons, Inc., Hoboken, 2011. |
| [4] | (b) Oishi, S.; Kamitani, H.; Kodera, Y.; Watanabe, K.; Kobayashi, K.; Narumi, T.; Tomita, K.; Ohno, H.; Naito, T.; Kodama, E.; Matsuoka, M.; Fujii, N. Org. Biomol. Chem. 2009, 7, 2872. |
| [4] | (c) Meanwell, N. A. J. Med. Chem. 2018, 61, 5822. |
| [5] | (a) Reddy, V. P. Organofluorine Compounds in Biology and Medicine, Elsevier, Amsterdam, 2015. |
| [5] | (b) Kirsch, P. Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications, WileyVCH, Weinheim, 2013. |
| [5] | (c) Berger, R.; Resnati, G.; Metrangolo, P.; Weber, E.; Hulliger, J. Chem. Soc. Rev. 2011, 40, 3496 |
| [6] | (a) Drouin, M.; Paquin, J.-F. Beilstein. J. Org. Chem. 2017, 13, 2637. |
| [6] | (b) Landelle, G.; Bergeron, M.; Turcotte-Savard, M.-O.; Paquin, J.-F. Chem. Soc. Rev. 2011, 40, 2867. |
| [6] | (c) Drouin, M.; Hamel, J.-D.; Paquin, J.-F. Synthesis 2018, 50, 881. |
| [7] | (a) Xu, J.; Ahmed, E.-A.; Xiao, B.; Lu, Q.-Q.; Wang, Y.-L.; Yu, C.-G.; Fu, Y. Angew. Chem., Int. Ed. 2015, 54, 8231. |
| [7] | (b) Jiang, Z.-T.; Huang, J.; Zeng, Y.; Hu, F.; Xia, Y. Angew. Chem., nt. Ed. 2021, 60, 10626. |
| [8] | (a) Wang, C.; Liu, Y.-C.; Xu, M.-Y.; Xiao, B. Org. Lett. 2021, 23, 4593. |
| [8] | (b) Dutheuil, G.; Paturel, C.; Lei, X.; Couve-Bonnaire, S.; Pannecoucke, X. J. Org. Chem. 2006, 71, 4316. |
| [8] | (c) Andrei, D.; Wnuk, S. F. J. Org. Chem. 2006, 71, 405. |
| [8] | (d) Schneider, C.; Masi, D.; Couve-Bonnaire, S.; Pannecoucke, X.; Hoarau, C. Angew. Chem.,Int. Ed. 2013, 52, 3246. |
| [9] | (a) Koley, S.; Altman, R. A. Isr. J. Chem. 2020, 60, 313. |
| [9] | (b) Ma, T.; Chen, Y.; Li, Y.; Ping, Y.; Kong, W. ACS Catal. 2019, 9, 9127. |
| [9] | (c) Li, J.; Rao, W.; Wang, S.-Y.; Ji, S.-J. J. Org. Chem. 2019, 84, 11542. |
| [9] | (d) Yang, L.; Ji, W.-W.; Lin, E.; Li, J.-L.; Fan, W.-X.; Li, Q.; Wang, H. Org. Lett. 2018, 20, 1924. |
| [10] | (a) Tian, P.; Feng, C.; Loh, T.-P. Nat. Commun. 2015, 6, 7472. |
| [10] | (b) Kong, L.; Zhou, X.; Li, X. Org. Lett. 2016, 18, 6320. |
| [10] | (c) Zell, D.; Dhawa, U.; Mu?ller, V.; Bursch, M.; Grimme, S.; Ackermann, L. ACS Catal. 2017, 7, 4209. |
| [10] | (d) Fuchibe, K.; Mayumi, Y.; Zhao, N.; Watanabe, S.; Yokota, M.; Ichikawa, J. Angew. Chem., Int. Ed. 2013, 52, 7825. |
| [11] | (a) Thornbury, R. T.; Toste, F. D. Angew. Chem., Int. Ed. 2016, 55, 11629. |
| [11] | (b) Xiong, Y.; Huang, T.; Ji, X.; Wu, J.; Cao, S. Org. Biomol. Chem. 2015, 13, 7389. |
| [12] | (a) Lu, X.; Wang, Y.; Zhang, B.; Pi, J.-J.; Wang, X.-X.; Gong, T.-J.; Xiao, B.; Fu, Y. J. Am. Chem. Soc. 2017, 139, 12632. |
| [12] | (b) Du, H.-W.; Sun, J.; Gao, Q.-S.; Wang, J.-Y.; Wang, H.; Xu, Z.; Zhou, M.-D. Org. Lett. 2020, 22, 1542. |
| [12] | (c) Dai, W.; Shi, H.; Zhao, X.; Cao, S. Org. Lett. 2016, 18, 4284. |
| [12] | (d) Zhou, L.; Zhu, C.; Bi, P.; Feng, C. Chem. Sci. 2019, 10, 1144. |
| [12] | (e) Xie, J.; Yu, J.; Rudolph, M.; Rominger, F.; Hashmi, A. S. K. Angew. Chem., Int. Ed. 2016, 55, 9416. |
| [12] | (f) Yu, L.; Tang, M.-L.; Si, C.-M.; Meng, Z.; Liang, Y.; Han, J.; Sun, X. Org. Lett. 2018, 20, 4579. |
| [12] | (g) Yang, H.; Tian, C.; Qiu, D.; Tian, H.; An, G.; Li, G. Org. Chem. Front. 2019, 6, 2365. |
| [12] | (h) Li, J.; Lefebvre, Q.; Yang, H.; Zhao, Y.; Fu, H. Chem. Commun. 2017, 53, 10299. |
| [13] | Lu, X.-Y.; Gao, A.; Ge, M.-Y.; Xia, Z.-J.; Liu, Q.-L.; Tao, T.-H.; Sun, X.-M. J. Org. Chem. 2022, 87, 4654. |
| [14] | Lu, X.-Y.; Ge, M.-Y.; Tao, T.-H.; Sun, X.-M.; Gao, M.-T.; Bao, S.-T.; Liu, Q.-L.; Xia, Z.-J.; Xia, J. Org. Chem. Front. 2022, 9, 831. |
| [15] | Lu, X.-Y.; Chen, X.-K.; Gao, M.-T.; Sun, X.-M.; Jiang, R.-C.; Wang, J.-C.; Yu, L.-J.; Ge, M.-Y.; Wei, Z.-H.; Liu, Z. Org. Chem. Front. 2022, 9, 4712. |
| [16] | Chen, Y.; Du, J.; Zuo, Z. Chem 2020, 6, 266. |
| [17] | (a) Xiao, T.; Zhou, L.; Huang, H.; Anand, D. Synthesis 2020, 52, 1585. |
| [17] | (b) Yu, X.-Y.; Zhao, Q.-Q.; Chen, J.; Xiao, W.-J.; Chen, J.-R. Acc. Chem. Res. 2020, 53, 1066. |
| [17] | (c) Xiao, F.; Guo, Y.; Zeng, Y. F. Adv. Synth. Catal. 2021, 363, 120. |
| [17] | (d) Sivaguru, P.; Wang, Z.; Zanoni, G.; Bi, X. Chem. Soc. Rev. 2019, 48, 2615. |
| [17] | (e) Xiao, W.; Wu, J. Chin. Chem. Lett. 2020, 31, 3083. |
| [17] | (f) Yu, X.-Y.; Chen, J.-R.; Xiao, W.-J. Chem. Rev. 2021, 121, 506. |
| [17] | (g) Lu, X.-Y.; Liu, C.-C.; Jiang, R.-C.; Yan, L.-Y.; liu, Q.-L.; Wang, Q.-Q.; Li, J.-M. Chem. Commun. 2020, 56, 14191. |
| [17] | (h) Lu, X.-Y.; Xia, Z.-J.; Gao, A.; Liu, Q.-L.; Jiang, R.-C.; Liu, C.-C. J. Org. Chem. 2021, 86, 8829. |
| [18] | (a) Nguyen, B.-N.; Cao, H.-T. Eur. J. Org. Chem. 2019, 20196, 5912. |
| [18] | (b) Matsumoto, A.; Nguyen, B.-N.; Honda, T.; Sakamoto, R.; Huang, X.; Sakaki, S.; Maruoka, K. Chem. Asian J. 2021, 16, 282. |
| [19] | (a) Chen, L.; Zhang, L.; Yan, G.; Huang, D. Asian J. Org. Chem. 2020, 9, 842. |
| [19] | (b) Lu, X.-Y.; Li, J.-S.; Wang, S.-Q.; Zhu, Y.-J.; Li, Y.-M.; Yan, L.-Y.; Li, J.-M.; Wang, J.-Y.; Zhou, H.-P.; Ge, X.-T. Chem. Commun. 2019, 55, 11123. |
/
| 〈 |
|
〉 |